Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Cytologic examination of a serous effusion is of paramount importance because the finding of cancer cells in such a specimen denotes that the patient has cancer that is not only advanced but also almost always incurable. Apart from the finding of cancer cells, cytologic examination of pleural, peritoneal, and pericardial effusions may also reveal information about inflammatory conditions of the serous membranes, parasitic infestations, and infection with bacteria, fungi, or viruses. It can also supply evidence of the presence of a fistulous connection with a serous cavity.
Although a serous effusion may be removed at the time of surgical exploration, it is usually sampled by the relatively simple procedure of inserting a wide-bore needle (under local anesthesia) through the body wall into the fluid-containing cavity. Peritoneal fluid is removed by abdominal paracentesis (colloquially referred to as paracentesis), pleural fluid by thoracentesis, and pericardial fluid by pericardiocentesis.
The fluid is collected into a clean, dry container, which need not be sterile, and sent to the laboratory as soon as possible. If the fluid cannot be sent immediately, it should be stored in a refrigerator at 4°C and not allowed to freeze. Anticoagulant or fixative does not need to be added to the fluid.
Formalin, alcohol, or any other kind of cellular preservative must not be added to specimens of serous fluid because formalin not only prevents cells from adhering well to a slide but also interferes with the quality of staining by the Papanicolaou method. Adding alcohol causes some precipitation of protein in virtually all specimens, thereby interfering with adherence of the cells to the slide. Apart from these criticisms of the use of formalin or alcohol, neither is necessary because keeping the specimen at refrigerator temperature, even for several days, preserves cells well ( Fig. 19-1 ).
The appearance to the naked eye of a serous effusion sometimes reveals clues about the cause of the effusion and the nature of its cellular contents. Therefore, for every serous effusion received by the laboratory, note should be made of its volume, color, clarity, and any unusual physical features, such as malodor, opalescence, or high viscosity.
Many serous effusions are noticeably bloodstained. A commonly held belief is that heavily bloodstained effusions are likely to be caused by cancer and that such effusions are more likely to contain cancer cells, although not substantiated.
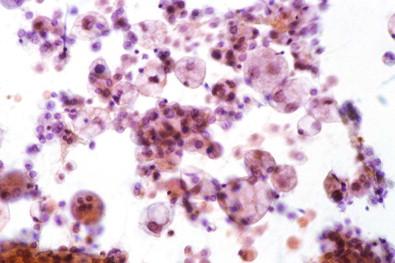
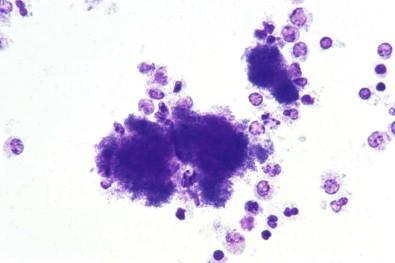
A serous effusion occasionally contains so many cancer cells that, if allowed to stand and sediment spontaneously, the cells form a thick, whitish-yellow layer at the bottom of the container. Spontaneously occurring sediment of similar appearance may develop in fluids containing numerous neutrophilic leukocytes.
Individual particles of cancer in a serous effusion may occasionally be large enough to be visible to the naked eye. Such particles, strikingly illustrated by de Vries, may be spheroids, ellipsoids, or similar shapes. Fig. 19-2 is a striking example of particles of metastatic squamous cell carcinoma in pleural fluid in which the particles were about the size and shape of sesame seeds. However, particles visible to the naked eye are usually spheroids, not more than 1 mm in diameter ( Fig. 19-3 ). Fluids containing particles of cancer visible to the naked eye produce excellent cell blocks.


Fluids containing numerous pigmented melanoma cells may be chocolate brown. Much lighter brown are effusions containing many hemosiderophages, a manifestation of old hemorrhage into the serous cavity. Effusions from patients who are jaundiced or that are a result of leakage of bile into the peritoneal cavity may have a rather dark brown-orange or greenish appearance that remains with the supernatant after the specimen has been centrifuged.
Serous effusions caused by malignant mesothelioma of epithelial type often contain a high concentration of hyaluronic acid, which may increase the viscosity of the fluid so much that it may have the consistence of thin honey. An effusion from the peritoneal cavity of a patient with pseudomyxoma peritonei (gelatinous ascites) is extremely difficult to aspirate because of its heavy mucoid consistence, and processing such a specimen is difficult because the cells it contains do not sediment during centrifugation.
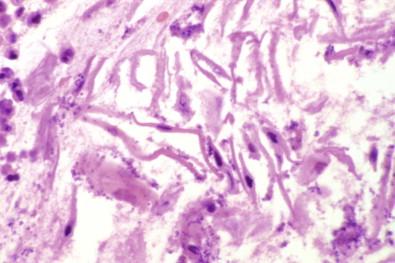
Effusions containing numerous cholesterol crystals are yellow and turbid and have a swirling, shimmering, gold paint appearance when agitated. The presence of cholesterol crystals can be confirmed by using a stained wet film (see “ Wet-film Technique ,” below) but the crystals are not seen in the permanent preparation stained by the Papanicolaou method, having been dissolved in the staining circuit. Chylous effusions have a milky white appearance with a creamy topmost layer due to their high concentration of emulsified lipid.
For serous effusion, three techniques can be used: a toluidine blue-stained wet film, a liquid-based preparation stained with the Papanicolaou stain, and cell blocks stained with hematoxylin and eosin. The usefulness of special stains, immunocytochemistry and molecular studies has been applied to cell block material. Although it is possible to apply the immunocytochemistry to Papanicolaou-stained smears without having to de-stain them, better results are obtained with cell blocks. For a comprehensive account of the application of electron microscopy to the cytology of serous effusions, readers should consult the monograph by Bedrossian although rarely used.
Remove any clots and extract all fluid from them by pressing them against the side of the container with a spatula or tongue depressor until left with a firm, rubbery mass. If the shrunken clot is large, put it into a Petri dish and cut it into small fragments with a pair of small scissors. Put the clot into 10% buffered formalin (as used for surgical specimens), allow it to fix for at least 30 min, and process the fragments as tissue.
Shake up the remaining fluid to disperse cells. Pour off an aliquot (up to 50 mL) into a centrifuge tube and centrifuge the sample for 5 min at 2000 rpm.
If the sediment is firm, pour off the supernatant by completely inverting the tube. If the sediment is not firmly compacted, as often happens with heavily bloodstained fluids, it tends to flow out of the centrifuge tube with the supernatant. In this situation, pipette off the supernatant, or with a pipette carefully “dredge” the buffy layer at the interface of the sediment and the supernatant. It may be necessary to recentrifuge the sediment removed this way.
A toluidine blue-stained wet film can be prepared as described below.
With a wire loop (as used in microbiology), remove a drop of the topmost layer of the sediment and transfer it onto the center of a glass slide.
