Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Anatomic neurovascular, muscular, and anatomy of each region is imperative.
Soft tissue fillers can refer to a multitude of substances that are injected into the skin or subdermal area for cosmetic or therapeutic use.
Cosmetically, soft tissue fillers are best used to volumize areas of hollowness, camouflage areas of gravitational changes of aging, or improve anatomic harmony by changing the relationships and proportions of various anatomic features.
There are many different types of soft tissue fillers; the most ubiquitous being those made from hyaluronic acid, which is temporary, reversible, and inert.
In assessing a patient for soft tissue filler application, it is crucial to perform a targeted history and physical examination and evaluate the patient as to their expectations and understanding of the risks involved.
As soft tissue filler is an invasive medical procedure, it is crucial that patients understand the risks, benefits, and alternative treatments for their concerns and sign a consent form that formally indicates that they have this understanding.
In choosing a soft tissue filler, it is important to consider the target area and the goals to ensure that the filler is appropriate for that application.
Soft tissue fillers can be injected via a traditional needle or through a cannula approach. Although arguably requiring more skill and time, a cannula approach is safer and leads to more natural results with less downtime and potential bruising and swelling.
All practitioners performing soft tissue filler should be fluent in the potential risks and side effects they may encounter.
All practitioners should have an emergency plan, including available products and referral pathways, for devastating vascular consequences such as tissue necrosis, blindness, or stroke.
“Soft tissue fillers” is an umbrella term for a wide array of substances intended for dermal or subdermal injection, with the goal of antiaging, cosmetic enhancement, functional improvement, or correction of medical, surgical, or trauma-related defects. The therapeutic effect of soft tissue fillers is through direct volume replenishment of the treated areas and/or in the biostimulation of volumizing/skin-strengthening byproducts such as collagen and elastin.
Recently, studies have revealed an increase in patients seeking minimally invasive treatments to correct aging changes and as a “rejuvenation” procedure to delay the appearance of facial aging or enhance their existing facial features.
Although commonly regarded by the lay public as a treatment for “wrinkles,” the major value of soft tissue fillers is not necessarily in filling the superficial dermal and cuticular divots related to the integrity of the skin—the cause of true wrinkling—but instead in restoring volume to areas of age-, disease-, or trauma-related volume deficits. In addition—especially in younger patients—fillers can provide additional volumetric augmentation to change the proportionality of cosmetic features to balance and harmonize overall facial/body structure.
The concept of soft tissue filler dates back to the 19th century when autologous fat grafting was first attempted. Autologous fat grafting—or the transfer of fat from one part of the body to another as a living transplant—persists to this day as a well-regarded permanent volumization technique, especially as an adjunct to other plastic surgery procedures such as rhytidectomy (facelift) or mastopexy (breast lift) procedures. A detailed discussion of the techniques and use cases of fat grafting is beyond the scope of this chapter and textbook.
Paraffin wax—a white or colorless solid wax made from saturated hydrocarbons—was the first reported nonautologous dermal filler. Its first documented use was in the mid-1890s, yet some form of paraffin wax injection was continued into the mid-20th century. Paraffin wax injection fell out of favor because of the high migration rates, vascular occlusion, and granuloma formation.
Purified bovine dermal collagen was the first U.S. Food and Drug Administration (FDA)-approved filler substance, developed in the 1970s as a xenogenic soft tissue filler and first approved in 1981 under the brand name Zyderm for correction of contour irregularities in the dermis. Although highly effective, given its xenogenic origin, it required two rounds of preapplication sensitivity testing, which made its application more complex. In addition, its irreversibility led to difficult complications.
A turning point for dermal fillers was in the development and approval of a hyaluronic acid (HA) filler in 2003. Relatively nonreactive and reversible, it ushered in a new era of safe and effective nonsurgical filling. This also served to popularize and normalize soft tissue filler as a reasonable treatment for aging and volume disproportionalities (see Figure 4.1 ).
In general, HA is a glycosaminoglycan disaccharide found in all human tissue but predominantly in the vitreous, skin, and synovial fluid. The skin comprises approximately 50% of the total body hyaluronic acid.
