Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Arthrofibrosis implies a loss of knee flexion, extension, or both compared with the contralateral normal knee.
Primary arthrofibrosis is caused by an exaggerated inflammatory response to an injury or surgical procedure followed by the production of fibroblastic cells and an increase in the deposition of extracellular matrix proteins. Proliferative scar tissue or fibrous adhesions form within the joint, which can be either localized or diffuse.
Incidence of arthrofibrosis following ligament reconstructive surgery varies.
Wide disparity exists among authors regarding the treatment of this problem.
Limitations in knee motion may occur owing to reasons other than arthrofibrosis:
Mechanical block from a displaced bucket-handle meniscus tear.
Impinged or improperly placed ACL graft.
Cyclops lesion.
Improper ACL graft tensioning.
Contracture of posterior capsular structures.
If not resolved, these problems may result in the development of secondary arthrofibrosis.
Most individuals have some amount of hyperextension to allow the normal “screw-home” mechanism to occur: knee stabilized in extension during the stance phase, with the quadriceps muscle relaxed.
Normal knee flexion averages 140 degrees in men and 143 degrees in women.
Patients not involved in athletics tolerate small flexion deficits. Significant flexion deficits <90 degrees cause problems with squatting, stair-climbing, and sitting.
Loss of just 5 degrees of extension may produce a flexed-knee gait, fatigue the quadriceps muscle, and cause patellofemoral pain.
Prevention of knee arthrofibrosis is paramount and preferred over the current treatment options for this complication.
Loss of a normal range of knee motion following an injury or ligament reconstruction is a potentially devastating complication. Many investigations and publications have appeared since the early 1990s regarding this problem, and it continues to be one of the most frequently reported complications after surgical procedures such as autologous chondrocyte implantation (ACI), knee ligament reconstructions, and total knee arthroplasty (TKA). Although the definition and use of the term arthrofibrosis varies among authors, it implies a loss of knee flexion, extension, or both compared with the contralateral normal knee. Stephenson and associates estimated that the number of patients who undergo major knee surgery in the United States that are subsequently affected by arthrofibrosis is at least 85,000 a year. Of these, 21,000 are at risk of requiring additional surgery, and 54,000 may have an unsuccessful outcome of treatment for this complication.
Primary arthrofibrosis is caused by an exaggerated inflammatory response to an injury or surgical procedure, followed by the production of fibroblastic cells and an increase in the deposition of extracellular matrix proteins. Proliferative scar tissue or fibrous adhesions form within the joint, which can result in either localized or diffuse involvement of all of the compartments of the knee and the extra-articular soft tissues ( Fig. 38-1 ). In the most severe cases, dense scar tissue obliterates the normal peripatellar recesses, suprapatellar pouch, intercondylar notch, and articular surfaces. Formation of dense scar tissue in the infrapatellar region may result in a patella infera and permanent limitation of normal knee flexion and extension. The consequent pain and restricted knee motion resulting from arthrofibrosis may lead to disabling events, including severe quadriceps atrophy, loss of patellar mobility, patellar tendon adaptive shortening, patella infera, and articular cartilage deterioration.
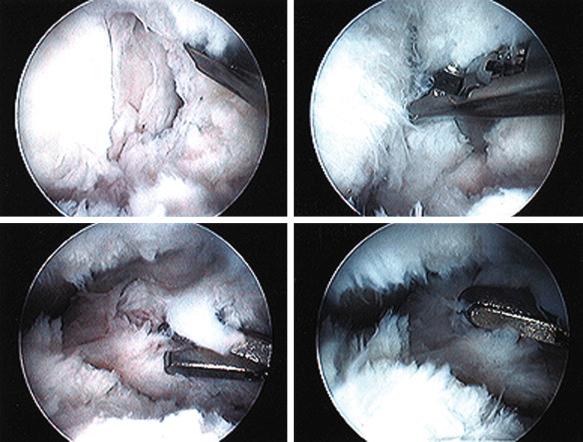
The incidence of arthrofibrosis after knee ligament reconstructive surgery varies according to several factors. In our experience, two of the most commonly noted factors are knee dislocations and major concurrent operative procedures ( Table 38-1 ). Studies show wide disparity among authors regarding the treatment of this problem, especially the postoperative time that intervention is warranted and what type of treatment should be implemented for losses of knee extension and flexion and patella mobility.
