Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Before sclerotherapy is performed, the examiner must obtain a focused history and perform a meticulous physical examination, including inspection and palpation (level 1). This combination places the patient into a proper clinical classification, and this, in turn, will dictate therapy. For example, primary venous insufficiency is characterized by telangiectasias, reticular varicosities and varicose veins without the stigmata of chronic venous disease. In contrast, the latter has characteristic hyperpigmentation, edema, ulceration or scarring from healed ulcers. The presence of chronic disease findings prompts the examiner to be more insistent regarding further evaluation.
Confirmatory diagnostic testing is performed after the history is taken and the physical examination is performed. The handheld continuous-wave Doppler historically was a routine part of the physical examination, but it is being replaced by compact, portable duplex ultrasound visualization. At present, the vascular laboratory (level 2) provides a reliable tool for acquiring anatomic and functional information that not only confirms the diagnosis but also formulates treatment. Elements of the diagnostic vascular laboratory have become part of a routine physical examination. This approach has been confirmed in the guidelines of the Society for Vascular Surgery and the American Venous Forum.
Invasive testing such as phlebography and ambulatory venous pressure (AVP) (level 3) measurements, while of historical significance, have been relegated to a secondary role as large amounts of data are obtained from duplex ultrasound. The level of diagnostic testing performed is dictated by the severity of the clinical condition.
A focused history is the beginning of every evaluation for any patient with a suspected vascular disorder. Obtaining a clear understanding and detailed description of the symptoms is the first step. Recording of the patient's concerns and reasons for seeking treatment should also be done at this time because patients concerned by pain at first interrogation may focus on esthetics in subsequent sessions following treatment of primary sources of reflux. Symptoms of venous insufficiency have been termed nonspecific . However, patients with clear symptoms of venous insufficiency typically do not ascribe these to varicose veins. Symptoms include aching, tiredness and far-ranging discomfort in the legs, relieved by sitting and leg elevation. Such discomfort increases as the day progresses, with standing and with increased vein stretching. In women, symptoms are exacerbated on the first days of a menstrual period because elevated progesterone levels cause increased vein swelling. Symptoms may also begin in pregnancy. It has been said that for males with varicose veins the lack of progesterone or estrogen results in fewer symptoms.
The magnitude of symptoms is not dependent upon the size of the varicosities. Telangiectasias and reticular varicosities may cause symptoms identical to those of gross varicose veins. Because symptoms are due to pressure of dilated veins on somatic nerves, it is not surprising that some patients experience burning near varices. This is classically thought to be ischemic neuropathy, but it may be capillary pressure–related. A sudden disabling pain that develops on standing or sitting with legs dependent and is relieved by muscular activity is termed venous claudication , a misnomer.
Several studies have suggested that a family history of varicose veins is common in this patient population. In patients with severe manifestations of chronic venous disease, a history of deep venous thrombosis is obtained in less than one third of patients. Nonetheless, a personal and family history of a diagnosis of venous thrombosis should be requested. If present, it is important to clarify the way in which this diagnosis was confirmed (phlebography, venous duplex or simply clinically). Patients should be prompted to recall specific aspects of their medical history, such as traumatic fractures, leg swelling, need for systemic anticoagulation, illness requiring bed rest, or major surgical intervention.
An international ad hoc committee of the American Venous Forum developed the CEAP classification for chronic venous disease in 1994 ( Table 5.1 ; see also Chapter 2 ) with the goal of stratifying clinical levels of venous insufficiency. The four categories and descriptors selected for classification were clinical manifestations (C), etiology (E), anatomy (A) and underlying pathophysiology (P). The CEAP classification has been endorsed worldwide, despite its acknowledged deficiencies. It has been adopted as the standard in many clinics in Europe, Asia, South America and the United States for categorizing chronic venous disorders, guiding treatment decisions and predicting prognosis. Its weakness is the inability to distinguish between levels of smaller superficial veins. The CEAP classification was revised in 2004 and is now referred to as advanced CEAP .
