Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Acute acquired concomitant esotropia (AACE) is not defined in the ICD10 or SNOMED CT and is described both as a relatively rare subtype of esotropia, distinct from paretic, incomitant strabismus and as the third most common form of strabismus. Rare or common depends somewhat on the definition but AACE certainly represents a diagnostic challenge. A typical presentation includes a sudden onset, concomitant, non-accommodative esotropia in a child ≥5 years of age with accompanying diplopia or visual confusion thought to have previously had straight eyes. Diplopia is often not expressed by younger children and one should ask about adaptations such as closing or covering one eye in children under 4 years who will adapt to their strabismus by rapidly suppressing the deviating eye. AACE is a diagnosis of exclusion and in these cases we need to ask: why has there been a loss of fusion? Most importantly, if we can’t identify a reason for this (e.g. a history of previous strabismus, occlusion therapy monocular visual loss or myopia) we need to explore whether the new onset strabismus is secondary to intracranial pathology. This is seen in a small number of cases (approximately 5%–10% neurological or secondary AACE) even in the absence of obvious neurological signs. Why patients with intracranial pathology develop AACE is uncertain as neither abducens nerve dysfunction nor hydrocephalus alone can account for all of the cases but tumors of the cerebellum, brainstem, pituitary region, and corpus callosum have all been reported to be associated with acute onset esotropia. Other causes include Arnold Chiari malformations and demyelinating disease.
There have been several attempts to subclassify patients presenting with AACE to guide further investigation and management. Burian’s seminal work classified these patients into one of three groups :
Those with temporary disruption of fusion by injury to an eye or occlusion (type I, Swan, occlusion-related AACE).
Those with a history of preceding illness, trauma, or physical or psychological stress with minimal refractive error <+3 spherical diopters and fusion (type II, Burian–Franceschetti, idiopathic AACE). A modern twist suggests that excessive mobile phone use should be added to this group.
Older children and adults with myopia and divergence insufficiency thought to result from excessive near work causing an inability to balance converging and diverging forces leading to increased medial rectus tone (type III, Bielschowsky AACE). This type is considered in more detail in the section on myopic esotropia.
More recent work suggests that this is an oversimplification, that there is likely overlap between types II and III and that it may be more helpful to describe two patterns, large or small angle. Decompensated monofixators with microdeviations are difficult to distinguish from type II and are also commonly described in the literature on ACCE, these are covered in more detail in the section on microtropia. Decompensated esophorias and more rarely cyclic esotropia and spasm of the near reflex (SNR) are also in the differential ( Box 79.1 ). The clinical picture of intermittent, usually small-angle esotropia, pseudomyopia with limitation of abduction and miosis means that SNR is normally easily differentiated from AACE but SNR has been described presenting with a constant large-angle esotropia, diplopia, and variable accommodative spasm and the absence of detectable miosis meaning that SNR needs to be kept in mind when seeing a case of acute, acquired, comitant esotropia. While SNR is normally considered a functional disorder it has been described in association with intracranial pathology including head trauma, multiple sclerosis, metabolic encephalopathies, Arnold–Chiari malformation, pituitary disease, pineal gland tumor, and sixth nerve palsy meaning that like AACE it is important to consider neurological evaluation and neuroimaging in the presence of atypical features.
Occlusion-related AACE
Idiopathic AACE
Myopia-associated AACE
Monofixators with microdeviations
Decompensated esophoria
Cyclic esotropia
Spasm of the near reflex
Neurological ACCE – tumors of the cerebellum, brainstem, pituitary region, and corpus callosum, Arnold–Chiari malformations, and demyelinating disease
Myasthenia gravis
Ophthalmoplegic migraine
Currently there is no evidence-based method to suggest which patients should undergo neuroimaging. If a cause for the esotropia is identified (e.g. significant uncorrected hypermetropia or myopia, a high accommodative convergence/accommodation [AC/A] ratio, anisometropic amblyopia or microtropia) without other suspicious features, imaging is not required.
Many pediatric ophthalmologists screen all patients presenting with AACE for the first time with a brain MRI while others suggest neuroimaging if neurological findings such as headache, papilledema, clumsiness, poor motor coordination, or nystagmus are present. A detailed history asking about common symptoms associated with posterior fossa tumor in children (nausea and vomiting, headaches and incoordination, lethargy, cranial nerve palsy, and diplopia ) is essential and assessment using head tilt test, evaluation for torsion, and alternating hypertropias in AACE may demonstrate extra signs pointing to intracranial pathology. Absence of motor fusion with prism or synoptophore correction of the angle may increase the likelihood of underlying neurological disease.
Intracranial disease should certainly be suspected if any of the following four risk factors are present: larger esodeviation at distance than near, recurrence of AACE, neurological signs and symptoms (papilledema, ataxia, and headache), and older age at onset (>6 years). Others suggest the detection of any nystagmus in AACE should prompt neurological and neuroradiological investigation.
Important factors relating to investigations are outlined in Fig. 79.1 and consideration of these and inquiry about neurological symptoms should help to identify patients with comitant esodeviation with underlying posterior fossa tumors, hydrocephalus, and Chiari malformation.