Put an approximately equal-sized drop of toluidine blue stain (see below) on the slide next to the drop of sediment.
Mix the two drops together with the corner of a coverslip and then place the coverslip over the mixture. This wet film can be examined immediately. Because the permanent smears show an identical cytologic picture, we discard the wet films after they have been examined. Wet films may be kept in a Petri dish in a refrigerator for many hours or longer on a piece of gauze moistened with tap water ( Fig. 19-4 ). If the edge of the coverslip is sealed with nail polish to prevent drying out, a wet film may be kept in a refrigerator and remain interpretable for months or years.

Prepare the permanent smears, technique rarely used.
These techniques are well described in Chapter 33 , on Cytopreparatory Techniques. Cytocentrifugation techniques such as Cytospin or filtration techniques such as ThinPrep are currently used by most laboratories. The CytoRich Red procedure may be used for bloody effusions.
After preparing the wet film and cytospin or ThinPrep for the Papanicolaou stain, prepare a cell block of the remaining sediment.
Several techniques for cell block preparation are available. For more details, see Chapters 33 and 36 . Here, we illustrate the plasma thrombin technique.
Add 2 or 3 drops of plasma to the sediment. Outdated plasma from a blood bank is used for this purpose. Mix the sediment and the plasma together.
Add 3 or 4 drops of thrombin solution (see the following) to the mixture. Mix again.
Allow the mixture to clot; this usually takes only a few seconds.
Add tinted 10% buffered formalin (see below) to the clot. Pour the clot and the formalin into a Petri dish that contains any spontaneously formed clot.
With a pair of small scissors, cut the clot(s) into small pieces and allow them to fix for 30 min. They may, as a matter of convenience, remain in the fixative much longer.
Process the fragments (spontaneous and induced clot) as tissue.
The formalin used to fix the cell block material can be colored pink-orange by adding a small amount of powdered eosin. The embedded specimen then stands out tinted in the paraffin wax, and the histotechnologists will find it much easier to cut the sections. Adding eosin to the formalin does not alter the appearance of the stained sections.
This is prepared by adding 10 mL of distilled water to a vial containing 5000 units of powdered thrombin.
Prepare the stain as follows:
Toluidine blue (0.5 g)
95% ethanol (20.0 mL)
Distilled water (80.0 mL).
To prevent fungal growth, store the stain in a refrigerator.
Gross inspection of the specimen
Centrifugation of an aliquot
Prepare a stained wet film from the sediment
Prepare the liquid-based preparation (cytospin, ThinPrep)
Prepare cell block from solidified sediment and any spontaneous clot.
The preparation of a wet film is extremely simple and quick, and the film is ready to be examined immediately. Stained wet films are the cytologic equivalent of frozen sections in histopathology. The advantages of this type of preparation are as follows:
A wet film frequently reveals a distinctly diagnostic cellular picture, enabling a report to be issued within 10–15 min after receiving the specimen in the laboratory ( Fig. 19-5 ).
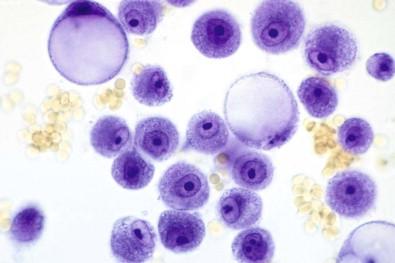
Wet films enable one to identify the “super-positive” serous effusions, those teeming with cancer cells. Not only are such specimens easily identified, but it is also possible to eliminate them from the routine staining circuit in order to avoid cross-contamination of other specimens.
A stained wet film may enable one immediately to identify unusual or interesting cytologic specimens, thereby providing an opportunity to prepare additional smears before the sediment is clotted for preparation of the cell block.
A large, solid fragment of neoplasm may absorb the toluidine blue stain very slowly or only at its periphery ( Fig. 19-6 ). Before they begin to absorb the stain, such fragments are easily overlooked
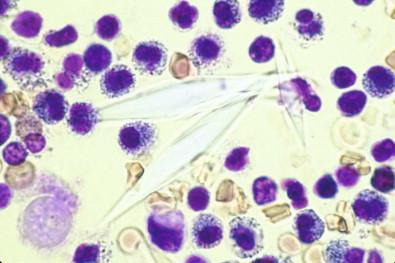
Apart from these advantages, stained wet films may reveal certain constituents in sediments obtained by centrifugation that are not seen in the permanent smears, such as cholesterol crystals, which are brilliantly highlighted by polarized light ( Fig. 19-7 ). Another crystalline curiosity revealed by the wet-film technique is Charcot–Leyden crystals ( Fig. 19-8 ), which may be found in effusions containing numerous eosinophilic leukocytes. Wet films reveal psammoma bodies, not because they take up the stain but because they do not react with toluidine blue and therefore stand out unstained against the surrounding stained cells ( Fig. 19-9 ). Bloodstained effusions may contain sheaves of brown hematoidin crystals, formed by the breakdown of hemoglobin.

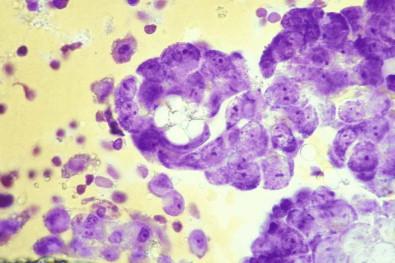
May allow rapid diagnosis and reporting
Allow “super-positive” specimens to be segregated
Reveal psammoma bodies and crystals.
The use of cell blocks of serous effusions is becoming standard practice in many laboratories. Specimens that are not anticoagulated frequently contain a clot that may be voluminous. Such a clot may have developed rapidly after the fluid was aspirated, enmeshing virtually all of the neoplastic cells in the specimen. Consequently, smears prepared from the remaining fluid after the clot has been removed may be devoid of neoplastic and other cells. In such a situation, sections of the spontaneously formed clot frequently reveal numerous, obvious neoplastic cells.
A discrepancy between positive smears and negative cell blocks can be explained by a slowly forming spontaneous clot, thereby allowing cancer cells to sink to the bottom of the container. In the cell block the spontaneously formed clot composed of dense magenta fibrin would contain very few, if any, cancer cells, whereas the induced clot prepared from the sediment obtained by centrifugation of the unclotted portion of the specimen would contain many ( Fig. 19-10 ). The reverse situation can also take place.
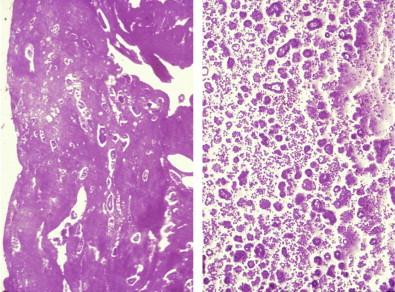
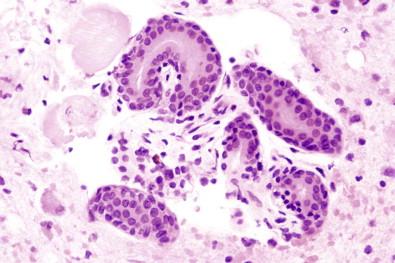
Cell block preparations may also reveal certain histologic aspects of a neoplasm such as papillary, acinar, or duct-like formations and also psammoma bodies, which may be difficult or impossible to detect in the liquid-based preparation (Cytospin, ThinPrep).