| Clinical Classification | |
| C0 | No visible or palpable signs of venous disease |
| C1 | Telangiectasias or reticular veins |
| C2 | Varicose veins—separated from reticular veins by a diameter of 3 mm as the upper limit of size of a reticular vein |
| C3 | Edema |
| C4a | Pigmentation, eczema (stasis dermatitis) |
| C4b | Lipodermatosclerosis, atrophie blanche (livedoid vasculopathy) |
| C5 | Healed venous ulcer |
| C6 | Active venous ulcer |
| Each clinical class is further characterized by a subscript for the presence of symptoms (S, symptomatic) or their absence (A, asymptomatic). Symptoms include aching, pain, tightness, skin irritation, heaviness and muscle cramps, as well as other complaints attributable to venous dysfunction. | |
| Etiologic Classification | |
| Ec | Congenital |
| Ep | Primary |
| Es | Secondary (postthrombotic) |
| En | No venous etiology identified |
| Anatomic Classification | |
| As | Superficial veins |
| Ap | Perforator veins |
| Ad | Deep veins |
| An | No venous location identified |
| Pathophysiologic Classification | |
| Basic CEAP | |
| Pr | Reflux |
| Po | Obstruction |
| Pr,o | Reflux and obstruction |
| Pn | No venous pathophysiology identifiable |
| Advanced CEAP | |
| Same as Basic with the addition that any of 18 named venous segments (below) can be used as locators for venous pathology. | |
| All items listed in C should be repeated. | |
| Venous Segments | |
| Superficial Veins | |
| 1 | Telangiectasias/reticular veins |
| 2 | GSV above knee |
| 3 | GSV below knee |
| 4 | SSV |
| 5 | Nonsaphenous veins |
| Deep Veins | |
| 6 | IVC |
| 7 | Common iliac vein |
| 8 | Internal iliac vein |
| 9 | External iliac vein |
| 10 | Pelvic: gonadal, broad ligament veins, other |
| 11 | Common femoral vein |
| 12 | Deep femoral vein |
| 13 | Femoral vein |
| 14 | Popliteal vein |
| 15 | Crural: anterior tibial, posterior tibial, fibular veins (all paired) |
| 16 | Muscular: gastrocnemial, soleal veins, other |
| Perforating Veins | |
| 17 | Thigh |
| 18 | Calf |
Within the CEAP classification, the clinical assessment is described as follows:
C0: no visible or palpable signs of venous disease
C1: telangiectatic and reticular veins (see definitions in Chapter 2 )
C2: varicose veins
C3: edema
C4a: pigmentation and/or eczema
C4b: lipodermatosclerosis and/or atrophie blanche
C5: healed ulcer
C6: active ulcer
The descriptor A is added for asymptomatic patients, and the designation S is used in case of symptoms.
All clinical features must be reported in the advanced CEAP classification; for example, a patient with telangiectasias, varicose veins, edema, pigmentation, active ulcer and pain is classified as C6S in the basic (classical) CEAP but as C1,2,3,4a,6,S in the advanced (revised) CEAP. This approach carries much more information.
Etiology is reported by descriptor E:
Ec: congenital (usually present at birth)
Ep: primary (degenerative, typically varicose veins)
Es: secondary (as in postthrombotic syndrome)
En: no venous etiology identified
The anatomy of the venous network involvement is described by the A classification, with involved venous segments recorded with a number (see Table 5.1 ):
As: superficial veins
Ap: perforator veins
Ad: deep veins
An: no venous location identified.
Last, pathophysiology is reported with the P descriptor, again with involved venous segments recorded by numbers (see Table 5.1 ):
Pr: reflux
Po: obstruction
Pr,o: reflux and obstruction
Pn: no venous pathophysiology recognized
When performing vascular testing, the data must include the date of the patient's evaluation. Classification of the level of examination performed is as follows: L1 (level 1) is clinical examination plus continuous wave Doppler, L2 is noninvasive (duplex ultrasound, plethysmography) and L3 includes complex imaging such as computed tomography (CT) and magnetic resonance imaging and invasive investigations such as venograms, intravenous ultrasound and blood pressure measurement.
