Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Individuals at risk for lower extremity peripheral artery disease (PAD) should undergo a vascular review of symptoms to assess walking impairment, claudication, ischemic rest pain, and the presence of nonhealing wounds.
Despite recent advances in noninvasive evaluation of lower extremity PAD, contrast angiography remains the gold standard for definitive evaluation. Contrast angiography provides detailed information about arterial anatomy and is recommended for evaluation of patients with lower extremity PAD when revascularization is contemplated.
Occlusive disease of the iliac arteries appears to occur in relatively young patients and may have a greater impact on productivity and lifestyle compared with infrainguinal lower extremity disease.
The excellent intermediate- to long-term patency rates following percutaneous intervention of the lower extremity arteries has led to its emergence as an attractive alternative to surgery in patients with suitable lesions. Endovascular intervention is recommended as the preferred revascularization technique for TransAtlantic Inter-Society Consensus (TASC) type A and B iliac and femoropopliteal artery (FPA) lesions.
Stenting is effective as primary therapy for common iliac and external iliac artery stenosis and occlusions.
Technical and clinical success rates of endovascular interventions for iliac artery stenosis exceed 90% with a fairly comparable intermediate- and long-term patency to that of surgical revascularization, making them the initial therapy of choice for most iliac stenoses.
Although controversial, surgery remains the preferred strategy for patients with common femoral and proximal profunda femoris obstructive PAD.
Lower durability of percutaneous intervention may be offset by the less invasive nature of endovascular interventions and resultant decreased morbidity and mortality compared with vascular surgery.
Interventions can be repeatedly done if they fail, but repeat surgery is technically more challenging and may be limited by availability of conduit.
In patients with anatomically appropriate lesions, most practitioners use endovascular interventions preferentially as the initial therapy of choice.
Peripheral arterial disease of the lower extremity is a common health problem. Epidemiologic studies have mostly used intermittent claudication (IC) as a symptomatic marker of the disease and an abnormal ankle-brachial index (ABI) to define the burden of asymptomatic peripheral artery disease (PAD). The prevalence of the disease is dependent on the age of the population studied and the underlying atherosclerotic risk profile of the cohort. It is estimated that the overall disease prevalence is in the range of 3% to 10%, increasing to about 15% to 20% in persons older than 70 years. More than half of all patients with lower extremity PAD may be asymptomatic.
History that includes standardized questionnaires, such as the Rose Claudication Questionnaire used in the Framingham Heart Study, and physical examination may grossly underestimate the true burden of lower extremity PAD. Criqui and colleagues evaluated the prevalence of lower extremity PAD in a population of 613 men and women in Southern California using four different modalities: the Rose Questionnaire, pulse examination, ABI, and pulse wave velocity. The detection rate of PAD with ABI and pulse-wave velocity was two to seven times higher than the detection rate of the Rose Questionnaire. Of interest, clinical examination of the pulse overestimated the prevalence of PAD by twofold.
To optimally manage patients with lower extremity PAD, whether symptomatic or asymptomatic, it is important to understand the global vascular disease burden, the natural history of the disease process, its impact on the patient’s lifestyle, and the risk factors for an individual patient. Such knowledge is key to reducing the mortality and morbidity of the individual patient.
The abdominal aorta bifurcates at the level of the fourth lumbar vertebra into two branches, the right and the left common iliac arteries, which further divide into the external iliac artery , which normally follows the same axis of the common iliac artery, and the internal iliac arteries , which take a posteromedial track in relation to the common iliac artery. In addition to its terminal branches, the common iliac artery gives branches to the surrounding tissues, peritoneum, psoas muscle, ureter, and nerves. Occasionally, the common iliac artery provides accessory renal arteries to a normal or ectopic kidney. Fig. 39.1 depicts the arterial circulation of the lower extremity.