| Study | Operation | Patients ( n ) | Type of Ligament Graft, Concomitant Procedures | Motion Problems Requiring Therapeutic Intervention (%) |
|---|---|---|---|---|
| JBJSA, 1990 | ACL reconstruction | 40 | Allograft, acute | 17 |
| JBJSA, 1991 | ACL reconstruction | 64 | Allograft only, chronic | 8 |
| 40 | Allograft + EA, chronic | 18 | ||
| JBJSA, 1992 | ACL reconstruction | 66 | Allograft only, chronic | 8 |
| 49 | Allograft + LAD, chronic | 2 | ||
| CORR, 1992 | ACL reconstruction | 90 | Allograft only | 4 |
| 52 | Allograft + EA | 10 | ||
| 52 | Allograft + meniscus repair | 12 | ||
| 13 | Allograft + MCL repair | 23 | ||
| AJSM, 1995 | ACL reconstruction | 34 | Allograft + MCL repair | 26 |
| 12 | Allograft + MCL conservative treatment | 17 | ||
| AJSM, 1997 | ACL reconstruction | 30 | PT autograft, acute | 10 |
| 57 | PT autograft, chronic | 2 | ||
| AJSM, 1997 | ACL reconstruction | 47 | PT autograft, men | 4 |
| 47 | PT autograft, women | 6 | ||
| AJSM, 1997 | Multiple ligament reconstruction for knee dislocation | 11 | Allograft + autograft; all ACL + PCL | 45 |
| 6 | Also + MCL | |||
| 6 | Also + PL | |||
| KSSTA, 2000 | ACL reconstruction | 219 | PT autograft only | 6 |
| 37 | PT autograft + PL procedure | 5 | ||
| 194 | PT autograft + meniscus repair | 8 | ||
| 17 | PT autograft + patellar realignment | 18 | ||
| 9 | PT autograft + MCL repair | 22 | ||
| AJSM, 2000 | HTO, closing wedge | 38 | PT allograft or PT autograft | 0 |
| 3 | HTO + ACL | 0 | ||
| JBJSA, 2004 | Meniscus transplantation | 18 | Transplant only | 0 |
| 16 | Transplant + OAT | 12 | ||
| 4 | Transplant + knee ligament procedure | 50 | ||
| JBJSA, 2005 | PCL reconstruction | 11 | Isolated | 18 |
| 8 | PCL + other knee ligament | 25 | ||
| AJSM, 2006 | HTO, opening wedge | 49 | HTO only | 0 |
| 4 | HTO + ACL | 0 | ||
| 2 | HTO + OAT | 0 | ||
| AJSM, 2007 | PL anatomic reconstruction | 2 | PL only | 0 |
| 12 | PL + knee ligament procedure | 17 |
Note that limitations in knee flexion and extension may occur because of reasons other than arthrofibrosis. Any acute injury causing pain and swelling may cause a short-term loss of knee motion, which usually resolves as symptoms subside. Other factors influencing the restoration of knee motion include a mechanical block from a displaced bucket-handle meniscus tear, an impinged or improperly placed anterior cruciate ligament (ACL) graft, a cyclops lesion, improper graft tensioning that constrains normal knee joint motion, and graft fixation at 30 degrees or more of extension. In addition, contracture of the posterior capsular structures may limit knee extension. If not resolved, these problems may result in the development of what is termed secondary arthrofibrosis , complicating the course of treatment.
Normal knee motion varies, but most individuals have some amount of hyperextension, which averages 5 degrees in men and 6 degrees in women. This degree of hyperextension is required so that the normal “screw-home” mechanism that allows the knee to be stabilized in extension during the stance phase may occur, with the quadriceps muscle relaxed. Soucie and colleagues provided the following ranges of knee motion, according to gender and age from a sample of 674 healthy subjects: (1) ages 9 to 19 years: 2.4 to 142.3 degrees for girls and 1.8 to 142.2 degrees for boys, (2) ages 20 to 44 years: 1.6 to 141.9 degrees for women and 1.0 to 137.7 degrees for men, and (3) ages 45 to 69 years: 1.2 to 137.8 degrees for women and 0.5 to 132.9 degrees for men.
It is generally assumed that at least 125 degrees of knee flexion is required for activities of daily living. Rowe and coworkers measured knee kinematics during functional activities in a group of 20 subjects aged 49 to 80 years and reported that 90 degrees of flexion is required for gait and slopes, 90 to 120 degrees for stairs and sitting, and 135 degrees for bathing. Patients not involved in athletics tolerate small flexion deficits. Significant flexion deficits of less than 90 degrees cause problems with squatting, stair climbing, and sitting. Athletes have a poorer tolerance for even minor losses of flexion, which affect running and jumping. Cosgarea and associates reported that deficits of 10 degrees or more of flexion were associated with decreased running speed.
All patients, regardless of their activity level, have greater problems with loss of knee extension. A loss of just 5 degrees of extension may produce a flexed-knee gait, fatigue the quadriceps muscles, and cause patellofemoral pain. Limitation of more than 20 degrees may cause a functional limb-length discrepancy. Many years ago, Perry and colleagues demonstrated the effect of a loss of extension on joint contact pressures, quadriceps muscle activity, and fatigue. These investigators measured the extensor forces required to stabilize the flexed knee during simulated weight bearing in cadaver specimens. The quadriceps force required to stabilize the knee was 75% of the load on the head of the femur at 15 degrees of flexion, which increased to 210% at 30 degrees of flexion and to 410% at 60 degrees of flexion. The quadriceps force was equal to 20% of average maximum quadriceps strength at 15 degrees of flexion but increased to 50% at 30 degrees of flexion. Surface pressure in the tibiofemoral and patellofemoral joints also increased with greater amounts of knee flexion.
It has become evident that the prevention of knee arthrofibrosis is paramount and preferred over using the currently available treatment options for this complication. This chapter discusses the risk factors and preventive measures for loss of knee motion after knee injury and surgery. In addition, conservative medical and therapeutic treatment intervention strategies are discussed that are usually successful in resolving a transient limitation of knee motion if initiated early postoperatively. Surgical procedures for severe cases of restriction of knee motion are presented. Our clinical studies involving 650 ACL-reconstructed knees are included to provide support for treatment recommendations.