The first step in evaluating a patient with venous disease is to establish his or her clinical class, which progresses from cosmetic concerns to chronic venous insufficiency. The next step is to correlate any symptoms, which place the limb being examined into one of the classes shown in Table 5.1 . The patient's clinical class will dictate the need for further evaluation. In patients with telangiectasias (class 1 or 2), the evaluation can be limited to a physical examination and evaluation of the superficial venous system with a handheld continuous-wave Doppler. Imaging of 83 limbs with clinical evidence of only telangiectatic vessels demonstrated that nearly 25% had insufficiency of the great saphenous vein (GSV) or small saphenous veins (SSVs), which was not apparent on physical examination. Patients with symptomatic class 2 varicosities and classes 4, 5 and 6 skin changes require a duplex venous reflux examination because surgical intervention may be indicated. Recalcitrant cases may require more extensive imaging studies to detect venous occlusive disease. Physiologic testing is relegated to documentation rather than to diagnosis, and phlebography should be performed only when venous reconstruction is considered.
The physician should discuss a patient's prior treatment for venous disease. However, he or she must realize that although proper ligation, with or without stripping of the main saphenous trunks, implies that reflux through the saphenofemoral junction (SFJ) and saphenopopliteal junction (SPJ) has been prevented, this is not always the case. Joshi et al found that reflux in the residual GSV was the most common cause of recurrent varicose veins in 419 legs among 298 patients who underwent ligation of the SFJ and stripping of the GSV. Up to 27% of patients have a duplication of the GSV, indicating that the removal of the GSV may be followed by the development of varicosity in the remaining GSV.
In 20% to 40% of patients, the SSV has a variable termination that is not in the popliteal vein at or above the popliteal fossa. Therefore, the actual SPJ must be correctly located, or it will lead to an apparent rapid recurrence with varicose changes occurring in the remaining segment of the SSV and its tributaries. In a number of patients, a recurrence of varicose veins in the upper thigh may be a result of incomplete ligation and division of the other tributaries arising at the level of the SFJ or of failure to accomplish the ligation flush with the femoral vein. In fact, in a review of 341 extremities that underwent repeat operations for varicose veins, Lofgren et al found that 61% had inadequate ligation. Thus, it is imperative that, even in a patient with a history of ligation, division and stripping, an examination for reflux through the SFJ and SPJ be performed.
The physician should consider responses to and complications due to all treatment modalities (e.g., sclerotherapy, laser). Certain complications, such as ischemic ulceration caused by inadvertent injection into an arteriovenous malformation, can be avoided more easily if a diagnosis is made before treatment. Given the predilection for these to occur in a particular anatomic distribution(s), the physician might avoid treating that area or use greater caution in the previously affected region. A history of prior hyperpigmentation, blushing or poor response to a particular sclerosing agent may support a variety of changes in treatment protocol, such as altering the sclerosant concentration, increasing the strength or duration of compression and paying greater attention to posttreatment thrombectomy.
The presence and severity of symptoms does not necessarily correlate with the size or severity of varicose veins present. Symptoms usually attributable to varicose veins include feelings of heaviness, tiredness, aching, burning, throbbing, itching and cramping in the legs ( Box 5.1 ). These symptoms are generally worse with prolonged sitting or standing and are improved with leg elevation or walking. A premenstrual exacerbation of symptoms is also common. Patients typically find relief with the use of compression in the form of either graduated support hose or an elastic bandage, but compliance can be a challenge. Weight loss or the commencement of a regular program of lower extremity exercise may also lead to alleviation of the severity of varicose vein symptoms. Clearly, these symptoms are not specific, as they may also be indicative of a variety of rheumatologic or orthopedic problems. However, their relationship to lower extremity movement and compression is usually helpful in establishing a venous origin for the symptoms. Significant symptoms suggestive of chronic venous disease should prompt further evaluation for valvular insufficiency and calf muscle pump dysfunction. If a venous etiology is suspected but all examinations are negative, repeat examination during a symptomatic period is warranted and often fruitful.