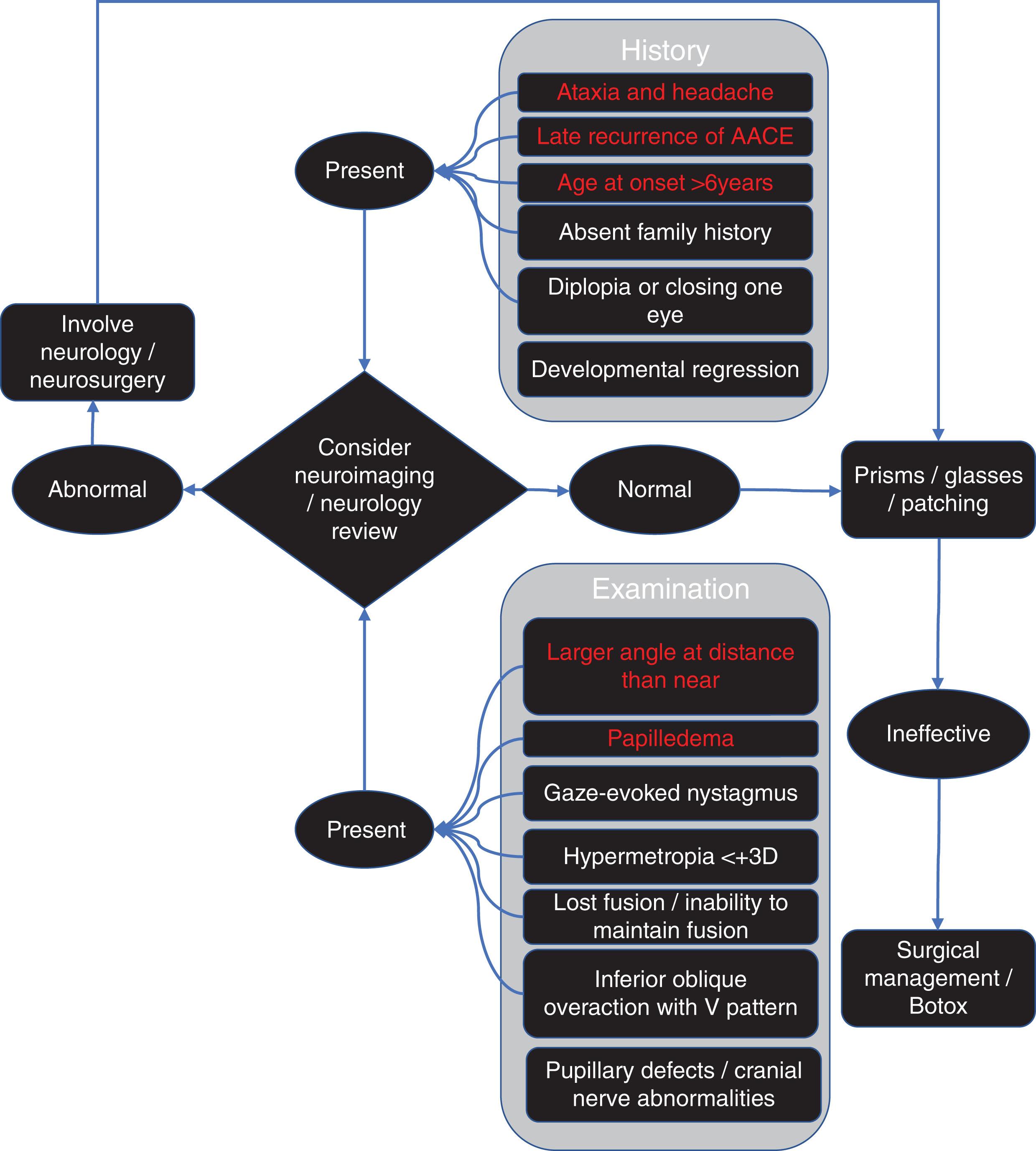
Whether to obtain neuroimaging in patients where the yield is low (subjecting them to added time, expense, and risk of intravenous contrast) or following them over time without scanning (with the risk of missing a potentially treatable disease) is the subject of ongoing debate. Choosing whether and how to image the brain will depend on what imaging you have available. CT has the advantage of ready availability and speed but increases the lifetime risk of developing leukemia and brain tumors (low absolute risk – for every 10,000 head CT scans performed for patients younger than 10 years it is estimated that one excess case of leukemia and one excess case of brain tumor occurs in the 10 years after the first scan ). MRI traditionally had the disadvantage of less availability, more expense, longer scan times and the need for sedation in many cases. Several developments including improved pediatric sedation, mock scanners, MR-compatible audiovisual systems, feed–sleep manipulation, play therapy, infant incubators/immobilizers, and more rapid MRI protocols may reduce barriers to MRI imaging for children where there is diagnostic uncertainty.
When assessing patients with AACE it is also important to consider alternative diagnoses such as myasthenia gravis which can cause any type of ocular misalignment. The history should cover variability of the deviation, effect of fatigue on the deviation, ptosis, limb girdle weakness, and speech or swallowing difficulties. Monocular visual loss can also result in concomitant esodeviations. It is important to check pupil reactions, visual acuity, and visual fields to exclude sensory esotropia. Finally, acute acquired transient esotropia can present in association with ophthalmoplegic migraine which will likely be clear from the history but may be mistaken for AACE on first presentation.