Terms used to describe limitation knee motion: arthrofibrosis, flexion contracture, ankylosis, infrapatellar contracture syndrome, motion loss.
The term arthrofibrosis describes a specific cause for a limitation of knee motion, which is the formation of diffuse scar tissue or fibrous adhesions within a joint.
Classification systems describe limitations in knee motion, based either on the anatomic location of scar tissue and adhesions, or on the amount of knee motion in the affected joint.
Our classification system is based on the anatomic sites where scar tissue, adhesions, and adaptive shortening of soft tissues occur.
A variety of terms have been used to describe, define, or classify a limitation of knee flexion and extension, including arthrofibrosis, flexion contracture, ankylosis, infrapatellar contracture syndrome, and motion loss. Some of the terms indicate a specific process, but others simply imply a limitation of motion compared with the contralateral normal knee. Ankylosis is one such generic term that indicates stiffness of joints that occurs for any reason and may represent loss of knee flexion, extension, or both. The term motion loss is another general phrase used to describe deviations from the amount of flexion and extension compared with the contralateral normal knee. Conversely, arthrofibrosis describes a specific cause for a limitation of knee motion, which is the formation of diffuse scar tissue or fibrous adhesions within a joint. Flexion contracture indicates a loss of extension attributable to any cause and is accompanied by a relative shortening of the posterior soft tissues (in either the capsule or muscles). Petsche and Hutchinson state that arthrofibrosis and flexion contracture are general terms, and it is necessary to indicate a specific cause for loss of motion.
Classification systems have been proposed by various authors to describe limitations in knee motion, based either on the anatomic location of scar tissue and adhesions or on the amount of knee motion in the affected joint ( Table 38-2 ). Sprague and coworkers first introduced a system based on the arthroscopically observed location of scar tissues and adhesions in 24 patients who had a severe limitation of knee motion. Paulos and associates described three stages of knee motion restrictions caused by infrapatellar contracture syndrome. Del Pizzo and colleagues and Shelbourne and coworkers developed classification systems based on the deviation of knee motion compared with the opposite knee. Blauth and Jaeger described a system based on the total arc of motion in the affected knee.
| Study | Classification System |
|---|---|
| Sprague et al (1982) |
|
| Del Pizzo et al (1985) | Based on deviation from full extension and amount of flexion present:
|
| Paulos et al (1987) | Three stages of infrapatellar contracture syndrome:
|
| Blauth & Jaeger (1990) | Based on total arc of motion:
|
| Shelbourne et al (1996) | Based on deviation from full flexion and extension of opposite, normal knee:
|
We have developed a classification system based on the anatomic sites at which scar tissue, adhesions, and adaptive shortening of soft tissues occur ( Fig. 38-2 and Table 38-3 ). This system is advantageous because it highlights the areas that must be addressed surgically, as is discussed.
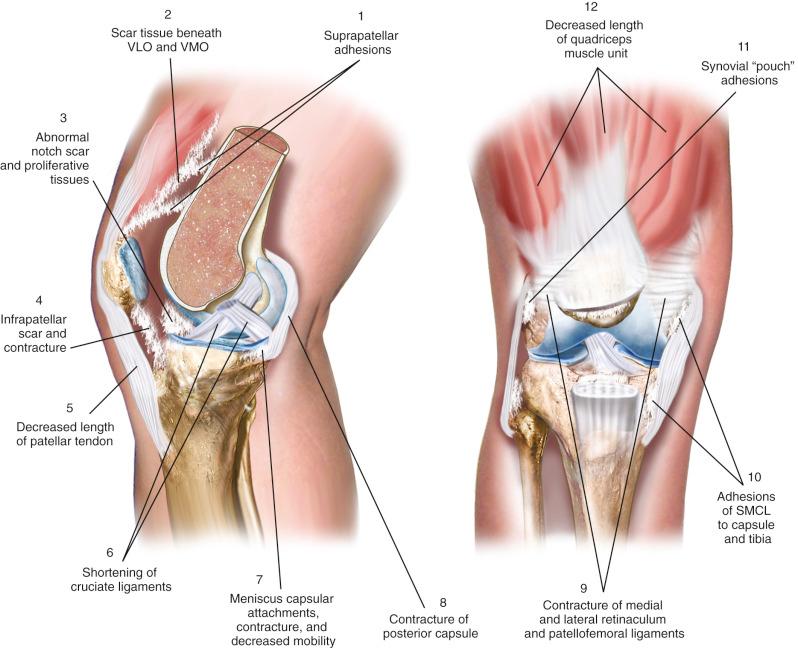
| Loss of Flexion |
| Quadriceps Muscle |
|
| Suprapatellar Pouch |
|
| Medial and Lateral Capsular Pouches |
|
| Patellar Retinaculum and Associated Ligaments |
|
| Patellar Tendon |
|
| Medial and Lateral Extraarticular Ligament Structures |
|
| Loss of Extension |
| Posterior Capsule |
|
| Femoral Notch |
|
| Hamstrings Muscle |
|
| Cruciate Ligaments |
|
Severity of injury: knee dislocations increase risk for developing motion complications. Surgery is delayed in all cases except those with ruptures to the posterolateral structures.