Aching
Heaviness, tiredness
Pain (throbbing, burning, sharp, tingling)
Itching
Cramping
Tightness
The recent development of an extremely painful area on the lower leg associated with an overlying area of erythema and warmth may be indicative of lipodermatosclerosis, which may be associated with insufficiency of underlying perforator veins or reflux from a proximal point; thus, examination for underlying perforator vein reflux should be performed. Lipodermatosclerosis may precede ulceration and has been shown to be improved by stiff compression and certain pharmacologic interventions.
Rarely, patients with a history of iliofemoral thrombophlebitis who describe ‘bursting’ pain with walking may have ‘venous claudication’. In these patients, an evaluation for persistent hemodynamically significant obstruction, possibly treatable with venous bypass surgery, is appropriate.
Complications such as ulceration and hemorrhage should be discussed with the patient, because this provides additional insight into both the severity and the probable locations of abnormality within the venous system. A history of ulceration of the medial aspect of the lower leg should prompt further examination of the GSV trunk, whereas involvement of the lateral aspect of the lower leg suggests an abnormality in the SSV in addition to the deep and perforating vein systems. A history of hemorrhage from telangiectasias in a particular area suggests further examination for underlying incompetent perforators and is an indication to treat all suspicious telangiectasias.
More extensive evaluation can provide essential information regarding both venous anatomy and function. Abnormalities of the superficial, perforating or deep systems can be diagnosed, and the exact sites of valvular insufficiency within the various systems (and therefore the sites where treatment must be directed) can be determined. The hemodynamic significance of each of these abnormalities may be defined, and the effect of correcting each site of reflux may be assessed. Insufficiency at a particular site within the venous system may be found to have no importance in the patient's pathologic venous hypertension or symptoms. This site may or may not be incorporated into the treatment plan to minimize the number of treatments. With the examination techniques discussed in the following sections, the result of sclerotherapy may be documented in a more accurate and sensitive manner than with simple observation and palpation. Treatment success may be enhanced significantly, with the goal being restoration of normal venous flow. If the presence of deep venous thrombosis (DVT) and/or deep venous valvular insufficiency is detected, these are absolute and relative contraindications to sclerotherapy. If diagnosed, serious complications in these noncandidates for sclerotherapy can be avoided.
The best way to approach examination of the venous system before sclerotherapy is to be methodical. Although the exact method of examination is a matter of personal preference, a systematic approach is advisable.
Using skills of clinical practice, the practitioner can obtain a degree of information regarding overall venous outflow from the leg, the sites of valvular insufficiency, the presence of primary versus secondary varicose veins and the presence of DVT.
The screening physical examination consists of careful observation of the legs. Any patient with the following conditions should be examined more fully: large varicose veins; bulges in the thigh, calf or the inguinal region, representative of incompetent perforating veins (IPVs) or a saphena varix ; signs of superficial venous hypertension, such as an accumulation of telangiectasias in the ankle region (corona phlebectatica); or any finding suggestive of venous dermatitis (pigmentation, induration, eczema). This includes patients with obvious cutaneous signs of venous disease, such as venous ulceration, atrophie blanche or lipodermatosclerosis. An obvious but often forgotten point is the necessity of observing the entire leg and not confining the examination simply to the area that the patient feels is abnormal. The importance of this is demonstrated in the patient shown in Figure 5.1 . This patient came for treatment of an obviously dilated anterior thigh vein, but further inspection revealed a saphena varix with incompetence at the level of the SFJ, thus defining the first step in her treatment. Similarly, patients often seek treatment of specific clusters of telangiectasia and do not notice the underlying reticular veins that should be treated initially or at the same time (see Chapter 12 ).
