Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Pancreatic ductal adenocarcinoma (PDAC) comprises the fourth most common cause of cancer lethality in Western countries with a less than 5% reported five-year survival . General increases in the life expectancy of citizens in industrialized countries primarily lead to an elevation in reported cases of PDAC with an annual incidence rate similar to its death rate. PDAC is characterized by late diagnosis, aggressive local invasion, early systemic dissemination, and resistance to chemo- and radiotherapy . Despite advances in surgical techniques and therapeutic regimens in the past 30 years, no substantial improvement in the survival of PDAC patients has been noted. We aim to provide an overview on the current status and major challenges of local and systemic treatments for PDAC. Further, novel rationales behind the development of promising next-generation therapy strategies for pancreatic cancer are explored.
Following diagnosis of PDAC, planning the patient’s treatment regimen is optimally based on the results of diagnostic and imaging tests, and in consultation with experienced multidisciplinary professionals, preferably in high-volume care centers. Variations in clinical evaluation and local management of pancreatic tumors exist among medical institutions in Europe and the United States. Nevertheless, general guidelines have been established for selection of the best treatment options for different subentities of pancreatic cancer ( http://www.nccn.org ). These involve proper assessment of tumor stage at the time of diagnosis, particularly with respect to discrimination between resectable, locally advanced or metastatic, unresectable disease. Accordingly, the assignment of stage-specific treatment strategy could be appropriately designed ( Figure 3.1 ).
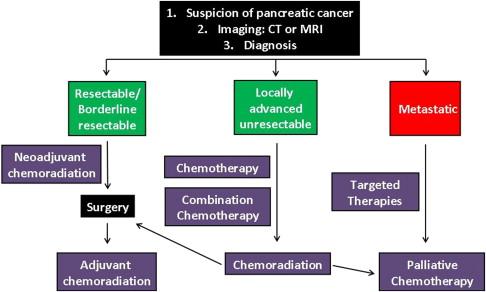
Only approximately one-fifth to one-sixth of patients diagnosed with PDAC are currently eligible for surgical resections, most of which display marginal clinical benefit and postoperative disease recurrences either locally (50–80%), peritoneally (25%), and/or in the liver (50%) . Identification of patients with resectable disease constitutes an initial challenge in the adequate management of pancreatic cancer. Classification of the resection state of cancer patients is highly dependent on preoperative radiographic imaging such as abdominal computed tomography (CT) and magnetic resonance imaging (MRI), as well as on endoscopic ultrasound-guided fine-needle biopsy, minimally invasive laparoscopy, and serum CA 19-9 levels . Abdominal CT imaging (optimized for pancreas) is the preferred method at most centers as the diagnostic tool for defining resectability . Based on CT assessment, tumors are considered resectable when three major criteria are achieved: (1) localized intrapancreatic disease; (2) no involvement of, or extension into, the celiac axis or superior mesenteric artery (SMA), and (3) patent confluence of the superior mesenteric-portal vein (SMPV) .
Minimally invasive laparoscopic distal pancreatectomy (LDP) and laparoscopic pancreaticoduodenectomy (LPD) were originally introduced in the mid-1990s . Studies of LDP have thereafter primarily focused on evaluating clinical outcome in patients with low-grade lesions rather than PDAC. Despite decreased blood loss, shortened hospitalization, and reduced complications, as recorded by a meta-analysis comparing LDP to open surgery, mortality rates observed for both operations were similar . Improvements made later in laparoscopic devices and incorporation of robotic platforms optimized the speed, mobility, precision, and aptitude of these minimally invasive surgeries as concluded by a comprehensive study of robotic-assisted LDP . However, decisive conclusions on the efficacy of LDP have been impeded by the particular nature and position of the pancreas, as well as its proximity to vascular structures and the insufficiency of available data outcomes.
Improved local tumor control and increased overall survival were reported in patients with gastrointestinal (GI) malignancies (pancreatic-rectal-stomach) exposed to chemoradiation succeeding their surgery . This improved outcome might be attributed to potency of chemoradiation in targeting microscopic lesions within the primary tumor area, or in decreasing the possibility of recurrence in regional lymph nodes . Up to approximately 90% success rate in controlling local disease has been reported in a population of pancreatic cancer patients after accurate diagnosis and multidisciplinary treatment consisting of surgery and chemoradiation .