Preoperative issues: performing knee ligament reconstruction within a few weeks of the injury or before the resolution of swelling, pain, quadriceps muscle atrophy, abnormal gait mechanics, and motion limitations correlates with postoperative knee motion problems.
Technical factors at surgery: improper ACL graft placement, overtensioning graft at surgery, performing concurrent MCL repair with ACL reconstruction.
Postoperative course and rehabilitation: immobilization is detrimental to all of the knee joint structures and may result in a permanent limitation of knee motion, prolonged muscle atrophy, patella infera, and articular cartilage deterioration. Begin knee motion, patellar mobilization, muscle-strengthening exercises the day after surgery. Treat joint effusion immediately.
Postoperative infection, complex regional pain syndromes, and reflex sympathetic dystrophy may cause knee motion problems.
A variety of factors appear to influence the requirement for treatment intervention for knee motion limitations ( Table 38-4 ) and the final amount of knee flexion and extension gained after ligament reconstruction. These factors are related to the severity of the injury, the timing of surgery, preoperative treatment, technical aspects of the ligament reconstruction, and the postoperative course and rehabilitation. Although it remains uncertain why some knees are more likely to form an abnormal scar response to trauma and surgery than others, knowledge of these factors may help reduce this problem.
|
Patients who sustain knee dislocations are at increased risk for developing motion complications ( Fig. 38-3 ). These injuries frequently occur from high-energy accidents that produce extreme soft tissue swelling and edema, hematomas, muscle damage, and other multiple trauma that must be resolved before consideration of knee soft tissue reconstructions. Early operative intervention when possible is advised for multiple ligament injuries that involve the lateral and posterolateral structures where acute repair and augmentation procedures are performed (see Chapter 17 ). For all other dislocations, surgery is delayed with the limb immobilized in a posterior plaster splint or bivalved cast with a posterior pad to prevent posterior tibial subluxation. Even in these severe knee injuries, it is possible to start immediate range of motion and prevent scar tissue formation that may compromise the outcome of a subsequent ligament reconstruction. Too often, these serious knee joint injuries are not treated with an early motion and function program. When surgical treatment is elected, the reconstructive and repair procedures of torn ligaments, capsular structures, and menisci are performed in a manner that allows immediate knee motion to be instituted postoperatively.
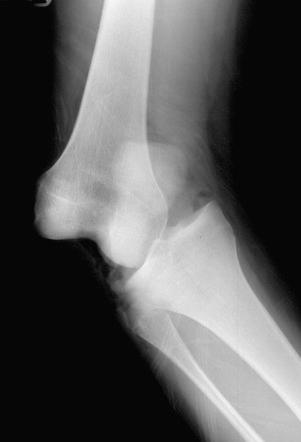
Performing anterior cruciate ligament (ACL) and other knee ligament reconstruction within a few weeks of the injury or before the resolution of swelling, pain, quadriceps muscle atrophy, abnormal gait mechanics, and motion limitations has been noted by many authors to correlate with postoperative knee motion problems. Shelbourne and associates noted an increased rate of arthrofibrosis in patients who underwent acute ACL reconstruction (within 1 week of the injury) compared with those in whom the reconstruction was delayed for at least 21 days. Similar findings were reported by Wasilewski and colleagues, who noted that arthrofibrosis was found in 22% of acutely reconstructed knees versus 12.5% of knees reconstructed with chronic ACL deficiency. Mohtadi and coworkers found that ACL reconstructions performed less than 6 weeks postinjury had a higher rate of knee stiffness than those done more than 6 weeks post-injury (11% and 4%, respectively).
Mauro and associates followed 229 patients who underwent ACL reconstruction to determine the incidence of loss of knee extension at 4 weeks after surgery. Fifty-eight patients (25%) had not regained full extension equal to the contralateral knee. When compared with the group of patients who had regained full extension, the group that had a limitation were noted to have had a shorter mean time from injury to surgery (60 days and 93 days, respectively; P < .05), a greater preoperative loss of extension (4 and 1 degrees, respectively; P < .05), and a greater percentage that underwent autograft reconstruction than allograft reconstruction (28.5% and 14%, respectively; P < .05).
Although Bach and colleagues did not observe a significant difference in knee motion complications between acute and chronic ACL-reconstructed knees, these authors stressed the need to delay surgery until patients achieved 120 degrees of knee motion and swelling had resolved. The authors found “no advantage to performing urgent acute ACL reconstruction.” Sterett and coworkers found no association between surgical timing and postoperative motion problems. However, in their series, all patients had achieved at least 0 to 120 degrees, had good quadriceps control, and could perform a straight-leg raise without an extensor lag before undergoing surgery. Eight percent of the patients in this investigation's acute subgroup required arthroscopic debridement of scar tissue. Meighan and associates also found no advantage in performing early ACL reconstruction and noted an increased rate of complications in patients who underwent surgery within 2 weeks of the injury compared with those who had surgery between 8 and 12 weeks postinjury. The delayed group had a faster return of knee motion and quadriceps strength.