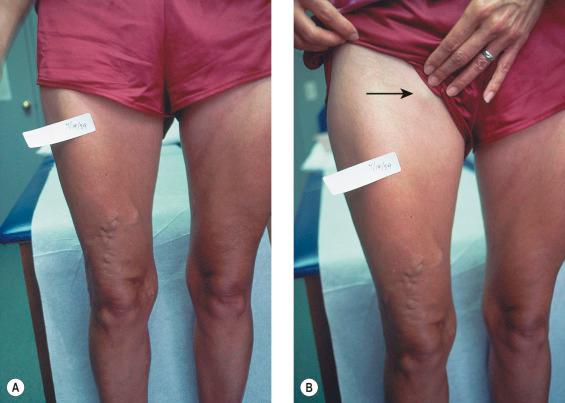
Because the veins of the leg empty into the pelvic and abdominal veins, inspection of the abdomen is very important. Dilation of veins on the abdominal wall or across the pubic region suggests an old iliofemoral thrombus or, rarely, a developmental anomaly of the venous system. Dilated veins along the medial or posterior aspect of the proximal thigh or buttocks most often arise from varicosities involving the pudendal or other pelvic vessels. These can be associated with vulvar varices that may remain symptomatic after the completion of the pregnancy during which they formed. Enlarged veins in the thigh or buttocks may also be quite symptomatic and respond well to treatment.
Historically, tests of venous function have been part of the physical examination of venous insufficiency. These tests have been slowly abandoned due to lack of specificity and sensitivity. As the ‘stethoscope’ of the venous examination, the continuous-wave Doppler examination has replaced most of these tests, while confirmatory duplex testing has supplanted all. An educated physician who treats venous insufficiency must have knowledge of these tests and their physiologic background. Also, knowledge of the Trendelenburg test (or Brodie-Trendelenburg test) is important for understanding venous physiology and is of historical significance.
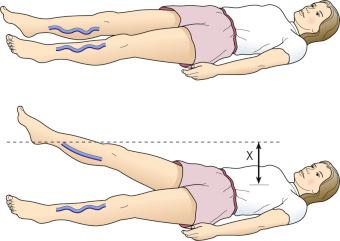
For the Trendelenburg test, a tourniquet may be placed around the patient's proximal thigh while the patient is standing. The patient then assumes the supine position with the affected leg elevated 45 degrees. The tourniquet is removed, and the time required for the leg veins to empty, which is indicative of the adequacy of venous drainage, is recorded.
When the affected leg is compared with the contralateral leg, the method just described may demonstrate a degree of venous obstructive disease. Another approach is to elevate the leg while the patient is supine and observe the height of the heel in relation to the level of the heart that is required for the prominent veins to collapse ( Fig. 5.2 ). Unfortunately, neither procedure is sufficiently sensitive or accurate or able to differentiate acute from chronic obstruction, which means neither of them is of much assistance in current medical practice. This emphasizes the important role of duplex ultrasound in modern evaluation of the superficial venous system. One study found that pneumatic tourniquets occluded only 27% of saphenous trunks. Several other physical examination maneuvers, described below, can provide information on the competence of the venous valves.
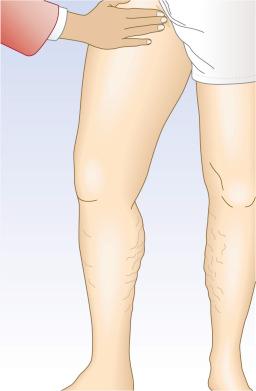
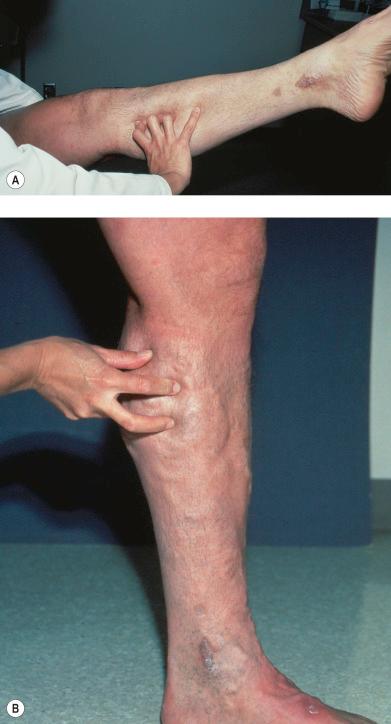
With the cough test, one hand is placed gently, without exerting pressure, over the GSV or SFJ, and the patient is asked to cough or perform a Valsalva maneuver ( Fig. 5.3 ). Simply palpating an impulse over the vein being examined may be indicative of insufficiency of the valve at the SFJ and below to the level of the palpating hand. This test, however, is not applicable to the examination of the SSV and SPJ (see following section). Palpation of a thrill during this maneuver is generally more diagnostic. One study found a low sensitivity of 0.59 and low specificity of 0.67 with this test.