To date, no standard adjuvant treatment has been unambiguously accepted for treatment of pancreatic cancer, in part due to the lack of sufficiently powered randomized trials and heterogeneity of in therapy protocols. An overview of some key clinical studies is provided in Table 3.1 . While chemotherapy is considered the standard treatment in Europe, centers in the United States provide chemotherapy alone, chemoradiotherapy, or chemotherapy followed by chemoradiotherapy . The efficacy of 5-fluorouracil (5-FU) and 5-FU-based chemoradiation after surgical resection of pancreas tumor was evaluated in an initial clinical trial that was first reported in 1985 and was led by the Gastro-Intestinal Tumor Study Group (GITSG) . As compared to the observation group (surgery alone), patients who received the adjuvant external beam radiotherapy (EBRT, 40 Gy) delivered in split course and chemotherapy (500 mg/m 2 5-FU), displayed significantly longer median survival times (20 vs. 11 months) and two-year survival (43% vs. 19%). However, this study was terminated due to the poor accrual of enrolled candidates (43 patients in eight years). Treatment of 30 additional patients in an expanded trial yielded a median survival of 18 months and a two-year survival of 46%, thus confirming the findings of the initial GITSG trial.
| Trial | Year | Patients | Assigned Treatment | Overall Survival (months) | p -value |
|---|---|---|---|---|---|
| GITSG | 1985 | 43 | 5-FU chemoradiation Observation (surgery alone) |
21 10.9 |
0.035 |
| GITSG expanded | 1985 | 30 | 5-FU chemoradiation | 18 | n/a |
| EORTC-40891 | 1999 | 114 | 5-FU chemoradiation Observation (surgery alone) |
17.1 12.6 |
0.09 |
| ESPAC-1 | 2004 | 289 | 5-FU/Leucovorin chemotherapy No chemotherapy 5-FU chemoradiation No chemoradiation |
20.1 15.5 15.9 17.9 |
0.009 0.05 |
| RTOG-9704 | 2006 | 388 | Gemcitabine → 5-FU chemoradiation → Gemcitabine 5-FU → 5-FU chemoradiation → 5-FU |
20.6 16.9 |
0.033 |
| CONKO-001 | 2007 | 354 | Gemcitabine chemotherapy Observation (surgery alone) |
22.1 20.2 |
0.06 |
| ESPAC-3 | 2010 | 1088 | Gemcitabine chemotherapy 5-FU/Folate chemotherapy |
23.6 23.0 |
0.39 |
In 1999, a second trial was reported by the European Organization for Research and Treatment of Cancer (EORTC-40891) investigating pancreatic head and peri-ampullary cancer . Patients were randomized to receive either 40 Gy EBRT in a split dose with 5-FU or no treatment (observation arm) after surgery. The median survival was 19 months for observation vs. 24.5 months in the chemoradiation group ( p = 0.2). Due to the ambiguity of data from both trials, the European Study Group for Pancreatic Cancer (ESPAC-1) initiated a randomized multicenter clinical trial investigating the effectiveness of chemoradiotherapy and chemotherapy after pancreas cancer resection . Patients were randomly allocated into four treatment groups: chemotherapy (425 mg/m 2 5-FU, 20 mg/m 2 leucovorin); chemoradiotherapy (40–60 Gy EBRT split course, 425 mg/m 2 5-FU, 20 mg/m 2 leucovorin); chemoradiotherapy followed by chemotherapy; or no further treatment. This study reported beneficial effects of adjuvant 5-FU-based chemotherapy as compared to surgery alone. However, addition of radiotherapy was associated with inferior survival as compared to surgery alone or adjuvant chemotherapy arms. The outcome of the ESPAC-1 trial is controversially debated in the community. In particular, the rationale behind different radiotherapy schemes administered in these trials, i.e., split-course radiotherapy, was conceived in the 1970s. In analogy to the EORTC-40891, for example, patients were treated for two weeks with 5 × 2 Gy fractions, then the progression of tumor was assessed, and eventually after an interval of two weeks, the treatment was repeated to a total dose of 40 Gy. Another example would have been two courses of 20 Gy in 10 fractions each followed by a three to four week rest period. Depending on the response and the patient’s