Other authors have not found a higher rate of postoperative motion problems after acute ACL reconstruction. However, it appears that delaying surgery until knee motion is regained, swelling is resolved, and a good quadriceps contraction is demonstrated is advantageous for decreasing the risk of postoperative arthrofibrosis. The inflammatory response to the initial injury varies among patients. Although some have little effusion and swelling, others have an exaggerated inflammatory response characterized by pain, soft tissue edema, and redness and increased warmth to tissues surrounding the knee. These knees are placed into an initial conservative treatment program to resolve these problems first and are also carefully monitored after ACL reconstruction for a similar exaggerated inflammatory reaction postoperatively. Knees with isolated ACL ruptures that have little swelling and demonstrate normal motion early after injury may be considered for earlier reconstruction. In addition, knees with multiple ligament ruptures that include the posterolateral structures are also candidates for early surgical repair, as discussed in Chapter 17 .
Improper ACL graft placement has been frequently noted to cause loss of knee motion. On the tibia, grafts placed anterior to the native ACL insertion result in impingement on the roof of the intercondylar notch when the knee is extended. Yaru and colleagues recommended that, with passive extension, there should be a 3-mm clearance between the anterior portion of the intercondylar notch and the graft to prevent impingement. Grafts placed lateral to the insertion site impinge on the lateral wall of the notch. In addition, Romano and coworkers found that ACL grafts placed too far anteriorly in the tibial tunnel can cause knee extension deficits, and grafts placed too far medially in the tibial tunnel can cause knee flexion deficits. On the femur, an excessive anterior graft placement causes deleterious forces on the graft, leading to limitations in flexion and potential graft failure ( Fig. 38-4 ).
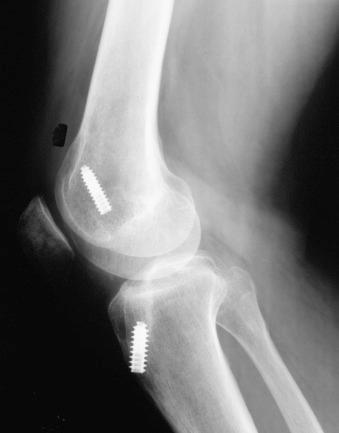
Overtensioning ACL grafts may lead to abnormal knee kinematics. In addition, the degree of knee extension during graft tensioning and fixation may affect postoperative motion. Austin and associates noted in a cadaver study that the amount of graft tension (44 N or 89 N) did not effect knee extension; however, tensioning the graft in knee flexion was associated with extension deficits. The authors reported that grafts tensioned and fixed at 30 degrees of flexion had more than a 12 degree increase in knee flexion after ACL reconstruction compared with those tensioned and fixed at 0 degrees of flexion. From a two-part biomechanic and clinical study, Nabors and colleagues suggested that grafts tensioned in full extension result in a low incidence of knee motion loss. In their series of 57 patients who underwent patellar tendon autograft ACL reconstruction, only one patient had a mild (5 degrees) loss of extension.
Concurrent medial collateral ligament (MCL) repair with ACL reconstruction has been associated with an increased risk of knee arthrofibrosis. Harner and associates postulated that concurrent MCL primary repair may cause a limitation of knee motion attributable to the disruption of the medial capsule because the procedure does not restore the normal tissue planes, resulting in scar formation and a heightened pain response. Our studies and the studies of Shelbourne and coworkers led to the recommendation many years ago to treat the majority of combined ACL/MCL ruptures conservatively. This allows healing of the medial-side injury followed by ACL reconstruction in appropriately indicated patients (see Chapter 19 ). There are exceptions to this rule, and any surgical procedure on the medial side of the knee joint should be carefully observed postoperatively for a limitation of motion or, in acute knee injuries, the development of heterotopic soft tissue ossificiation requiring treatment.
There is consensus in the literature that immobilization is detrimental to all of the knee joint structures and may result in a permanent limitation of knee motion, prolonged muscle atrophy, patella infera, and articular cartilage deterioration. Early knee joint motion decreases pain and postoperative joint effusions, aids in the prevention of scar tissue formation and capsular contractions that can limit normal knee flexion and extension, decreases muscle disuse effects, maintains articular cartilage nutrition, and benefits the healing ACL graft. Modern rehabilitation programs incorporate immediate knee motion and muscle-strengthening exercises the day after surgery, both of which have been shown to be safe and not deleterious to healing grafts (see Chapter 10, Chapter 11, Chapter 12 ). Importantly, the immediate motion program must include patellar mobilization (inferior, superior, medial, and lateral directions) to avoid an infrapatellar contracture.
Patient compliance with postoperative rehabilitation is essential in recovering full knee motion. In our experience, the small percentage of patients who have permanent restrictions in extension or flexion have often been unwilling to perform the required motion, strengthening, and patellar mobilization exercises postoperatively. In addition, in instances in which an early postoperative limitation of motion has been recognized, these patients are also unwilling to undergo treatment recommendations such as overpressure exercises, extension casts, and other modalities that are usually effective in resolving these problems. Thus in a majority of cases, the inflammatory and fibrotic response that follows surgery and initially limits knee motion is treatable if no delay occurs in instituting a gentle motion and overpressure program along with appropriate antiinflammatory medications. There is a distinct group of patients, in the range of 1% to 2%, who demonstrate a pathologic exaggerated fibrous tissue proliferative response most likely from a genetic basis, in which the treatment is prolonged and may not be successful. This unique group of patients is discussed in a later section of this chapter.