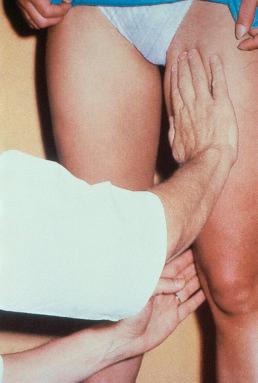
With the percussion/Schwartz test, one hand is placed over the SFJ or SPJ while the other hand is used to tap very lightly on a distal segment of the GSV or SSV ( Fig. 5.4 ). The production of an impulse in this manner implies insufficiency of the valves in the segment between the two hands. Confirmation of valvular insufficiency can be achieved by tapping proximally while palpating distally. This test can also be used to detect whether an enlarged tributary is in direct connection with the GSV or SSV by palpating over the main trunk and tapping lightly on the dilated tributary, or vice versa. The presence of a direct connection results in a palpable impulse being transmitted from the percussing hand to the palpating hand. As might be expected, these tests are far from infallible. In a study of 105 limbs, Chan et al found that these clinical examination techniques correctly identified SFJ incompetence in only 82% of limbs. False negatives were believed to be caused primarily by obesity and previous groin surgery with resultant scarring. However, false positives were the result of variations in venous anatomy, such as a dilated tributary emptying into the common femoral vein (CFV) adjacent to the GSV or the absence of valves in an otherwise normal CFV and external iliac vein (seen in 5–30% of patients) (M. Schadeck, personal communication, September 1989). Researchers in another study found that this test had a low sensitivity of 0.59 and a high specificity of 0.92. A further source of error with the cough and/or percussion test is simply a misinterpretation of the muscle contraction that occurs with coughing as a reflux impulse.
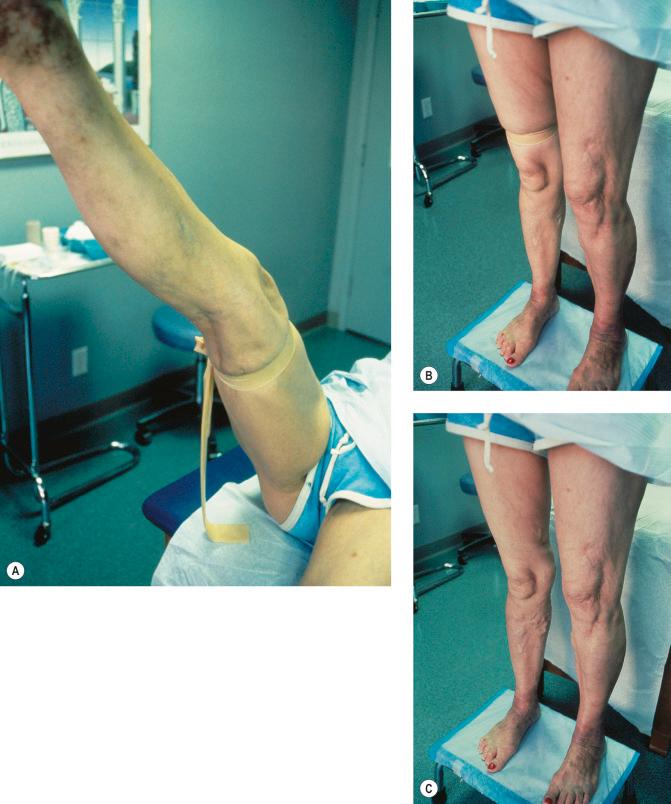
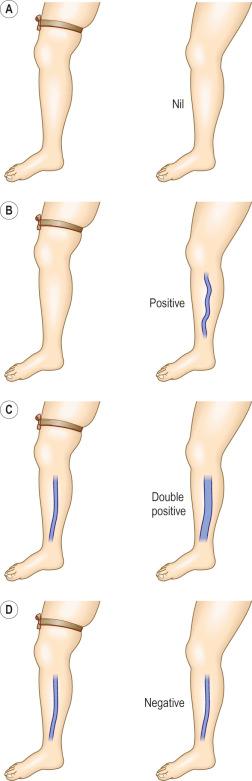
The Brodie-Trendelenburg test traditionally involves the manual obstruction of the proximal end of the GSV (or SSV) while the patient lies supine with the leg elevated, after the vein is stroked in a cephalad direction to empty it of blood. The patient then assumes the standing position, and the leg is observed for 30 seconds ( Fig. 5.5 ). In a ‘nil’ test, the veins fill slowly from below and release of the compression does not result in rapid filling from above, indicating competence of valves in deep and perforating veins and at the SFJ ( Fig. 5.6A ). Rapid filling of the GSV or more distal tributaries that occurs only after release of the compression constitutes a ‘positive’ test, indicating the presence of an insufficient valve at the SFJ ( Fig. 5.6B ). In the ‘double-positive’ test, some distention of the veins occurs within the initial 30 seconds while the compression is maintained, as well as additional filling once the compression is released ( Fig. 5.6C ). This is taken as evidence of incompetent deep and perforating veins as well as reflux through the SFJ. A ‘negative’ test occurs when the veins fill within the initial 30 seconds with no increased filling