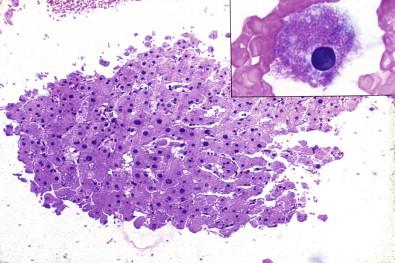
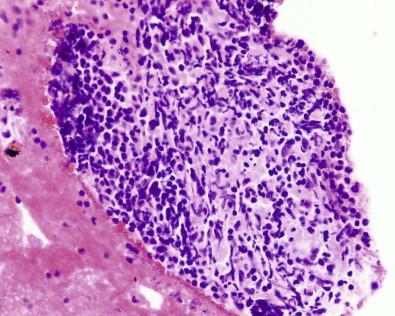
Cell blocks reveal other interesting histologic or cytologic entities, some mere curiosities but others of importance. Fig. 19-11 illustrates a fragment of liver in the cell block of peritoneal fluid obtained when the paracentesis needle traversed the liver on its way to the peritoneal cavity; retrospective analysis of the smears revealed only an occasional hepatocyte. Cell blocks can also reveal granulation tissue ( Fig. 19-12 ), a manifestation of pleural inflammation, cholesterol clefts in fibrin, skin, squamous epithelial cells ( Fig. 19-13 ), skeletal muscle ( Fig. 19-14 ), cartilage, colonies of microorganisms ( Fig. 19-15 ), accessory skin structures in subcutaneous adipose tissue ( Fig. 19-16 ), fragments of hyperplastic mesothelium with collagenous stroma ( Fig. 19-17 ), fibroblastic tissue, vegetable cells, and pulmonary parenchyma. Many of these entities either may not be present or are not recognized in the smear preparations.
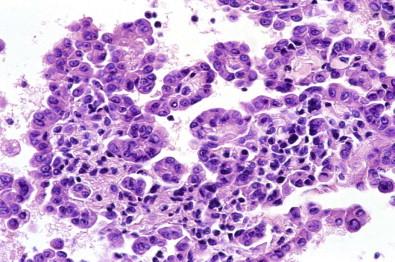
Spontaneous clot may enmesh virtually all the cells
Cell blocks will increase percentage of positive results
Cell blocks may reveal histologic aspects of neoplasm
Cell blocks may reveal entities not visible in smears.
The embryonic coelomic cavity gives rise to the serous cavities: pleural, peritoneal, and pericardial. The term serous refers to the small amount of serum-like fluid each cavity contains. The term is also applied to the cavity partly surrounding each testis formed by the tunica vaginalis testis, an embryonic extension of the peritoneal cavity. For a comprehensive account of the cytology of hydroceles, readers should consult the monograph by Spriggs and Boddington.
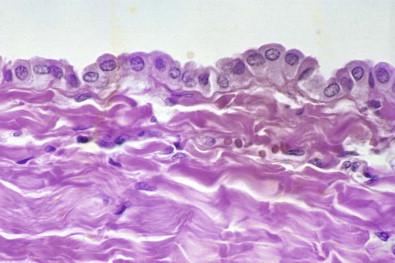
The serous cavities are commonly referred to as body cavities . Under normal conditions, the cavities are collapsed sacs invaginated by the heart, lungs, and intestines. Each cavity is completely closed (except the peritoneal cavity at the points where it receives the fimbriated ends of the fallopian tubes), and each contains a small amount of fluid. The outer layer of each serous cavity is the parietal layer; the layer directly in contact with the enveloped organ is the visceral layer. Apart from a thin film of fluid, these layers are in contact with each other; thus, under normal conditions, each cavity is only a potential cavity. When a cavity contains excess fluid, it becomes an actual cavity. Each cavity is lined by a monolayer of mesothelial cells, beneath which is a layer of connective tissue, supplied with blood vessels, lymphatics, and nerves ( Fig. 19-18 ). The close proximity of blood and lymphatic vessels to mesothelium may partly account for the ready spread of neoplastic cells into a serous cavity.
Accumulation of fluid in a serous cavity in excess of the normal small amount is referred to as an effusion. Effusions are designated according to their location: pleural, peritoneal, or pericardial. The condition of peritoneal effusion is frequently referred to as ascites and the fluid as ascitic fluid. Air may sometimes be introduced into a serous cavity by trauma (including surgical trauma), for radiologic diagnostic purposes, for therapy, or secondary to a pathologic process, producing the condition of pneumothorax, pneumoperitoneum, or pneumopericardium. When these conditions are accompanied by effusion, the terms are expanded to indicate its presence: pneumohydrothorax, pneumohydroperitoneum, and pneumohydropericardium.
Transudates are effusions characterized by a low protein content, usually <3.0 g/dL, and low specific gravity, usually <1.015. Transudates accumulate by the filtration of serum across physically intact capillary walls under conditions in which the outflow of fluid through a serous membrane exceeds the normal reabsorptive process. This may take place as a result of increased venous pressure, as in congestive heart failure or cirrhosis of the liver, or in hypoproteinemia in renal failure. Transudates generally have a lower cellular and fibrin content than exudates. The cellular content usually consists of mesothelial cells and macrophages, with an occasional lymphocyte or neutrophilic leukocyte.
The cellular content is higher and is likely to contain many inflammatory cells in exudates caused by inflammation or, in exudates caused by neoplasm on the serosal surface, many neoplastic cells.
Pleural inflammatory exudates are likely to be caused by pneumonia, pulmonary infarct, pulmonary abscess, pleuritis, or secondary bacterial infection of a transudate. Peritoneal inflammatory exudates are likely to be caused by peritonitis, either spontaneous bacterial or secondary to infarct or inflammation of the bowel, spontaneous or traumatic rupture of a viscus, or pelvic inflammatory disease in women. Pericardial effusions that are exudates are likely to be caused by viral or bacterial inflammation of the pericardium or uremic pericarditis. In the less prosperous nations of the world, tuberculous inflammation of a serous membrane is always foremost in the minds of clinicians, whereas in affluent societies it is far less frequent.
Serous effusions caused by neoplasm may be transudates, resulting from failure of resorption of serous fluid due to mechanical interference by neoplasm, or they may be exudates caused by a neoplasm that damages the capillaries of the serous membranes.
