Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
It was estimated that there were 8580 anal squamous cell carcinomas diagnosed and 1160 deaths from this disease in the United States in 2018.
The age-adjusted incidence rate is 1.7 per 100,000 men and women per year with a female predominance.
Anal cancer is associated with human papillomavirus (HPV) infection, which mediates transformation of the anal squamous epithelium through a progression of precancerous lesions (anal intraepithelial neoplasia) to invasive cancer.
The American Joint Commission on Cancer (AJCC) tumor-node-metastasis (TNM) staging system for anal cancer is a clinical staging system. Notably, the T stage is based on tumor size rather than depth of invasion as it is in other gastrointestinal cancers. A revised nodal staging system was introduced in the AJCC eighth edition staging system.
Physical examination, with evaluation of lesion size, location, mobility, and inguinal nodal involvement, is critical in staging patients and for a baseline comparison to determine response.
Computed tomography, proctoscopy, endoanal ultrasound, and magnetic resonance imaging and/or positron emission tomography are helpful in evaluation of the extent of the primary lesion and the presence of regional nodal disease or distant metastatic disease.
Tumors of the anal canal are primarily squamous cell carcinomas.
Nonsquamous histologic types are rare and include anal adenocarcinomas, melanomas, and sarcomas.
Primary therapy for anal squamous cell carcinomas consists of definitive chemoradiation with concurrent mitomycin C, 5-fluorouracil–based chemotherapy, and radiotherapy.
Surgical resection is used only as salvage for patients who have persistent or recurrent disease after chemoradiation.
Induction chemotherapy, radiation dose intensification, and maintenance chemotherapy have not been shown to improve outcomes over standard chemoradiation for anal cancer.
The outcomes for anal squamous cell carcinoma are good, with a colostomy-free survival of 72% and overall survival of 78% at 5 years with pelvic radiotherapy and concurrent 5-fluorouracil and mitomycin C.
More advanced tumors are associated with a higher risk of local recurrence and distant failure.
Although rare, anal cancer provides a paradigm for organ preservation in the management of cancer. The treatment for anal cancer has evolved from the abdominoperineal resection (APR), which required a permanent colostomy, to sphincter-preserving nonsurgical therapy with concurrent radiation therapy and mitomycin C and 5-fluorouracil (5-FU)–based chemotherapy. However, the favorable disease-free survival associated with definitive chemoradiotherapy has been tempered by a significant rate of both acute and long-term toxicities. Improvements in pelvic radiotherapy techniques to minimize the exposure of normal tissues to potentially damaging radiation doses have demonstrated promising results in reducing acute toxicities. Investigations into alternative chemotherapeutic regimens have not yielded positive results to date. Studies evaluating targeted agents and less toxic regimens are ongoing. Further investigations are warranted to assess intensification of therapy in more advanced anal cancers and reduction of treatment in early-stage disease with the goal of improving both the tolerance to chemoradiotherapy and long-term, disease-free quality of life.
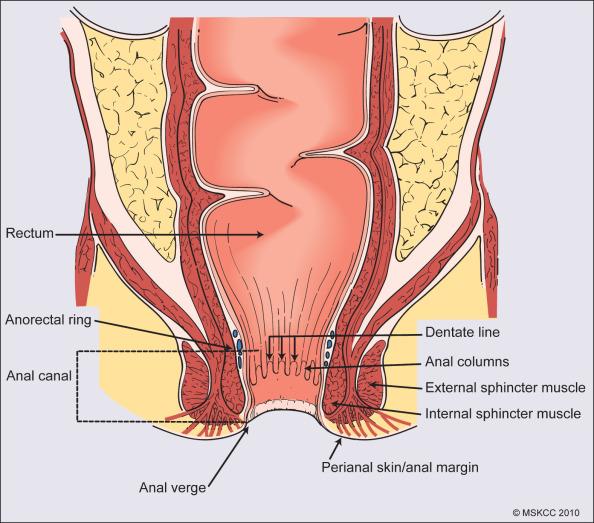
The anal canal extends cranially from the anorectal ring, where the rectum passes through the puborectalis sling to the anal verge, measuring approximately 3 to 4 cm in length ( Fig. 76.1 ). The anal margin or perianal skin is the hair-bearing skin within a 5-cm radius immediately beyond the anal verge, and neoplasms involving the anal margin have been traditionally managed as skin cancers. The anal verge is the junction of stratified squamous epithelium of the anal canal and the keratinized squamous epithelium of the perianal skin. The anorectal ring, which is palpable on digital rectal examination, is composed of a muscular bundle at the junction of the internal sphincter, the puborectalis muscle, and the external sphincter. The puborectalis muscle is one of the three muscles (the iliococcygeus, pubococcygeus, and puborectalis) which comprise the levator ani muscle or pelvic floor. The mucosa overlying the anorectal ring is a columnar epithelium which extends approximately 1 cm caudally to the dentate line, the histologic transitional zone between the columnar and stratified squamous epithelium. Tumors arising near the dentate line have historically been referred to as cloacogenic or transitional cell tumors and are composed of nonkeratinizing squamous cells; however, such tumors have similar prognoses and are treated in the same fashion as squamous cell cancers of the anal canal. Tumors that develop from mucosa (columnar, transitional, or squamous) are true anal canal cancers, whereas tumors that arise from skin distal to the anal verge are termed perianal skin cancers.