after the compression is released, implying only deep and perforating valvular insufficiency ( Fig. 5.6D ). The reverse may not be true; that is, filling in longer than 30 seconds does not imply competence of perforating veins. In a study of 901 extremities, Sherman found that 95% had a nil Trendelenburg test, but surgical exploration later showed incompetent perforators in 90% of these patients. Another study showed a high sensitivity of 0.91 with a low specificity of 0.15.
The Brodie-Trendelenburg test thus can be an important method of localizing the most proximal site of reflux in most dilated superficial veins by obstructing the GSV, SSV or whichever vein is suspected of refluxing into a more distal vein. The physician can also place the examining finger over palpable fascial defects in the leg while the patient is supine and then release the obstructions one by one after the patient is standing. This allows the sites of insufficient perforators, or ‘points of control’ (considered so crucial in Fegan's technique of sclerotherapy), to be defined, because the superficial veins distal to the insufficient perforator fill rapidly once the obstructing fingers are removed (see Chapter 9 ).
With this technique, described well in many papers, the practitioner first marks on the leg the sites of all dilated varicosities. The patient then assumes the supine position with the leg elevated to approximately 60 degrees to empty the veins. After at least 20 seconds, or when the distended veins are flattened, the leg is gently and rapidly palpated to detect any defects in the fascia. With experience, these can be detected easily as locations that allow the entrance of the examining finger without the use of any pressure. Fascial defects can be caused by many abnormalities other than perforating veins; thus, the practitioner continues the examination by compressing the individual fascial defects with his or her fingers and then having the patient stand. The fingers are then released one by one, starting with the most distal defect, and rapid filling of more distal varicosities is noted ( Fig. 5.7 ). Those defects that cause distal filling when released are assumed to correspond to sites of IPVs. In the presence of a dilated GSV or SSV, these points of reflux first must be controlled with either digital compression or a tourniquet to evaluate the lower volume reflux through the perforators. For the evaluation to be helpful, compression of the defects must first cause sustained flattening of the varicosities when the patient initially stands. If the veins fill before any of the fingers are released, the test must be restarted and other sites compressed until the sites responsible for the reflux are located. This examination is associated with 50% to 70% accuracy compared with findings at surgical exploration. Repeated examination at different times and improvement of edema allow the detection of increased numbers of perforators.