Every pleural, peritoneal, and pericardial effusion contains cells, often numerous, occasionally scanty. The non-neoplastic cells commonly found in serous effusions are those derived from blood (erythrocytes, leukocytes, and histiocytes) and from the serosal lining, the mesothelial cells. The proportion of the different types of these cells varies considerably, depending on certain circumstances such as the cause and the duration of the effusion and the presence or absence of inflammation.
Examples of normal cells rarely found in spontaneously occurring serous effusions are megakaryocytes, hepatocytes, and cells derived from the alimentary or respiratory tract via a fistula. Cells detached from the fimbriated end of the fallopian tubes are occasionally found in peritoneal fluid obtained by culdocentesis, laparoscopy, laparotomy, or dialysis catheter.
It is difficult to ascertain what is a normal range for cells in serous effusions, because normally the serous cavities contain only a small amount of fluid. It is only when there is an excess of fluid, an abnormal situation, when it is possible to aspirate it. It is known, however, that the cellular content of peritoneal fluid varies considerably in different animal species. Yamada aspirated pleural fluid from healthy soldiers and found that the cellular content ranged from 1700 to 6200 cells/mL, with most of the cells being classified as “large mononuclear cells,” presumably histiocytes, and the remainder as mesothelial cells and lymphocytes. Culdocentesis specimens from apparently normal persons may contain papillary clusters of mesothelial cells in which there are psammoma bodies.
Pericardial fluid may be easily obtained by aspirating the pericardial sac at the time of thoracotomy. Spriggs and Boddington examined the sediment of 14 samples of pericardial fluid obtained in this way. The cells were classified as mesothelial or histiocytes, and mitotic figures were found in some of the cells. They also noted that serous effusions collected at necropsy generally contained large numbers of mesothelial cells that had exfoliated after death. Ramsey and colleagues obtained pericardial fluid from patients undergoing thoracotomy, some for cardiac surgery. These fluids contained small histiocytes and mesothelial cells, some in papillary clusters.
Despite the uncertainty about what is a normal range of the various cell types in serous effusions, it is common practice to send these fluids to the hematology laboratory for total and differential cell counts. Such counts should be performed only on fluids that have been anticoagulated; otherwise, any clot that forms is likely to entrap cells and give spurious results.
Only a few characteristic (though not specifically diagnostic) cytologic pictures are demonstrated by total and differential cell counts, such as the cytologic profiles of acute purulent inflammation, eosinophilic effusion, and lymphocytic effusion. Under non-neoplastic conditions, the cytologic picture is usually that of a mixed population that, when analyzed quantitatively, does not give any specific diagnostic findings.
Mesothelial cells readily undergo hypertrophy and hyperplasia in response to a wide variety of stimuli, such as inflammation of the serous membrane, inflammation or necrosis of the underlying parenchyma, the presence of foreign substances such as blood or air in the serous cavity, and even the presence of a longstanding sterile effusion such as may occur in the peritoneal cavity in hepatic cirrhosis.
In the presence of an effusion, whatever its cause, mesothelial cells exfoliate, often in large numbers, either as isolated cells or as cohesive clusters of cells or both. It is entirely possible that mesothelial cells exfoliated into a serous effusion continue to proliferate because they are in a natural medium. In fact, of all the benign cells seen in cytopathologic practice, mesothelial cells are the most likely to show mitotic figures, evidence of their ability to proliferate in a serous effusion.
The prototypic mesothelial cell exfoliated into a serous effusion ( Fig. 19-19 ) is round, about 25 µm in diameter, and has a single central or eccentric nucleus. With the Papanicolaou stain, the cytoplasm is dense and gray-green or slightly eosinophilic and tends to become less dense at the periphery of the cell, imparting an indistinct foamy or even scalloped appearance to the cell membrane. The nucleus is round or oval and possesses a well-defined, smoothly contoured membrane. The chromatin is uniformly granular, and nucleoli are readily identified.
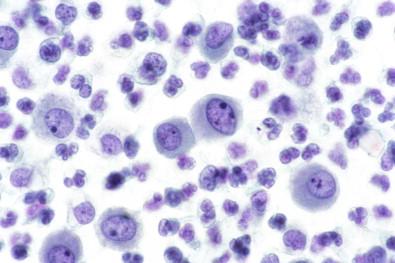
Mesothelial cells vary considerably in size, from small cells about 9 µm in diameter with relatively little cytoplasm to giant cells more than 60 µm in diameter. Large, hypertrophied and much smaller mesothelial cells are frequently seen joined to each other. Giant mesothelial cells are almost always multinucleated, often with ≥10 nuclei ( Fig. 19-20 ).
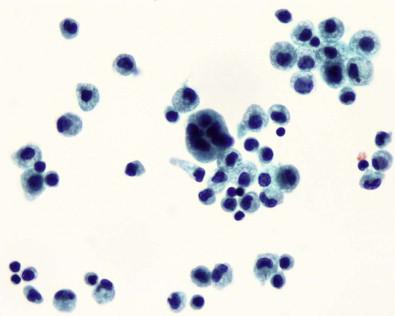
Cytoplasmic vacuolation, when present, may take several forms:
One or more tiny, unobtrusive vacuoles scattered throughout the cytoplasm but more likely to be near the nucleus
A large, solitary vacuole that seems to displace the nucleus to the periphery of the cell
An elongated, perinuclear vacuole that curves around a segment of the nuclear membrane like a tiny sausage.
It is not uncommon to find a mesothelial cell with a large, solitary vacuole and an eccentric nucleus joined to a typical non-vacuolated mesothelial cell, clearly denoting the mesothelial lineage and benign nature of the vacuolated cell ( Fig. 19-21 ).
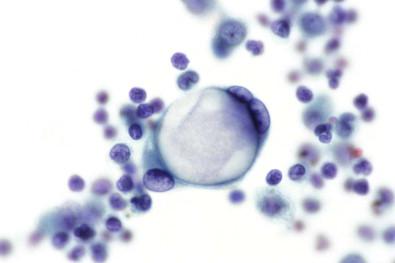
Mesothelial cells articulate with each other in a characteristic manner. One form of articulation consists of little chains of cells joined at flattened or curved apposing surfaces ( Fig. 19-22 ). Groups composed of two to four mesothelial cells are frequently seen to have this type of articulation. Between the apposing cellular surfaces, clefts or “windows” may develop, suggesting that the conjoined cells are about to become detached from each other. Less commonly seen, although with the same type of articulation, are larger sheets of mesothelial cells with a mosaic appearance composed of about ≥10 cells ( Fig. 19-23 ). Another articulation characteristic of mesothelial cells is illustrated in Fig. 19-24 , in which the cytoplasm of one cell appears to be clasping another cell. It should be mentioned that all of these types of articulation can be seen, although much less frequently, in adenocarcinoma cells in serous fluids.