HA for fillers is extracted from bacterial fermentation and lacks animal-derived proteins. Later, the HA undergoes stabilization to prevent enzymatically or oxidative degradation once injected into the body. In addition, cross-linking with 1,4-butanediol diglycidyl ether creates a polymer. The degree of cross-link will modify the rheology properties and longevity of the filler Table 4.1 .
| Product | HA Concentration mg/mL | % Cross-Linked |
|---|---|---|
| Restylane | 20 | 1.20% |
| Restylane Lyft | 20 | 1.20% |
| Restylane Silk | 20 | 1.20% |
| Restylane Kysse | 20 | 7% |
| Restylane Contour | 20 | 7% |
| Restylane Defyne | 20 | 8% |
| RHA 2 | 23 | 3% |
| RHA 3 | 23 | 3.50% |
| RHA 4 | 23 | 4% |
| Juvederm Ultra | 24 | 6% |
| Juvederm Ultra Plus | 24 | 8% |
| Juvederm Voluma | 20 | Highly (proprietary) |
| Juvederm Volbella | 15 | Highly (proprietary) |
| Juvederm Vollure | 17.5 | Highly (proprietary) |
| Revanesse Versa | 25 | 7% |
| Belotero | 22.5 | Cannot be measured |
Rheology is the science that studies the consistency, compression, and elasticity properties of liquids and gels. As of the writing of this chapter, HA is by far-and-away the most used type of filler globally.
Aside from various modifications of HA—both in manufacturing processes and additives (like lidocaine)—three additional synthetic filler substances entered the market after the first HA: poly-L-lactic acid (PLLA) (Sculptra), calcium hydroxyl-apatite (CaHA) (Radiesse), and polymethyl methacrylate (Bellafill). Unlike HA, all three of these additional filler substances have biostimulator properties and thus prompt the body to self-volumize by creating more collagen. Biostimulating fillers are considered “semi-permanent”: although collagen is not predictably broken down by the body, over time, it is subject to the same aging degradation that all collagen sustains ( Table 4.2 ).
| Product | Brand Name |
|---|---|
| Polymethyl-methacrylate microspheres | Artefill, Belafill |
| Calcium hydroxylapatite | Radiesse |
| Polyalylimide | Aquamid |
In addition to the synthetic fillers discussed earlier, a newer category of filler comes from donated fat that is purified to a cell-less allograft adipose matrix (Renuva). A combination of collagen, proteins, and growth factors, it is thought to provide a targeted scaffolding for the patient’s own body to build fat. It is commonly used to create permanent volumetric changes and in cases where fat grafting is required but the patient does not have adequate donor fat ( Table 4.3 ).
| Product | Brand Name |
|---|---|
| Human Tissue Collagen Matrix | Dermalogen, Dermaplant |
| Acellular Human Dermal Matrix | Alloderm |
As technology will surely continue to develop, research and development will continue to expand on our understanding of soft tissue fillers.
Fillers can be categorized in countless ways according to clinical applications, molecular structure, or scientific properties. We find it most helpful to organize soft tissue fillers into four general categories, each with their own benefits and potential risks and drawbacks ( Table 4.4 ):
biodegradable synthetic (e.g., HA, CHA, and PLLA)
nonbiodegradable synthetic (e.g., polymethylmethacrylate microspheres, polyacrylamide hydrogel polymers, and silicone)
allogenic biologic (e.g., collagen, allogenic purified fat)
autologous biologic (e.g., fat transfer)
| Category | Substances | Common Brand Names | Pros | Cons |
|---|---|---|---|---|
| Nonbiodegradable | Calcium hydroxylapatite | Radiesse |
|
|
| Nonbiodegradable | Polymethyl-methacrylate microspheres | Artefill, Belafill | Contains collagen |
|
| Biodegradable | PLLA | Sculptra |
|
Nonreversible |
| Biodegradable | Hyaluronic acid | Aptique, Esthélis, Elevess, Hylaform, Juvederm, Perlane, Prevelle, Puragen, Restylane | Reversible |
|
| Nonbiodegradable | Polyalkylimide | Aquamid |
|
Inflammatory reaction, nodule formation, granuloma formation |
There are several other materials that have been used outside of the United States which currently do not have FDA approval for distribution in North America, and these are briefly discussed below.
Aquamid (Contura, Soeborg, Denmark) is a polyacrylamide gel which is biocompatible, stable, and nonresorbable. It consists of 2.5% cross-linked polyacrylamide and nonpyrogenic water. It has been commerically used in Europe for decades.