A clever variation of the Brodie-Trendelenburg technique was proposed by Bracey in 1958 ( Fig. 5.8 ). He used a flat, 3.8-cm-wide rubber tourniquet and two rubber rings covered with latex, with inside diameters of 7 cm and 8.2 cm. The smaller ring is used between the ankle and knee and may also be used for the thigh if the patient is thin. If not, the larger ring is used for the thigh. With the patient standing, the small ring is rolled over the foot to just above the ankle, and the rubber tourniquet is then placed below the ring to obstruct any upward flow of blood through the superficial veins. The small ring is then slowly rolled upward, emptying the superficial veins as it moves. As soon as it passes an IPV, the blood enters the superficial vein that connects with it, causing a dilation of the vein. The exit site of the perforating vein may then be marked. This reflux of blood can be accentuated by asking the patient to repetitively dorsiflex the foot. When the ring reaches the knee, the tourniquet is moved up to the knee, just below the ring. Either the smaller or larger ring is then used similarly to examine the thigh.
The Perthes test has several uses, including distinguishing between venous valvular insufficiency in the deep, perforator and superficial systems and screening for DVT ( Table 5.2 ). To localize the site of valvular disease, the physician places a tourniquet around the proximal thigh with the patient standing. When the patient ambulates, a decrease in the distention of varicose veins suggests a primary process without underlying deep venous disease because the calf muscle pump effectively removes blood from the leg and empties the varicose veins. Secondary varicose veins do not change caliber (if there is patency of the deep venous system) because of the inability to empty blood out of the veins as a result of impairment of the calf muscle pump. In the setting of a concurrent DVT, these veins may increase in size. If there is significant chronic or acute obstructive disease in the iliofemoral segment, the patient may note pain (venous claudication) as a result of the obstruction to outflow through both the deep and superficial systems. Information regarding the presence of deep venous valvular insufficiency and thrombosis is important to note to aid in patient selection. This avoids causing catastrophic complications, such as pulmonary embolism resulting from an undiagnosed and worsened DVT or venous claudication caused by further impairment of venous return. Indeed, these two complications are serious enough to warrant the use of a much more sensitive and accurate method; therefore, the Perthes test is now of more historical than actual clinical importance.
| Finding | Interpretation |
|---|---|
| Decreased diameter of varicose veins | Primary varicose veins |
| No change in diameter of varicose veins | Secondary varicose veins |
| Deep venous patency | Impairment of calf muscle pump |
| Increased diameter of varicose veins | Deep venous obstruction |
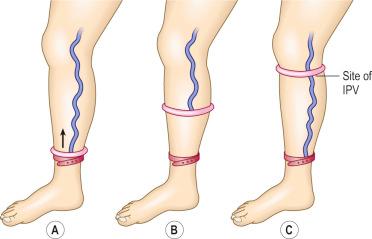
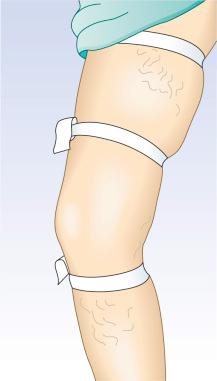
To test for perforator valvular defects, the physician may embellish the traditional Perthes test by placing a tourniquet around the calf just below the popliteal fossa. If the dilated superficial veins in the calf and ankle become less prominent as the patient ambulates, this implies that the blood is being drawn into the deep system through competent perforating veins. However, if the veins become increasingly dilated, the perforating veins must be incompetent. A more involved test, the Mahorner-Ochsner comparative tourniquet test, similarly localizes the site(s) of reflux by observing the leg while the patient walks with the tourniquet placed at various levels on the leg (upper, middle and lower thigh) ( Fig. 5.9 ).
The preceding decades have provided a wealth of noninvasive technology that has revolutionized vascular diagnosis. A thorough description of all these techniques is beyond the scope of this book, but those not presented here may be found in several excellent texts. Some of the new technologies have real utility in the everyday performance of sclerotherapy, and the following discussion attempts to acquaint the reader with their uses and limitations.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here