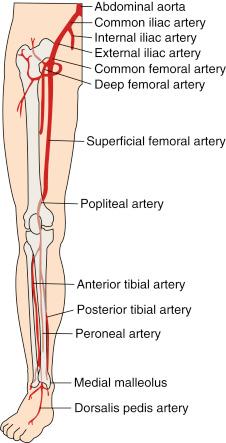
The external iliac arteries are larger than the internal iliac arteries. They descend along the medial border of the psoas major muscle and enter the thigh posterior to the inguinal ligament to become the common femoral arteries. The inferior epigastric artery arises medially from the distal external iliac artery and ascends behind the rectus abdominis muscle. This vessel is a useful landmark in predicting higher bleeding risk in arterial punctures proximal to its origin. The external iliac artery gives off other branches—namely, the deep circumflex, cremasteric, and several muscular and cutaneous branches—before it continues as the common femoral artery (CFA). Together the common iliac and the external iliac arteries contribute to the inflow of the lower extremity.
The CFA starts as a continuation of the external iliac artery, giving multiple branches to surrounding tissues, such as the pudendal arteries and the superficial circumflex artery; then it becomes the superficial femoral artery (SFA) after giving rise to the deep femoral artery (DFA) roughly 3.5 cm distal to the inguinal ligament. The DFA arises laterally and posteriorly from the CFA, whereas the SFA continues its pathway to end as the popliteal artery when it passes through the abductor canal. The DFA gives off perforating branches, usually three—the lateral and medial circumflex arteries and muscular branches—and the end of the DFA is the fourth perforating branch. The popliteal artery continues through the abductor’s canal, and after giving muscular, cutaneous, genicular, and sural branches, it terminates into the anterior tibial artery and the tibioperoneal trunk. The CFA, SFA, DFA, and the popliteal artery comprise the outflow of the lower extremity.
The anterior tibial artery runs between the two heads of the tibialis posterior muscle and then through the upper part of the interosseous membrane to the front of the leg medial to the head of the fibula. It descends down to the ankle and then continues to the dorsum of the foot, where it becomes the dorsalis pedis artery. The posterior tibial artery arises from the popliteal artery distal to the origin of the anterior tibial artery as the tibioperoneal trunk. After giving rise to the peroneal artery, the tibioperoneal trunk continues as the posterior tibial artery behind the leg and passes behind the medial malleolus to end by giving rise to the arteries of the foot, namely the calcaneal, which anastomoses with the calcaneal and malleolar branches of the peroneal and medial and lateral planter arteries. The anterior tibial artery, posterior tibial artery, and the peroneal artery are considered the runoff vessels.
Patients with lower extremity PAD may be asymptomatic (disease is detected during physical examination or screening tests), or they may present with IC, rest pain, nonhealing ulcers, or intractable foot infections. In light of the limitations of clinical assessment in defining patients with asymptomatic PAD, additional assessment of patients with risk factors for atherosclerosis may be required in identifying patients with asymptomatic advanced PAD. Symptomatic patients can be classified based on the severity of ischemia based on either the Fontaine or Rutherford categories ( Table 39.1 ).
| Fontaine | Rutherford | |||
|---|---|---|---|---|
| Stage | Clinical | Grade | Category | Clinical |
| I | Asymptomatic | 0 | 0 | Asymptomatic |
| IIa | Mild claudication | I | 1 | Mild claudication |
| IIb | Moderate to severe claudication | I | 2 | Moderate claudication |
| I | 3 | Severe claudication | ||
| III | Ischemic rest pain | II | 4 | Ischemic rest pain |
| III | 5 | Minor tissue loss | ||
| IV | Ulceration or gangrene | IV | 6 | Ulceration or gangrene |
The ABI is a ratio of the blood pressure in the dorsalis pedis or the posterior tibial artery, whichever is higher, to the blood pressure in the brachial arteries. ABI measurement is one of the most cost-effective methods for assessing PAD of the lower extremity, and it typically is done with a handheld Doppler ultrasound device. This is a noninvasive, fairly reproducible, and inexpensive test. The resting ABI should be used to establish the lower extremity PAD diagnosis in patients with suspected lower extremity PAD, defined as individuals with one or more of the following: exertional leg symptoms, nonhealing wounds, age 65 and older, or 50 years and older with a history of smoking or diabetes. The ABI should also be measured in both legs in all new patients with PAD of any severity to confirm the diagnosis of lower extremity PAD and establish a baseline. ABI results should be uniformly reported: noncompressible values are defined as greater than 1.40, normal values are 1.00 to 1.40, borderline is 0.91 to 0.99, and abnormal is 0.90 or less. The most widely accepted definition of lower extremity PAD is a resting ABI of less than 0.9, which is usually associated with 50% or greater angiographic arterial stenosis with a reported sensitivity of 95% and close to 100% specificity. A resting ABI of 0.4 to 0.9 is suggestive of mild to moderate PAD, and a resting ABI below 0.4 is suggestive of severe PAD ( Table 39.2 ). Resting ABI measurement can be artifactually high in the setting of tibial artery calcification usually seen in diabetics. The toe-brachial index (TBI) should be used to establish a diagnosis in patients in whom lower extremity PAD is clinically suspected but in whom the ABI test is not reliable because of noncompressible vessels (usually patients with long-standing diabetes or advanced age). A TBI below 0.7 is considered abnormal.