Ellansé (Sinclair Pharmaceuticals, London, UK) is a biodegradable, collagen-type I biostimulant consisting of polycaprolactone microspheres in carboxymethyl cellulose gel. Ellansé was introduced in Europe in 2009. Its biodegradation occurs by hydrolysis of ester linkages, with CO 2 and H 2 O as end products that are ultimately eliminated from the body. Ellansé has four presentations with different in vivo longevity of 1 to 4 years, respectively (Ellansé-S, Ellansé-M, Ellansé-L, Ellansé-E).
Other important subjective and objective considerations for soft tissue fillers are:
Duration—how long does the product create change in the tissues? While each product has a certain duration profile as specified by the manufacturer and based upon clinical trials, this has also been found to vary based on placement and the patient’s own, non-quantifiable ability to break down filler. Officially quoted filler duration is somewhat biased and based upon clinical study endpoints in the filler approval process, and many practitioners agree that even HA fillers can last many years past their stated duration.
Affinity for water (hydrophilic properties)—all fillers, especially HA fillers, have a particular affinity for water, which is naturally occurring in human tissue. Depending on a particular filler’s hydrophilic nature, a certain amount of water will incorporate itself within the filler. This affinity for water is important to consider depending on the intended effect and injection area. For example, particularly, hydrophilic fillers should never be injected in the area underneath the eyes, an area that is associated with a significant amount of efflux and influx of water. This can create fluctuating infraorbital puffiness that is not aesthetically desireable.
Amount of Cross-linking—quantifies the cross-linking molecules per every 100 units of HA. The extent of cross-linking results in fillers that feel firmer and leads to enhanced longevity of fillers. Nevertheless, as cross-linking is a non-organic modification of HA, excessive cross-linking may lead to biocompatibility issues and higher risk of complications such as swelling. The physiologic reason is that alterations increase the risk of foreign body reaction. This immune response may lead to delayed inflammatory reactions such as swelling, granulomas, and nodules. ,
G-Prime—this refers to the “rheology” of the filler, or its viscosity, flexibility, and elasticity. In other words, it is a value that indicates the runniness and pliability of a filler. A higher G-prime filler tends to be more appropriate in areas where structure and “lifting” are required, whereas a lower G-prime filler is more appropriate in areas where there is more movement or the filler is injected more superficially.
Reversibility—this refers to the ability to deactivate or dissolve a filler. This is an important factor to consider, as filler that is reversible can be considered safer, given the practitioner’s ability to dissolve it should any issues occur. Reversability also tends to be reassuring, as patients are comforted to know that they can have the filler easily removed. The only reversible fillers that are currently available are those that are constructed of HA.
Biostimulatory properties—fillers that result in biostimulation create lasting, more permanent change by encouraging the body to create more collagen.
Approved areas of use—depending on the particular location, all available soft tissue fillers have approved techniques and areas of use. Although fillers are frequently used “off-label,” it may be important to consider where and how they are approved for injection. Injecting “off-label”—while common—poses some additional liability concerns for the practitioner.
Risk profile—soft tissue fillers can vary in terms of the potential risks.
Over the past 20 years, soft tissue fillers have become ubiquitous. Especially with the advent of social media and the newfound focus on photography and online presence, fillers are viewed as a panacea for antiaging.
Although soft tissue fillers can be profound in their ability to reduce signs of aging and positively change anatomic proportionality, they have significant potential side effects and, if not used correctly and in the appropriate patient, can have a range of consequences from relatively harmless to serious and even life-threatening.
As such, it is important to understand the indications for treatment in patient selection, how to guide the patient, and to plan for injection.