Significant lower extremity muscle atrophy represents an unresolved problem after ACL reconstruction because the magnitude of quadriceps atrophy and strength loss may exceed 20% to 30% in the first few months postoperatively. Prolonged quadriceps atrophy may affect the ability to regain normal knee motion and maintain knee extension. Chronic swelling may also cause a limitation of knee motion by inhibiting the function of the quadriceps muscle. It is important that a knee joint effusion be treated to lessen its deleterious effect on quadriceps function.
Infection may result in loss of knee motion after ACL reconstruction. The rule followed in our clinical practice is to always consider first that an exaggerated inflammatory response with joint swelling, synovitis, and early limitation of joint motion is caused by a joint infection until proved otherwise. Even when an infectious process appears to have been excluded, a knee joint that does not respond to the gentle modalities to regain knee motion or that has continued pain or lack of patellar mobility should undergo repeat aspiration, cell count, culture, and diagnostic studies. In our experience, the most severe cases of arthrofibrosis that initially were presumed to be caused by a genetic abnormality were subsequently proven to have an occult unrecognized infectious etiology. In patients with an established infectious process, the principles of treatment have been discussed (see Chapter 7 ), and it is important that gentle motion and overpressure programs continue to maintain the joint motion. Even in knees with swelling of soft tissues and the initial host response to an infection, it is possible within days after arthroscopic lavage, debridement, and appropriate antibiotics to initiate motion exercises and prevent a flexion contracture or patella infera. Although aggressive management of this complication usually leads to resolution of the infection, studies report permanent loss of extension and flexion in the majority of patients treated which may be avoidable.
Recent reports by Csintalan and associates and Nwachukwu and coworkers identified female gender as a significant risk factor for the development of arthrofibrosis after ACL reconstruction. Csintalan and associates studied a cohort of 14,522 primary ACL reconstructions and reported a hazard ratio (HR) for reoperations related to arthrofibrosis of 2.48 for women compared with men ( P <.001). In addition, these authors reported any prior surgery was also a significant risk factor (HR, 3.02; P = .005). Nwachukwu and coworkers studied 933 consecutive ACL reconstructions performed in patients aged 7 to 17 years and reported an overall arthrofibrosis rate of 8.3%. Statistically significant risk factors were female gender ( P = .0001), patient aged 16 to 18 years ( P = .007), patellar tendon autograft ( P = .03), and concomitant meniscal repair ( P = .007).
Complex regional pain syndromes and reflex sympathetic dystrophy may cause knee motion problems as a result of quadriceps atrophy, chronic swelling, and an increased sensitivity to pain. These issues are discussed in detail in Chapter 40 . Appropriate management using a variety of medical specialists is essential to diagnose and treat these problems.
Cytokines or growth factors responsible for constant signaling occurring both between local cells and within cells.
TGF-β is a key cytokine that initiates and ends process of tissue repair. Its actions enhance the deposition of extracellular matrix and regulate the actions of other cytokines.
Overexpression of TGF-β1 results in progressive accumulation of matrix in tissues and an elevated number of myofibroblasts, leading to fibrosis.
Specific ASMA-expressing myofibroblasts generate tissue contraction, are responsible for collagen overproduction during fibrotic diseases. TGF-β is capable of upregulating ASMA and collagen in fibroblasts.
ASMA is involved in scar formation and scar tissue contraction during the course of primary arthrofibrosis.
Collagen type VI could play a contributory role in the deposition of extracellular matrix that leads to arthrofibrosis.
Primary arthrofibrosis may be the result of an immune response.
The effectiveness of various agents such as decorin, hyaluronic acid, chitosan, mitomycin C, and various IL-1 receptor antagonists (anakinra), as well as low-dose irradiation, in high-level human studies for early or later-stage arthrofibrosis has not been investigated to date.
It is hopeful that chemical agents will become available to treat fibrotic response to injury by controlling elevated levels of myofibroblast up regulators and fibrogenic growth factors.
In 1972, Enneking and Horowitz published one of the first studies on the pathophysiology of a joint contracture after immobilization in humans. The investigators described in a series of case reports the presence of fibrofatty connective tissue in the infrapatellar fat pad, suprapatellar pouch, and posterior recesses of the knee joint with eventual obliteration of the joint cavity. Over time, this tissue was replaced with mature fibrous connective tissue.
Tissue homeostasis is maintained by the normal level of cell growth and proliferation along with the production and turnover of the extracellular matrix. Polypeptides known as cytokines or growth factors are responsible for the constant signaling that occurs both between local cells (paracrine) and within cells (autocrine). Cytokines exist as small proteins that signal cells in response to injury and infection. One cytokine, interleukin-1 (IL-1), has been found to stimulate platelets and macrophages to release a variety of growth factors, including transforming growth factor-β (TGF-β). TGF-β is thought to be one of the factors (among others ) mediating Dupuytren's contracture. Cytokines regulate all aspects of tissue remodeling and may act positively or negatively on tissue damage. This variability results in a wide range of potential responses to injury among patients.