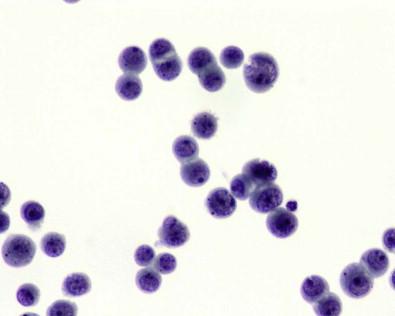
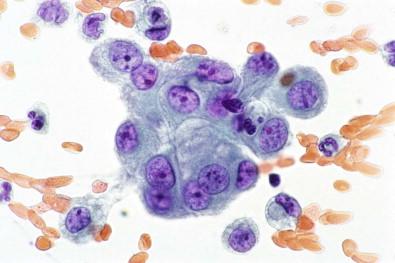
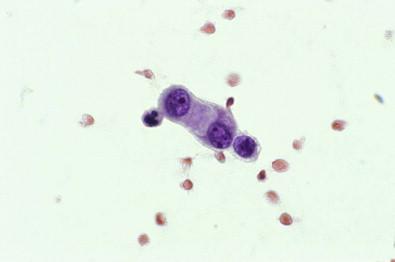
Tissue fragments composed of mesothelial cells characteristically have knobby or scalloped contours due to individual cells protruding from the periphery ( Fig. 19-25 ). In smears, it is the presence of these more visible protruding cells that allows the mass of cells to be identified as mesothelial. Occasionally, however, large, round, smoothly contoured clusters of mesothelial cells are found, a situation in which it is more difficult to determine the mesothelial nature of the cells. It should be possible, however, to detect at the edge of the cluster a fine rim of cytoplasm showing the peripheral cytoplasmic fading characteristic of mesothelial cells. Cell block preparations can be extremely useful in this situation, because they may more readily reveal the benign nature of these fragments. Should any doubt remain about whether the cells are mesothelial or adenocarcinomatous, immunocytochemistry applied to cell block material usually enables one to distinguish between mesothelial and adenocarcinoma cells (see “ Mesothelioma ,” below).
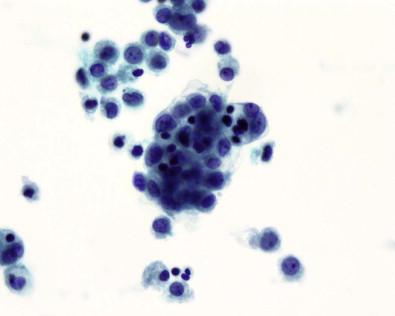
Giant multinucleated mesothelial ( Fig. 19-20 ) cells are not uncommon, having been reported in 26% of 396 serous fluids. They may be misinterpreted as giant multinucleated histiocytes, a rarity in serous effusions, found almost exclusively in specimens from patients with rheumatoid pleuritis or pericarditis. Furthermore, the cytoplasm of giant multinucleated mesothelial cells does not exhibit the fine, diffuse vacuolation typical of histiocytes. It may contain several well-defined vacuoles, with the residual non-vacuolated cytoplasm being typical of mesothelial cells in general. In contrast to the nuclei of histiocytes, the nuclei of giant multinucleated mesothelial cells are smoothly contoured with well-defined membranes, and they contain prominent nucleoli. Giant multinucleated mesothelial cells are usually accompanied by numerous smaller cells whose mesothelial lineage is easily recognized.
We commonly come across reports of “atypical” or “reactive” mesothelial cells, designations given to mesothelial cells that are hypertrophic ( Fig. 19-26 ) and often hyperplastic. Furthermore, mesothelial cells that are not well spread and well stained, especially if the serous effusion is bloody, may appear dark, thereby creating diagnostic uncertainty – hence, the use of the terms atypical and reactive.
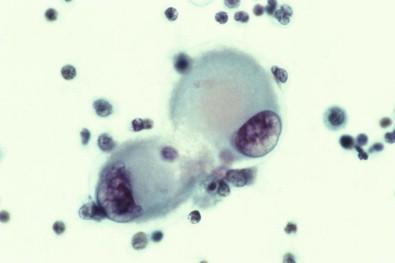
The morphologic features of mesothelial cells seen in Papanicolaou-stained smears are also visible in toluidine blue-stained wet films, with several important differences. First, mesothelial cells in toluidine blue-stained wet films are noticeably larger than those in permanent, wet-fixed preparations. (This size difference applies to all types of cells examined in wet films.) Another difference is that mesothelial cells in wet films frequently contain tiny, glistening, golden, cytoplasmic lipidic inclusions not seen in the permanent preparations because they are dissolved by solvents in the fixative or staining circuit. Finally, the types of articulation characteristic of mesothelial cells are usually more difficult to discern in wet films, although careful observation reveals that they are present.
Mesothelial cells in cell block preparations generally do not present any particular diagnostic difficulty. Because these preparations are stained with hematoxylin and eosin, the cytoplasmic staining reaction is one of various shades of pink. Except for this, all of the previously mentioned morphologic features of mesothelial cells seen in Papanicolaou-stained smears are discernible in cell block preparations, although not as readily as in smears.
Papillary fragments of mesothelium that become detached from the serosa, especially during a washing procedure, may consist of a connective tissue core covered by a layer of mesothelial cells. Such connective tissue cores may become calcified to form psammoma bodies, illustrating that malignancy must not be diagnosed on the presence of psammoma bodies alone; the diagnosis must be based on the accepted nuclear changes of malignancy in any accompanying cells.
Becker and coworkers reported an example of overdiagnosis due to the presence in pericardial fluid of exfoliated micropapillary excrescences covered by mesothelial cells. Benign papillary proliferations may develop on any serous surface and are usually attributed, especially in the female pelvis, to the effects of previous inflammation, including those induced by surgical operation. They do not cause effusion.
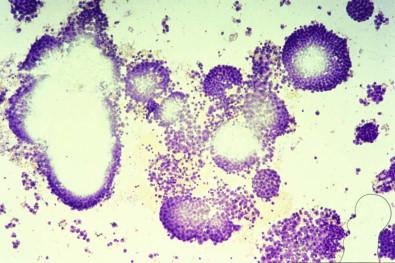
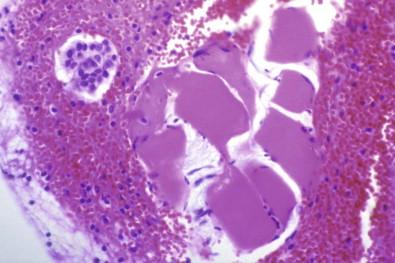
With the Papanicolaou stain, such fragments of collagenous connective tissue covered by mesothelium are frequently seen as smoothly contoured, homogeneous, pale green bodies covered by a layer of flattened cells ( Fig. 19-27 ) – hence the term collagen ball that we adopted to describe this entity. Collagen balls are virtually confined to washings of the peritoneal cavity or specimens obtained by culdocentesis. We have rarely seen a collagen ball in a spontaneous pleural, peritoneal, or pericardial effusion; in such specimens, collagen balls are almost always small and unobtrusive and may be overlooked. The 3-dimensional, spheroidal nature of peritoneal collagen balls is realized by focusing up and down on the ball and finding that the covering cells are visible at different planes. An inexperienced observer might regard these characteristic fragments as adenocarcinoma cells distended by mucin. Apart from the fact that their nuclei do not possess the features of malignancy, any doubt about the composition of these fragments can be settled by cell block preparations, which show the characteristic eosinophilic collagenous cores.