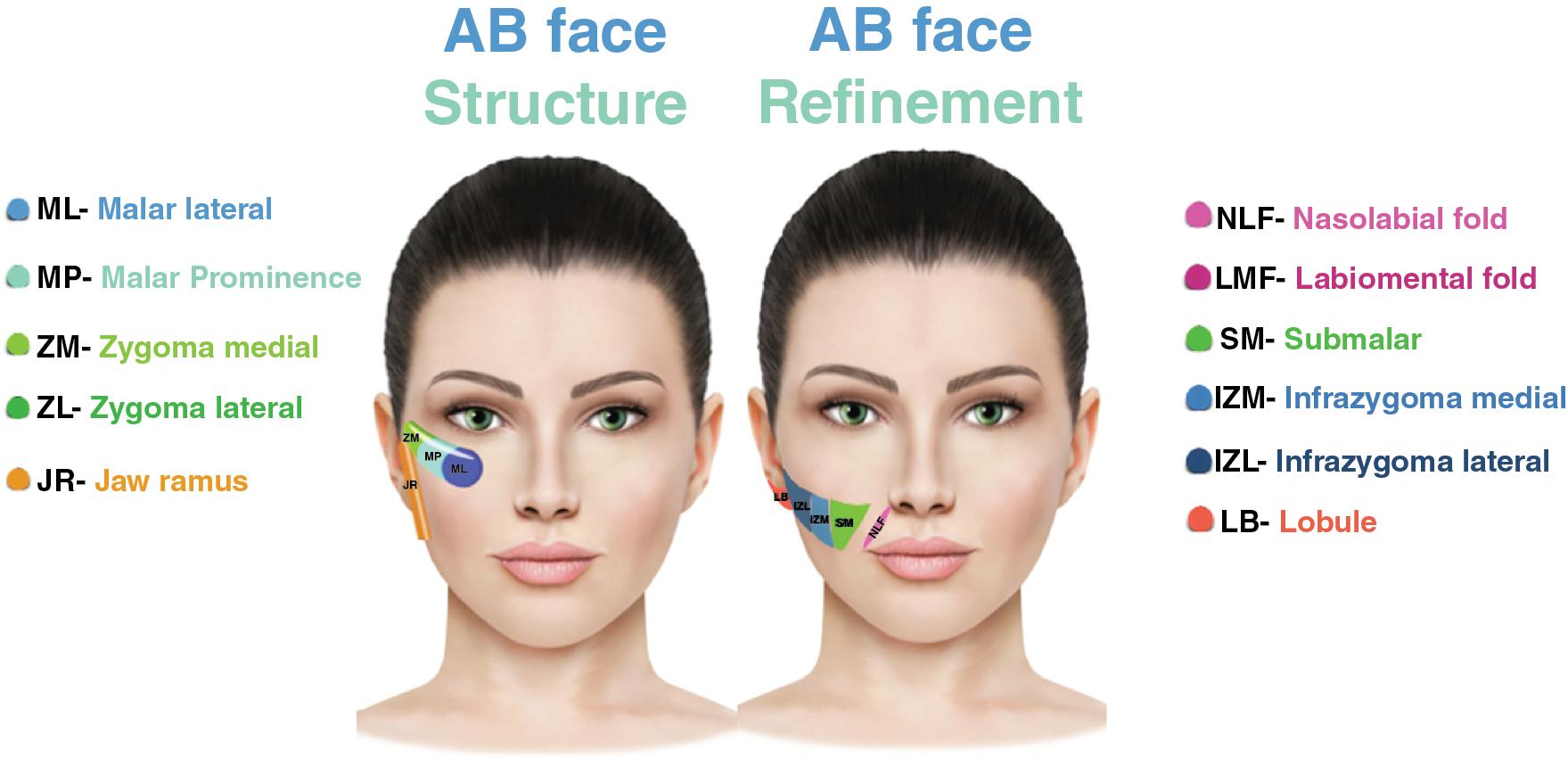
Tissue augmentation occurs in a three-dimensional pattern. As such, a complete understanding of the anatomy, tissue resilience, elasticity, and resistance is important to predict the filler’s behavior once injected.
The best-known and most popular application for dermal fillers is cosmetic augmentation. Cosmetic augmentation falls into three main categories; however, these categories are not mutually exclusive.