TGF-β, released by platelets, tendon fibroblasts, and the joint capsule, has been identified as a key cytokine that both initiates and ends the process of tissue repair. Although other growth factors are involved in tissue remodeling, TGF-β is unique in its actions that enhance the deposition of extracellular matrix and regulate the actions of other cytokines. An overexpression of TGF-β 1 results in progressive accumulation of matrix in tissues and an elevated number of myofibroblasts, leading to fibrosis. Border and Noble believe that repeated injury or trauma may also cause autoinduction of TGF-β beyond normal levels, creating a “chronic, vicious circle of TGF-β overproduction” resulting in tissue fibrosis that may also occur in organ systems such as the kidney, liver, and lung. Investigators have documented that elevated levels of myofibroblasts and TGF-β occur rapidly, within 2 weeks following injury in experimental models, and that these changes are similar to those observed in humans with chronic elbow joint contractures.
Zeichen and coworkers compared tissue samples of 13 patients with arthrofibrosis obtained a mean of 16 months after the inciting knee ligament surgery with those of 8 patients undergoing primary ACL surgery. Histologic examination from the patients with arthrofibrosis showed marked synovial hyperplasia, infiltration of inflammatory cells, and vascular proliferation with an increased level of collagen types VI and III. None of the control subjects demonstrated these findings. These authors noted that this confirmed the observations of Murakami and associates regarding the association between an inflammatory process and proliferation of fibroblasts. The suggestion was made that collagen type VI could play a contributory role in the deposition of extracellular matrix that leads to arthrofibrosis.
There is evidence that during the exaggerated fibrotic response that occurs in knee arthrofibrosis, a certain amount of tissue contraction or shrinkage occurs. Specific α-smooth muscle actin (ASMA)-expressing myofibroblasts generate tissue contraction during wound healing and are responsible for collagen overproduction during fibrotic diseases. TGF-β is capable of up regulating ASMA and collagen in fibroblasts.
Unterhauser and associates measured the amount of ASMA-containing fibroblastic cells in arthrofibrotic tissue to determine whether the number of these cells was increased over that found in normal tissue. Tissue biopsies were obtained from 9 patients who underwent surgery for arthrofibrosis that had developed after ACL reconstruction. The patients all had primary arthrofibrosis and underwent the debridement procedure between 4 and 12 months after the ACL reconstruction. Tissue samples were also taken from 5 patients who underwent ACL reconstruction for chronic ligament deficiency and from 8 patients who had follow-up arthroscopy after ACL reconstruction for meniscus pathology; all 13 of these control patients showed no signs of arthrofibrosis. Significantly higher expressions of ASMA-positive fibroblastic cells were found in the collagen bundles and remaining fatty tissue in the study group compared with both control groups (23.4% of the total cell count vs 2.3% and 10.8%, respectively; P < .001). These authors concluded that this cell type is involved in scar formation and scar tissue contraction during the course of primary arthrofibrosis. They speculated that the expression of ASMA is most likely variable according to the time from the onset of the disease to tissue biopsy and probably decreases as the disease progresses.
Unterhauser and associates also reported that the fatty tissues in the arthrofibrotic knees were replaced by a dense collageneous network with parallel fiber orientation. A significantly higher total cell count and lower vessel density were measured in the arthrofibrotic knees compared with the control group that underwent ACL reconstruction. Similar findings were noted by Mariani and colleagues who biopsied 17 knees with arthrofibrosis that occurred after a variety of surgical procedures. The tissue samples were obtained less than 6 months from the onset of stiffness in 9 patients, from 6 to 12 months in 5 patients, and more than 12 months in 4 patients. The histological evaluations demonstrated collagen-producing fibroblastic tissue with differing amounts of cellularity and vascularization. This study supports the hypothesis that arthrofibrotic tissue undergoes progressive remodeling and acquires the characteristics of mature collagen, all within the first 6 months after the onset of joint stiffness. There was no association between the amount of knee motion and time from surgery or the degree of tissue maturation. The severity of loss of motion was associated only with the location and amount of adhesive tissue in the knee joint.
Many investigations have suggested that antifibrotic agents may be of potential use in preventing or combating arthrofibrosis because they inhibit the TGF-β signaling pathway. However, the effectiveness of various agents such as decorin, hyaluronic acid, chitosan, mitomycin C, and various IL-1 receptor antagonists in high-level human studies for early or later-stage arthrofibrosis has not been investigated to date.
The use of powerful antiinflammatory medications, typically used for rheumatoid arthritis, may be useful in the future in patients who do not respond to high doses of oral corticosteroids. Magnussen and coworkers reported a series of four patients who were treated with an intraarticular injection of Kineret (anakinra), an IL-1 receptor antagonist, to treat persistent postoperative inflammation and scarring after ACL reconstruction. The injections were performed on postoperative days 36, 38, 41, and 97. The patients regained normal or nearly normal knee motion; three did not undergo other procedures but one did require debridement of scar tissue from the anterior compartment. Brown and associates also used intraarticular anakinra injections in eight patients, four of whom had chronic refractory arthrofibrosis and four of whom had “limited” arthrofibrosis with varying results. There was no control group or validated rating system used, and range of motion and joint effusion were based on “clinician estimate.” With so few data published to date, no recommendation can be made regarding the use of Kineret for knee arthrofibrosis at present.