In summary, there is currently no single clinical sign that reliably indicates which patient with acute esotropia is harboring intracranial pathology and no reliable way to distinguish the uncommon but worrying cases of AACE associated with serious neurological disease. A very high index of suspicion should be maintained and neuroimaging considered when the etiology is unclear, when fusion is not regained after treatment, and when neurological signs or other atypical features are present. As one group put it, “beware, AACE are armed and dangerous” and careful initial and long-term observation is important to identify patients harboring intracranial pathology.
Where pre-existing high-grade binocularity was present prior to development of AACE the potential to re-establish motor and sensory fusion is excellent. Indeed as good binocular potential is a defining characteristic of this group their surgical results are better than patients with other indications for squint surgery such as unilateral vision loss. Early treatment of accommodative esotropia is associated with improved sensory and motor outcomes and it is suggested that an acquired esotropia in a young child should be treated as a daytime emergency. In AACE this may not be the case as age of onset appears to be more critical than duration of misalignment in terms of regaining bifoveal fusion. Children presenting after 4 years of age have the best outcomes although difficulties assessing sensory outcomes in younger children make this conclusion somewhat uncertain. It is also important to note that patients in this group achieve excellent binocular outcomes but that re-emergence of stereopsis can take time (even several years).
Glasses should be prescribed for the full cycloplegic error. If the eyes are not re-aligned, treatment options include prisms, botulinum toxin injections or strabismus surgery. The role of botulinum toxin injections is still debated in spite of evidence indicating that chemodenervation was non-inferior to incisional strabismus surgery at 6 months with benefits in terms of reduced duration of general anesthesia, time in the post-anesthesia care unit, and cost. To date, uptake of botulinum toxin for strabismus in children has been poor and further high-quality research to support its use and confirm the duration of effect is required. Several factors may reduce the attractiveness of botulinum toxin, including the lack of electromyographic (EMG) guidance for injection of anesthetized children, the expense of the drug itself, and doubts about its long-term efficacy. These problems may be partially addressed by Mendonça's forceps, which are commercially available. These have similar efficacy to EMG-guided injection making the expensive EMG machine and electrode needles unnecessary. The apparent long-term stability of reconstituted botulinum toxin may reduce costs and the American Society for Dermatologic Surgery consensus statement on reuse of botulinum toxin makes it clear that its use more than 24 hours after reconstitution and on more than 1 patient per vial is appropriate and consistent with the safe and effective practice of medicine. While results of studies show equivalent results at 6 months, less is known about long-term outcomes with botulinum toxin injections. Evidence exists for long-term action including micro-anatomical changes in the neuromuscular junction and muscle fibre remodeling in addition to central adaptive responses from several animal model studies. When surgery is planned the role of prism adaptation in deciding preoperative squint angle is debated.
Myopia is associated in childhood with both exotropia and esotropia. In one study from Japan, about 5% of pathologically myopic children were esotropic and 8% exotropic. Myopic esotropia can be sub-categorized into progressive comitant esotropia, usually associated with low- to mid-range esotropia, and the esotropia or hypo-esotropia associated with high myopia. Myopia is a feature of a number of pediatric conditions including retinopathy of prematurity, Down syndrome, Stickler syndrome, Marfan syndrome, and Noonan syndrome, some of which are associated with high myopia ( Fig. 79.2 ).

Progressive esotropia related to myopia tends to affect young adults but may present in childhood. The esotropia tends to be greater in the distance (divergence insufficiency type), may be associated with mild abduction weakness and often presents with intermittent distance diplopia.
In children, it is important to exclude neurological causes of distance esotropia even if there is no clear evidence of lateral rectus weakness. Herlihy et al., in a retrospective analysis, found that over 90% of children with distance esotropia had an acute onset deviation and had underlying neurological disorders including meningitis, hydrocephalus, and raised intracranial pressure due to other causes (such as transverse sinus thrombosis) or tumors (including those associated with NF1). Most of these children presented with the neurological condition and were subsequently referred with distance diplopia but three children presented to the ophthalmologists with distance diplopia and were then diagnosed with a neurological cause. All had apparently full ocular rotations. Progressive distance esotropia has also been reported associated with demyelinating disease even in the absence of other symptoms.
Esotropia in myopic children may be caused by over-correction of the myopia inducing accommodative convergence. Careful refraction is important. Xia et al. reported a series of young adults (mostly with mild to moderate myopia) who developed distance esotropia related to excessive near work. Refraining from near work for 3 months significantly reduced the deviation although all but one needed treatment with prisms or surgery.
Other reports of insidious onset distance esotropia with normal ductions in young, moderately myopic adults also attributed the onset to excessive near work. Recently, excessive smartphone use has been implicated in the development of myopic esotropia. Further evidence that excessive accommodative effort contributes to the development of myopic esotropia is provided by Campos, who treated patients with atropine over several months which progressively reduced the esotropia.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here