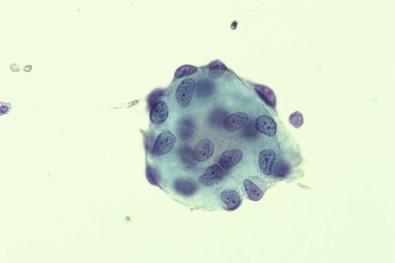
It was our belief that collagen balls were probably detached from microexcrescences that are normally present on the surface of the ovary. However, Sasaki and colleagues demonstrated that “serosal balls,” their name for the entity, may be found with at least equal frequency in peritoneal washings of men. Consequently, they suggested, probably correctly, that serosal balls in peritoneal washings originate by detachment of minute fragments of serosa and underlying stroma during a surgical operation; these detached fragments then round up to produce balls.
To anyone embarking on cytopathology, it soon becomes obvious that the mesothelial cell in all of its variations offers the greatest potential for being misinterpreted as metastatic adenocarcinoma. The important question is how can one eliminate or at least diminish the chance and frequency of such error?
The first step is to rely on only well-prepared specimens in which the cells are wet fixed (if the Papanicolaou stain is being used), not overstained, and easily visible. With such preparations, the mesothelial nature of individual cells should be obvious. Smears that are too thick or very bloody increase the potential for overdiagnosis. In such smears, the staining reaction of mesothelial cells is darker, with the consequence that their nuclei appear hyperchromatic and suggestive of malignancy.
When the cytologic picture remains ambiguous or when mesothelial cells appear to be hypertrophic or hyperplastic (“atypical” or “reactive”), immunocytochemistry can be helpful.
Round, great size variation, usually one nucleus
Giant forms with many nuclei
Cytoplasm gray-green to slightly eosinophilic
Mosaic clusters, monolayer strips, and collagen balls in washings.
The sediment obtained by centrifugation of most serous effusions, even those that appear not to be bloodstained, contains a layer of red blood cells visible to the naked eye.
Red blood cells are readily recognized in Papanicolaou-stained smears as neatly round orange-red discoids about 7 µm in diameter, although many may become distorted into elongated forms in the process of spreading the cellular sample. Fixation may cause red blood cells to undergo lysis, leaving residual empty cell membranes with a slightly cyanophilic staining reaction. Hemoglobin released from these cells may remain as part of the proteinaceous background of a smear, thereby imparting a reddish cast to the smeared material, both to the naked eye and under the microscope. In the toluidine blue-stained wet films, red blood cells are unstained, having a light straw yellow color.
Apart from these artifacts, normal red blood cells generally show little morphologic variation.
Intact red blood cells may be phagocytosed by macrophages. When red blood cells disintegrate, their hemoglobin is converted into hematoidin or hemosiderin, which, if present in a large enough amount, imparts a distinctly yellow (xanthochromic) appearance to the supernatant. In stained wet films, hematoidin pigment may occasionally be seen as sheaves of fine, elongated, yellow-brown extracellular crystals, whereas hemosiderin is seen as dark blue granules in the cytoplasm of macrophages. In Papanicolaou-stained smears, hematoidin retains the same appearance, but the staining reaction of hemosiderin is golden brown to olive green. Hemosiderin gives a positive staining reaction for iron (Perls test), whereas hematoidin does not.
Seen in virtually every specimen
Biconcave discoids, frequently lysed
Shape possibly distorted by processing
May degenerate to produce hematoidin and/or hemosiderin.
Almost every specimen of serous effusion contains neutrophil leukocytes. Their number varies from just the occasional cell to highly cellular purulent effusions in which all or almost all of the cells are neutrophils. Purulent effusions are of a light yellow turbidity and a creamy consistency; if infected, they may be malodorous. Neutrophils are readily recognized in toluidine blue-stained wet films, Papanicolaou-stained permanent smears, and cell block preparations.
Inflammation, infarction, and rupture of an organ are the principal causes of serous effusion containing numerous neutrophils. Hypertrophied mesothelial cells may also be present in the early stages of acute inflammation, but, as the neutrophils dominate the picture, the mesothelial cells die and disintegrate. Even in purulent effusions, a small percent of the cells may be histiocytes or lymphocytes or both.
In wet films, the cytoplasm remains unstained or weakly stained ( Fig. 19-28 ).
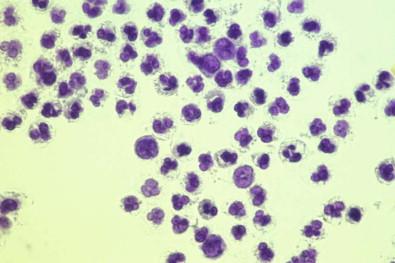
Neutrophils in non-infected effusions are generally well preserved. In infected purulent fluids, neutrophils degenerate; in some effusions, virtually every neutrophil is necrotic. Necrotic neutrophils become transformed into ill-defined, light gray to gray-blue particles without any visible nuclear material or cytoplasmic granularity ( Fig. 19-29 ). The nuclei of necrotic cells may also condense into round, solitary, deeply cyanophilic masses, or they may become fragmented.
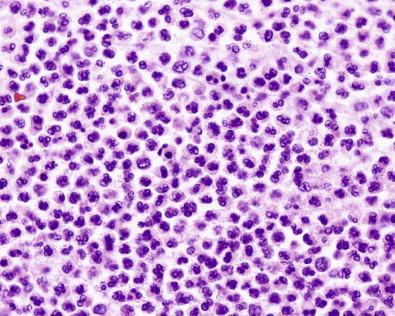
Readily identifiable in all types of preparation
Cytoplasm granules not prominent
Nuclei usually trilobed
Possibly necrotic in inflammatory lesions.
Several comprehensive reviews during the early days of cytodiagnosis attest to the fascination that eosinophilic effusions, especially pleural effusions, have held for clinicians. However, the arbitrary criterion for designating a pleural effusion as eosinophilic has varied from an effusion with as few as 5% to one with at least 50% of the cells being eosinophils; we have adopted the criterion of a concentration of 10% or more as used by Koss.