Decorin is a human proteoglycan that has been shown in many experimental models to inactivate the effects of TGF-β and have a beneficial effect in arthrofibrotic tissues. Several years ago, Fukushima and coworkers reported that injection of decorin improved both the muscle structure and function of lacerated muscle to near-complete recovery in mice. More recently, Zhang and associates found that decorin reduced the effect of TGF-β in regard to intrinsic cell expansion in cultured synovial fluid explants from osteoarthritic knees. Abdel and colleagues assessed the effects of four intraarticular injections of decorin that were performed 8 weeks after an operation that was done to induce a knee joint contracture in rabbits. There was no significant effect on improving the flexion contractures. However, genetic analysis showed a significant alteration in several fibrotic genes that were likely associated in the arthrofibrosis process. These authors suggested that earlier injection might produce better results in terms of preventing joint contractures. Several other experimental models have demonstrated the effectiveness of decorin in inhibiting adhesion formation in intraarticular adhesions, skeletal muscle injury, kidneys, and lungs. There are no current human trials on the use of decorin for the prevention or treatment of knee arthrofibrosis.
Emami and coworkers investigated the effects of bevacizumab (Avastin), an agent that inhibits vascular endothelial growth factor, on preventing arthrofibrosis after surgery in a rabbit model. Given in either one or two injections, these authors reported reduced joint fibrosis, with better clinical and histological findings in the two-injection group. This was especially evident in the statistically significant reduced number of fibroblasts ( P = .002), inflammatory cells ( P = .01), vascularity ( P < .05), and collagenous matrix deposition ( P < .05).
Liang and colleagues applied topical hydroxycamptothecin, a chemotherapeutic drug, to rabbit knees in an attempt to prevent intraarticular adhesions after surgery in which cortical bone was removed from the femoral condyles. After 4 weeks, there was a significant reduction of adhesions in knees that received 1.0 mg/mL or 2.0 mg/mL compared with lower doses and controls ( P <.05). Another well-known chemotherapeutic agent, mitomycin C, was investigated by Yan and coworkers in the rabbit model. The agent was applied following removal of cortical bone from the femoral condyle and the effects evaluated 4 weeks later. There was a dose-dependent significant effect on suppression of the formation of adhesions, with 0.1 mg/mL required to reduce the adhesion score, the hydroxyproline content, and the fibroblast number compared with controls.
Wang and associates studied the effects of weekly intraarticular injections of 0.3 mL hyaluronic acid in immobilized rabbit knees performed for 8 weeks. Compared with the control group, the experimental group had significantly greater range of motion ( P = .002), a lower mean adhesion score ( P = .01), and lower mean collagen content ( P = .001).
In a small animal model, Bedair and associates showed that an angiotensin II systemic receptor blocker decreased fibrosis and enhanced muscle regeneration in acutely injured skeletal muscle and hypothesized that the blocker modulated TGF-β 1 . Hagiwara and colleagues reported that the joint capsule has the potential to produce TGF-β 1 and connective tissue growth factor, both of which play a role in causing fibrosis.
Two small case series used a single dose of low-level irradiation (700 to 800 rads) before repeat revision TKA in patients with long-standing arthrofibrosis. The idea was derived from the established use of low-dose irradiation for the prevention of heterotopic ossification before total hip arthroplasty in high-risk patients, and the potential effects of irradiation in preventing the differentiation of mesenchymal stem cells into osteoblastic precursors. Farid and associates employed this treatment method in 14 patients with severe idiopathic arthrofibrosis in an effort to suppress fibroblastic and fibroosseous proliferation postoperatively. Major improvements in knee motion and function were experienced in all but one patient, and there were no complications related to the irradiation therapy. Baier and colleagues described three patients who had similar treatment and marked improvements in knee flexion. However, these authors cautioned that the influence of radioactivity on soft tissues has not been investigated, and bony nonunion may occur. Both series were retrospective, and no control group or randomization procedures were performed.
Bosch and colleagues advanced the hypothesis that primary arthrofibrosis is the result of an immune response. Tissue samples obtained from 18 patients between 4 and 48 months from the triggering event to surgical debridement demonstrated synovial hyperplasia with fibrotic enlargement of the subintima and infiltration of inflammatory cells. These authors believed their findings supported an immune response as the cause of capsulitis leading to formation of diffuse scar tissue. The clinical implication was to avoid further surgery or aggressive manipulation while patients have acute capsulitis and to wait 3 to 6 months for the inflammatory response to resolve before debridement is considered. As is discussed, too often, there is a delay in treatment that results in a permanent limitation of knee motion. Unfortunately, even though a delay in treatment may allow tissues to “quiet down,” the resulting fibrotic tissue that forms becomes well organized and dense and treatment is thus more difficult. It is anticipated that in the future, chemical agents will become available to treat fibrotic response to injury by controlling elevated levels of myofibroblast upregulators and fibrogenic growth factors.
After ACL injury, surgeon-therapist team manages pain, effusion, quadriceps weakness, knee motion limitations, gait abnormalities, other knee ligament injuries before ACL reconstruction.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here