clinical status, another 10–20 Gy in 5–10 fractions was administered as a final boost. Moreover, irradiation was administered sometimes with only two fields (anterior-posterior and posterior-anterior). Likewise, 5-FU-based chemotherapy was started, e.g., prior to radiotherapy as bolus or at 25 mg/kg per 24 h (at max. daily dose of 1500 mg). This is in contrast to continuous administration of 5-FU or gemcitabine-based chemotherapy delivered with modern radiotherapy techniques, i.e., four or more fields, allowing, e.g., integrated boost concepts using IMRT or additional dose escalation with IORT in doses of 45–54 Gy. Therefore, the relatively high local tumor recurrence rates constituting 62% of all recurrences (as compared to the more common pattern of distant recurrences observed in this disease), and the excess mortality observed primarily in the second year, suggest a potential role for late radiation toxicity in surrounding normal organs, such as kidney damage, and suboptimal doses prescribed to the tumor region among possible mechanisms discussed for the poor outcome of the radiotherapy arm in ESPAC-1 trial . In conclusion, the low quality of radiation administered at different centers in the ESPAC-1 clearly indicates the need for randomized prospective trials using modern radiotherapy techniques to ultimately evaluate the role of this therapeutic modality in the adjuvant treatment of PDAC.
The Radiation Therapy Oncology Group (RTOG-9704) investigated the efficacy of gemcitabine (1000 mg/m 2 /week) or 5-FU (250 mg/m 2 /day) chemotherapy administered before and after 5-FU-based chemoradiation (50.4 Gy EBRT) in 388 patients with pancreatic head cancers . Evaluation of data revealed prolonged overall survival in patients treated with gemcitabine (20.6 months median survival) as compared to 5-FU (16.9 months median survival). In contrast to the European ESPAC trials, the RTOG investigators have shown that failure in adhering to the specified radiation therapy protocols was associated with decreased survival in RTOG-9704 .
Additional evidence for the beneficial effects of adjuvant chemoradiation in PDAC is further provided by two large volume U.S. centers, Johns Hopkins University and the Mayo Clinic, which together investigated more than 1200 patients. They found an improved overall survival (OS) after 5-FU-based adjuvant chemoradiation (50.4 Gy EBRT) as compared to surgical resection alone (median survival 21.1 vs. 15.5 months, p < 0.001) . Their data indicate sustained benefits of chemoradiation with improved two- and five-year OS rates as compared to surgery alone (44.7% vs. 34.6%; 22.3% vs. 16.1%, p < 0.001).
Encouraging five-year survival rate (55%) was obtained by an adjuvant interferon-based chemoradiation phase II trial performed at the Virginia Mason University and followed up by the American College of Surgeons Oncology Group (ACOSOG) . However, these data were not confirmed by the randomized phase III trial led by the University of Heidelberg . In this study, patients were treated after surgery with cisplatin, interferon alpha2b, 5-FU, and radiotherapy followed by two cycles of 5-FU monotherapy (arm A) or with six cycles of 5-FU monotherapy (arm B). Median survival for the patients who received at least one dose of the study medication was 32.1 months in the combination arm A and 28.5 months in 5-FU alone arm B ( p = 0.49). Patients treated with the combination therapy more frequently experienced grade III/IV toxicities as compared to the 5-FU alone arm (85% vs. 15%). Nonetheless, this study demonstrates that management of operable PDAC patients with surgery and chemotherapy and/or chemoradiotherapy has improved over time as the relatively high median survival in both arms suggest. Currently, a phase III trial conducted by the RTOG-0848 is joining American and European efforts (EORTC) in evaluating efficacy of postoperative erlotinib treatment after five months of gemcitabine-based chemoradiation .