Eosinophilic pleural effusion has been reported in association with a wide variety of conditions: allergy, autoimmune disorders, pneumonia (including viral pneumonia), pulmonary infarct, fungal infection, parasitic infection, malignant neoplasms, pulmonary tuberculosis, artificial and spontaneous pneumothorax, and hemothorax, including that induced by thoracic trauma. A total of 81 of the 127 (64%) cases of eosinophilic pleural effusion analyzed by Spriggs and Boddington and 8 of 30 (27%) analyzed by Veress and coworkers were associated with some form of thoracic trauma.
Spriggs concluded that pleural eosinophilia is the normal reaction of the pleura to the introduction of air into the pleural cavity and suggested that the reaction is more likely a result of suspended particles of animal or vegetable origin than of the air itself.
When pneumothorax can be excluded, the commonest causes of eosinophilic pleural effusion seem to be pulmonary infarct, pneumonia, and neoplasm. Hodgkin's disease is rarely associated with eosinophilic pleural effusion. Only a minority of cases can be attributed to recognizable hypersensitivity states, including parasitic infestation. Another cause of pleural eosinophilic effusion is the benign type of effusion associated with the inhalation of asbestos; in a series of 60 cases, 26% showed eosinophilia of various degrees.
When all of the possible causes of eosinophilic pleural effusions have been eliminated, there is a substantial residue of cases in which the cause cannot be identified.
Despite the uncertainty surrounding the pathogenesis of idiopathic eosinophilic pleural effusions, they seem to have a favorable prognosis, even in patients with a previous history of cancer.
Because it has not been possible to identify a single linking pathologic mechanism behind the formation of eosinophilic pleural effusion, it has been suggested that these effusions exist in two forms: an effusion of relatively acute onset related to an allergic reaction or to thoracic trauma and the more chronic form with a longer clinical course.
Eosinophilic peritoneal effusions are rare. Examples of eosinophilic peritoneal effusion associated with malignant neoplasm, various allergic states, parasitic infection, eosinophilic gastroenteritis, and chronic peritoneal dialysis have been reported. It is possible that one or more of the agents used in peritoneal dialysate, such as antiseptic, talc, particles of tubing, and peritoneal catheters, provoke a hypersensitivity reaction characterized by an efflux of eosinophils. In addition to eosinophilia, chronic peritoneal dialysis may stimulate mesothelial hypertrophy and hyperplasia.
Eosinophilic pericardial effusion is even rarer than eosinophilic peritoneal effusion. A few examples in association with pulmonary eosinophilia, including one associated with the use of the drug cromolyn sodium, have been reported. Examples of Hodgkin's disease and of lymphocytic lymphoma first being manifested by eosinophilic effusion have been reported.
Most eosinophils have bilobed nuclei, although many nuclei have three or occasionally four lobes. The lobes are slightly larger and more neatly round than those of neutrophils. A pyknotic form in which the nucleus is represented by a solitary chromatinic mass with no lobes is occasionally seen, especially in fluids that have stood for some time after aspiration ( Fig. 19-30 ).
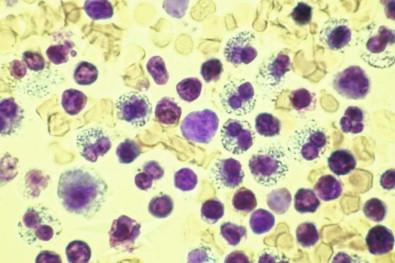
In Papanicolaou-stained smears, eosinophils are smaller than in wet films and the cytoplasmic granules are much less obvious, frequently being manifested by only a fine eosinophilic granularity. Instead, the cytoplasmic staining reaction may be light green with little or no granularity, depending on the quality of the stain; such cells are recognized as eosinophils by their size and the bilobation of their nuclei. Cell block preparations demonstrate eosinophils well ( Fig. 19-31 ). Eosinophils may undergo necrosis, although we have not observed the widespread necrosis of eosinophils in smears similar to that seen in neutrophils in empyema fluids.
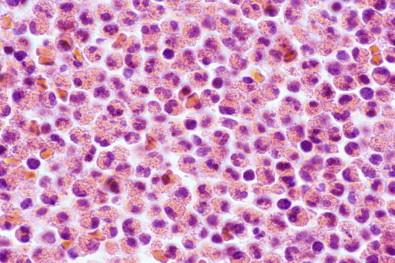
Slightly larger than neutrophils and nuclei usually bilobed
Cytoplasmic granules well seen in wet films and cell blocks
May disintegrate to form Charcot–Leyden crystals.
Basophil leukocytes and mast cells are small round cells whose cytoplasm contains round granules that are about 0.5 µm in diameter and that stain purple-red with basic dyes. In staining, these granules often acquire a color different from that of the dye, the phenomenon of metachromasia. Mast cells presumably originate in connective tissues of the body, whereas basophils originate with other leukocytes in the bone marrow, from where they enter the blood stream.
Mast cells and basophils are readily recognized with a Romanowsky stain, but they are not recognizable in Papanicolaou-stained smears or in cell blocks stained with hematoxylin and eosin. They can be recognized in toluidine blue-stained wet films ( Fig. 19-32 ), in which the granules are a delicate purple-red, quite different from the deep blue of the stain itself.
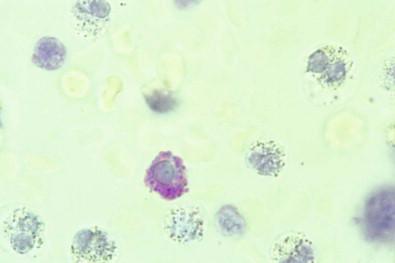
Mast cells, slightly larger than basophils, are about the size of small mesothelial cells. Their nuclei are round or oval and centrally placed, whereas the nuclei of basophils are usually bilobed. The cytoplasm of mast cells is entirely occupied with granules; the cytoplasm of basophils contains far fewer.
Identifiable in stained wet films
Small round cells with cytoplasmic granules
Granules purple-red with toluidine blue stain.
Histiocytes are found in various proportions in almost every serous effusion. Their size varies considerably, from about 15 to 100 µm in diameter, with most within the range of 20–40 µm. In smears, the typical histiocyte is easily identified by its size, eccentric round or bean-shaped nucleus, and lightly stained lacy cytoplasm ( Fig. 19-33 ). Because histiocytes are phagocytic, their cytoplasm may contain leukocytes, nuclear particles ( Fig. 19-34 ), red blood cells, carbon particles, lipid droplets, melanin, or hemosiderin.