Squamous cell carcinoma of the anus is relatively rare and comprises only 6% of all large bowel cancers. The Surveillance, Epidemiology, and End Results (SEER) Cancer Statistics Review of the National Cancer Institute (NCI) estimated that 8580 new cases would be diagnosed in the United States and 1160 deaths would occur from the disease in the United States in 2018. From 2005 to 2009, the median age at diagnosis was 61 years of age. The age-adjusted incidence rate was 18 per 100,000 men and women per year, with a slight female predominance (2.1 per 100,000 women and 1.5 per 100,000 men). There has been a steady increase in the incidence of anal cancer in recent years in both men and women, with an annual percent change of 2.7% per year among males and 2.8% per year among females in the SEER regions from 1992 to 2004. There were some differences in anal cancer incidence by race and ethnicity from 1998 to 2003, with white women demonstrating a significantly higher rate of anal cancer than black women. Conversely, in men, blacks had a higher rate of anal cancer than whites, and Hispanic men had the lowest incidence. Another analysis of the SEER registry between 1973 and 2009 demonstrated that there was a clear inflection point in the incidence of both anal carcinoma in situ and invasive anal cancer cases. Before 1997 the combined incidence of anal carcinoma in situ and anal cancer increased at an annual rate of 2.2%, and after 1997 the combined incidence increased at a rate of 7.2%, likely a result of the increasing rate of human papillomavirus (HPV) infection and an increase in screening. Mortality rates from anal cancer also rose, on average, 3.2% each year from 2004 to 2013. Fortunately, at diagnosis, 48% of patients have localized (stages I/II) disease and only 13% have metastatic disease. The 5-year overall survival (OS) rate for all patients diagnosed between 2006 and 2012 was 66%, with 81% of patients with localized disease surviving 5 years.
Several factors have been implicated in the pathogenesis of anal cancer, including HPV, immunosuppression, and cigarette smoking. The most important risk factor for squamous cell anal cancer is infection with HPV, especially types 16 and 18. These high-risk HPV types act as carcinogens in the development of anogenital cancers. A meta-analysis has suggested that HPV-16 is found more frequently (75%) and HPV-18 less frequently (10%) in anal carcinomas than in cervical carcinomas. Moreover, approximately 80% of anal cancers demonstrated more than one HPV genotype.
As in cervical neoplasia associated with HPV, the viral proteins E6 and E7 mediate oncogenic transformation of the anal squamous epithelia. The viral E6 protein binds to the E6-associated protein (E6 AP) and ubiquinates the protein p53, which in turn leads to the proteosomal degradation of this major cellular transcription factor, leading to loss of the cell cycle arrest and apoptotic mechanisms that allow for deletion of errors in DNA replication. The viral E7 protein binds to the product of the retinoblastoma (Rb) gene, and consequently accelerates the cell cycle, allowing cells to progress from the G 1 into the S phase of the cell cycle.
As anal lesions progress from condylomata to low-grade dysplasia and then high-grade anal intraepithelial neoplasia (AIN) and invasive cancer, there is an accumulation of mutant p53 expression, emphasizing its role in tumor development. A prior history of anal condylomata has been reported in as many as 50% of homosexual and 30% of heterosexual patients diagnosed with anal carcinoma. The E2 protein allows HPV to escape intracellular detection by facilitating attachment of the HPV DNA to the host chromatin, an effect that allows steady replication of the virus in tandem with epithelial cells. Therefore through concerted actions of the HPV proteins E2, E6, and E7, the anal epithelium accumulates genetic errors leading to proliferation and eventually carcinogenesis.
Immunosuppression, in particular impaired cellular-mediated immunity, is another important risk factor in the development of anal cancer. The impact of suppressed cell-mediated immunity may be related to a reduced host response that would clear the HPV virus and prevent the establishment of a prolonged viral presence. Support for this finding comes from the observation that anal cancer rates are increased in both human immunodeficiency virus (HIV)–positive patients and patients who undergo renal transplantation, in which cell-mediated immunity is suppressed. In one series there was an approximate 100-fold increased risk of developing anogenital cancer in renal transplant patients. In another series, patients receiving chronic steroid therapy for amelioration of autoimmune disorders were also found to be predisposed to HPV-associated anal lesions.
Data from the prospective US Military Natural History Study (1985–2008) suggest that in HIV-infected men the incidence rate of anal cancer has been rising dramatically, even with the introduction of highly active antiretroviral therapy (HAART), reaching 128 per 100,000 men in 2006 to 2008. In addition, the risk of developing anal cancer increased with the duration of HIV infection, with a 12-fold higher rate of anal cancer in men infected with HIV for at least 15 years versus those infected for less than 5 years. Fortunately, the use of HAART appears to delay progression to anal cancer in HIV-positive men who have sex with men (MSM).
Cigarette smoking is also associated with the development of anal cancer, much as it is with cervical cancer. The risk of anal cancer appears to be related to pack-year history of smoking, with more extensive histories associated with a higher risk. The mechanism of smoking-associated tumors is unclear, but smoking may act as a cocarcinogen in the context of HPV infection.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here