Toward further development in the direction of chemotherapy alone in the adjuvant setting, the ESPAC-3 trial compared the influence of gemcitabine to that of 5-FU/folate–based adjuvant chemotherapies . Although no differences in median survival were observed for both treatment modalities (23.6 vs. 23 months), better tolerance to gemcitabine (revealed by fewer adverse effects) was detected in corresponding patients. In a randomized trial initiated in Germany and Austria by the Charité Onkologie Clinical Studies in GI Cancer-001 (CONKO-001), patients treated with gemcitabine also revealed significantly longer median disease-free survival (13.4 months) as compared to the surgery alone group (6.9 months) . Consequently, based on these studies postoperative chemotherapy has become standard in treatment of operable PDAC in Europe. Later on, the Japanese Study Group of Adjuvant Therapy for Pancreatic Cancer conducted a randomized phase III trial comparing gemcitabine with surgery, but only in 118 patients with resectable disease . Treatment with gemcitabine (1000 mg/m 2 ) contributed to prolonged disease-free survival as compared to surgery alone (11.4 vs. 5 months).
Several rationales favor the implementation of radiochemotherapy prior to surgery in a “neoadjuvant” setting . First, surgery itself is associated with increased risk of morbidity and mortality, which may consequently result in delayed/canceled postoperative adjuvant treatment. Therefore, preoperative chemo- or radiotherapy may ensure full dose delivery, unlike for adjuvant treatment. Second, development of metastatic disease during the time frame of neoadjuvant therapy would select patients with advanced systemic disease that might benefit less from more aggressive local regimens including surgery. Third, improved delivery of chemotherapeutic agents may be possible by the still intact blood supply prior to surgery. Fourth, disrupted perfusion caused by the interruption of the tumor’s blood flow after initial surgery reduces oxygen levels within tumors, rendering cells resistant to radio- as well as chemotherapy. Fifth, reduced adverse and toxic effects have been reported in patients with GI malignancies exposed to preoperative treatment regimens, as shown by a German Rectal Cancer Study .
Successive phase II clinical trials testing the efficacy of neoadjuvant therapy have been initiated at MD Anderson Cancer Center . In 1992, efficacy of 5-FU (300 mg/m 2 /day) and irradiation (50.4 Gy, in certain cases intraoperative radiation therapy (IORT)) was tested in 28 patients with cytologic or histologic proof of localized adenocarcinoma of the pancreas head . Four to five weeks of neoadjuvant chemoradiation provided a window for metastatic development in approximately 40% of enrolled patients, as displayed by pathological restaging. The remaining patients were treated with 5-FU (300 mg/m 2 /day) and irradiated with rapid fractionation (30 Gy in 2 weeks, i.e., 10 daily fractions of 3 Gy, ±10–15 Gy intraoperative radiotherapy (IORT) in 74% of patients) . Around 40% of subjects displayed a pathologic partial response to treatment (>50% nonviable tumor cells). Both irradiation modalities (standard 50.4 Gy vs. rapid ) yielded similar local tumor control. Median survivals recorded were 18 and 25 months for standard and rapid chemoradiation regimens, respectively. This study concluded that favorable locoregional control may require a combination of preoperative 5-FU/rapid fractionation chemoradiation, surgery, and IORT . Investigation of the influence of paclitaxel (60 mg/m 2 ) in combination with rapid fractionation (30 Gy EBRT), conducted later on resectable pancreas cancer, showed no advantage of this neoadjuvant therapy over 5-FU-based chemoradiation . Of note, paclitaxel-based neoadjuvant chemoradiation demonstrated a higher relative toxicity (nausea, dehydration, vomiting) as compared to conventional 5-FU-based regimens. Subsequently, two phase II trials, including 86 and 90 resectable pancreatic cancer patients, evaluated preoperative chemoradiation using gemcitabine alone (400 mg/m 2 ) or in combination with cisplatin (30 mg/m 2 ) and radiotherapy (30 Gy in 10 fractions), respectively . Patients treated with gemcitabine displayed better pathological findings, i.e., higher rates of microscopic tumor clearance (R0 resection) and longer overall survival than those treated with 5-FU– or paclitaxel-based neoadjuvant treatments. Addition of cisplatin with gemcitabine had no obvious improvement on patient outcome as compared to gemcitabine alone . In conclusion, the MD Anderson data clearly suggest favorable outcomes for primarily operable patients using a neoadjuvant gemcitabine-based hypofractionated (30 Gy in 10 fractions ± IORT) radiochemotherapy regimen.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here