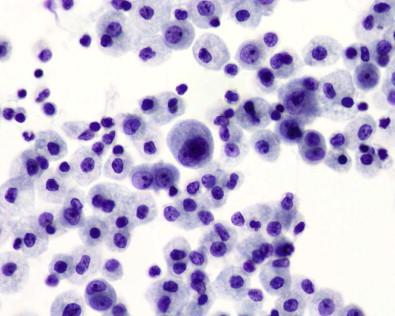
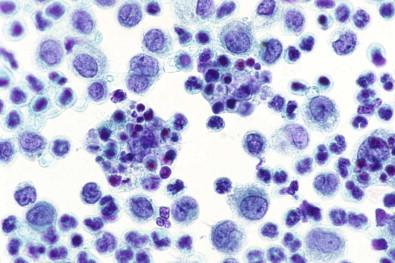
Histiocytes tend to be isolated but may coalesce, presumably because of their long microvilli becoming entangled. This usually gives a loose, sheet-like appearance to a group of macrophages. Such sheets do not have the tight cohesiveness and sharp definition of sheets of mesothelial cells; they tend to straggle at the periphery, and the overall cohesiveness of the group has a loose quality, with spaces between individual cells. Furthermore, the lacy, porous appearance of the cytoplasm contrasts sharply with the dense cytoplasm of mesothelial cells. Fig. 19-35 exemplifies in a smear of pericardial effusion the cohesive property of histiocytes, in this case to form balls; the patient had fibrinous pericarditis.
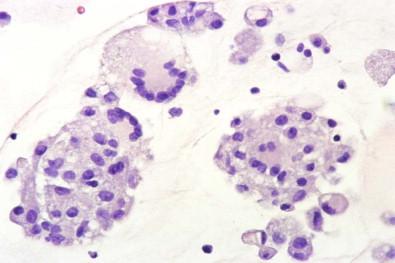
Histiocytes may contain large solitary or loculate cytoplasmic vacuoles that appear to displace the nucleus to the periphery of the cell ( Fig. 19-36 ), a picture suggestive of signet ring adenocarcinoma. In cell block preparations of a spontaneously formed clot, groups of macrophages that have become tightly packed against each other as the clot contracted may be seen; these compact groups of histiocytes may be mistaken for fragments of adenocarcinoma.
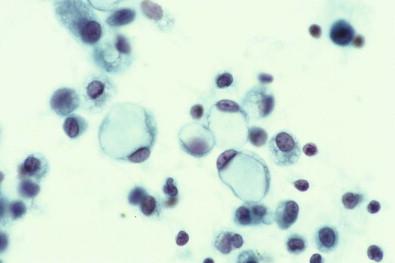
Histiocytes usually have a single nucleus, although binucleation is not uncommon. The finding of giant multinucleated histiocytes formed by fusion of a number of macrophages or by mitotic division of a nucleus is a rarity, virtually confined to effusions caused by rheumatoid pleuritis or rheumatoid pericarditis (see “ Rheumatoid Disease ,” below). Giant multinucleated mesothelial cells are seen with much more frequency, and whenever a cell is regarded as a giant multinucleated histiocyte, the question should be raised about whether it is really a mesothelial cell.
20–40 µm in diameter; larger when multinucleated
Isolated or coalescent because of long microvilli
Cytoplasm lightly stained
Ovoid, reniform, or round nuclei
Possibly phagocytic.
Lymphoid cells in effusions are similar to those in lymphoid tissue in general, with most being of the small, mature type. T and B lymphocytes cannot be distinguished in routine preparations but can be identified by immunocytochemical staining using monoclonal antibodies. Most lymphoid cells in benign effusions are of T-cell origin.
Almost every serous fluid contains at least a few lymphoid cells, readily recognized in Papanicolaou-stained smears as small cells having little or no cytoplasm and being dominated by round, deeply staining nuclei ( Fig. 19-37 ). Their size varies according to the amount of drying occurring during preparation of the smear and before fixation. In the thicker parts of a smear, where air drying is minimal, the lymphoid cells are smaller, whereas at the periphery of a smear, where some air drying is almost inevitable, lymphoid cells have a larger diameter and appear flatter and less darkly stained. This variation in size and staining reaction is not seen in cell block preparations.
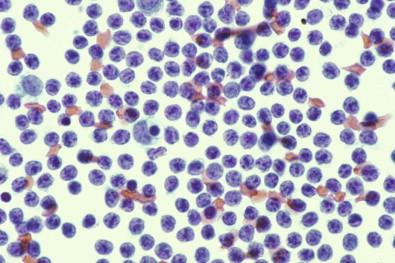
The nuclei of mature lymphocytes are neatly rounded, although some may be slightly indented or notched. Nucleoli are difficult to find in the Papanicolaou smears, although they are frequently visible in wet films. When lymphocytes are present in large numbers, it is customary to find a few less mature, slightly larger lymphoid cells. In these less mature forms, the chromatin is paler and nucleoli are visible. Cytoplasm, although scanty, is most readily seen in stained wet films and in Papanicolaou-stained smears which had undergone momentary drying before fixation.
Mature plasma cells similar to those in bone marrow may be found, usually in effusions containing many lymphoid cells ( Fig. 19-38 ). Larger, less mature-appearing plasmacytoid cells are also frequently seen in such effusions; they may contain two or more nuclei.
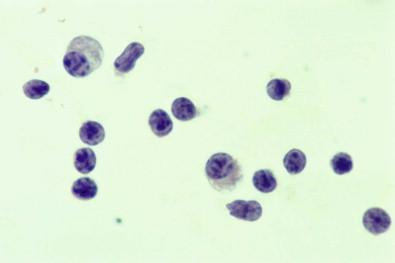
Apart from their size and shape, one of the most important features distinguishing lymphoid cells from those of small-cell carcinoma is their property of remaining separated from each other. In contrast, the cells of small-cell carcinomas form cohesive clusters. This property of lymphoid cells being separated from each other is obvious even in cell block preparations.
In contrast to purulent effusions, those dominated by non-neoplastic lymphoid cells contain only a very small percent of necrotic cells. A prolonged search under high power is usually needed to find what seems to be a necrotic lymphocyte. Such a necrotic lymphocyte may show the type of karyorrhexis consisting of tiny round particles reminiscent of a dispersed drop of mercury (mercury drop karyorrhexis). This type of karyorrhexis, difficult to find in benign lymphocytic effusions, is frequently seen in effusions containing lymphoma cells.
Small, round, with usually undetectable cytoplasm and nucleoli
Non-coalescent
Rarely necrotic in inflammatory lesions.
Morphologically normal megakaryocytes are a rare find in serous fluids ( Fig. 19-39 ). Almost all of the reported examples were associated with a myeloproliferative disorder, lymphoma, or metastatic carcinoma, situations in which bone marrow was replaced by either neoplastic cells or fibrous connective tissue, resulting in extramedullary hematopoiesis immediately adjacent to a serous cavity.
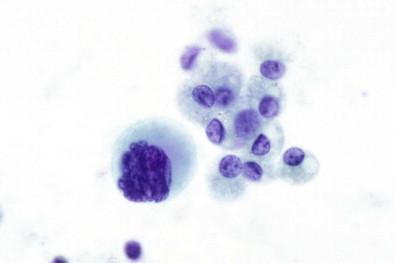
Rarely seen in serous effusions
Usually associated with a myeloproliferative disorder
Large cells with large polymorphous nuclei.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here