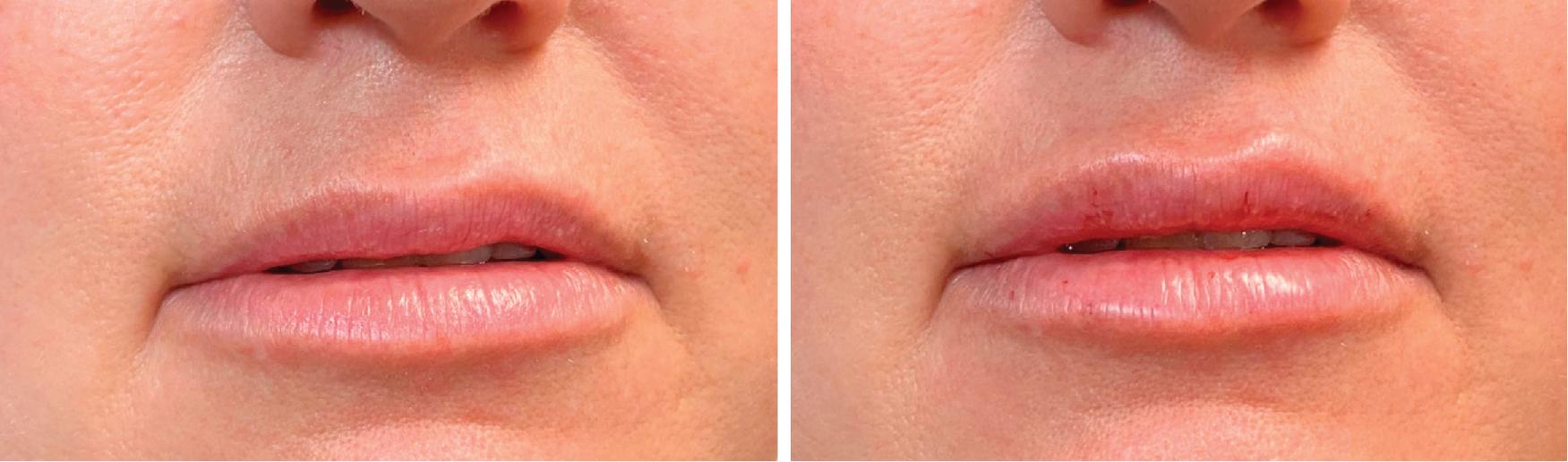
More traditionally, dermal fillers restore volume to areas where age-related volume loss has occurred. An example of this is a patient who has age-related volume loss in their lateral cheeks and would like the volume restored to where it once was (see Figure 4.2 ).
A different but overlapping category is the use of dermal filler to camouflage the stigmata of aging related to gravitational changes. The classic example of this is the patient who has jowling along their anterior jawline, in which filler is placed on either side of the jowl to reduce the transitional contour irregularities and shadowing caused by the jowl.
The third broad category of cosmetic augmentation is the use of filler to augment areas of the face or body that have not necessarily lost volume or are suffering from age-related changes, but instead to alter the proportions amongst various related anatomic features. As a younger and younger audience seeks out fillers, this use case comprises a larger percentage of filler patients. An example is nonsurgical rhinoplasty, which uses filler to reshape the nose to a more aesthetically pleasing shape, one that has a better relationship to the surrounding facial features.
Common areas of soft tissue filler augmentation are:
FACE
Upper and lower mucosal lip
Chin
Lateral cheeks
Medial cheek
Prejowl sulcus (anterior jawline)
Posterior jawline
Marionette folds
Nasolabial folds
Brow
Superorbital hollows (A-frame deformity)
Temples
Earlobe rejuvenation
Nose
Neck and chest
Superficial skin wrinkling
Periorbital
BODY
Buttocks
Breasts
Hands
Many patients seek dermal fillers to correct skin rhytids. Rhytids are folds/lines within the skin itself, related to generalized skin aging—called static wrinkles —or connected to movement in the area that creates a stereotypical pattern of wrinkling—called dynamic wrinkles . As a person ages, the dynamic wrinkling of the face corresponds to what eventually develops as the “relaxed skin tension lines” (RSTLs), which are stereotypical patterns of wrinkling that corresponds to natural facial movement patterns.
Dermal fillers can be used to correct rhytids—however, rhytid correction with filler is fraught with complexity and risk and should be cautiously approached. In particular, dynamic wrinkling can create challenges, as the area inherently moves, and filler—being a static gel—can look unnatural. In addition, true rhytids exist in the skin organ itself. Volumizing these lines can pose a challenge, as it requires the placement of filler into an inherently dense dermis. In the author’s experience, even the term “dermal fillers” is a misnomer, as fillers are best utilized to volumize when placed in sub-dermal locations.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here