| Supine Resting ABI | Postexercise ABI | |
|---|---|---|
| Normal | >1.0 | >1.0 |
| Mild | 0.8–0.9 | >0.4 |
| Moderate | 0.4–0.8 | >0.2 |
| Severe | <0.4 | <0.2 |
a Postexercise ABI is measured following treadmill exercise at 1–2 mph, 10% to 12% grade, for 5 min or is symptom limited.
The use of exercise ABI may be helpful in equivocal cases. For this, the patient walks on the treadmill at a constant speed of 1 to 2 miles per hour and at a 10% to 12% incline for 5 minutes, or the exercise can be done with active pedal plantar flexion. A decrease of at least 15 mm Hg in the ankle systolic pressure following the exercise challenge is considered an abnormal test. Patients with no significant PAD are expected to have an increase or no change in their ankle systolic pressure. Exercise treadmill tests may also be performed in individuals with claudication who are to undergo exercise training (lower extremity PAD rehabilitation), so as to determine functional capacity, assess nonvascular exercise limitations, and demonstrate the safety of exercise.
Plethysmography is used to detect volumetric changes in the lower extremity blood flow and is performed with pressure cuffs inflated to 60 to 65 mm Hg at various segments of the lower extremity. A normal tracing will have a rapid systolic upstroke and downstroke with a prominent dicrotic notch. This pattern changes as PAD develops and progresses, with a noted attenuation and widening of the arterial waveform. Ultimately, the waveform becomes flat (nonpulsatile) in patients with advanced PAD ( Fig. 39.2 ). Pulse volume recordings are reasonable to establish the initial lower extremity PAD diagnosis, assess localization and severity, and follow the status of lower extremity revascularization procedures.
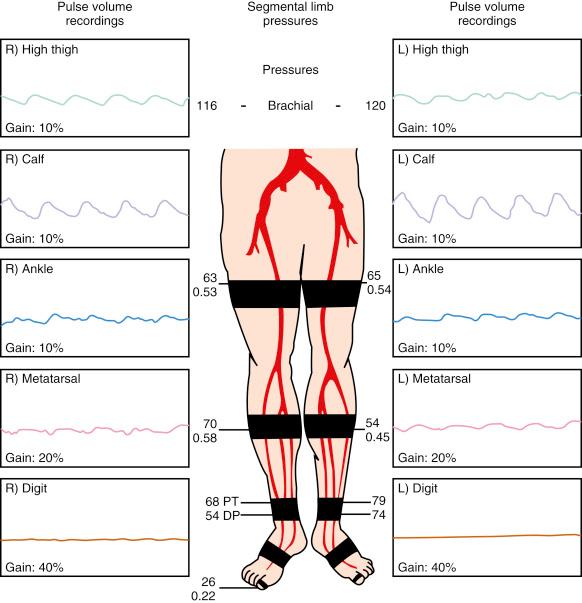
For this test, a series of blood pressure cuffs are placed at the level of the thigh (one or two cuffs), calf, ankle, foot, and the big toe. These cuffs are then inflated sequentially to about 20 mm Hg above the systolic pressure in that segment. The cuff pressure is then released slowly, and a continuous-wave Doppler probe is used to obtain the pressure at each segment. A decrease between two consecutive levels of 30 mm Hg or more indicates the presence of a stenosis in the segment proximal to the blood pressure cuff. Also, the presence of a 20- to 30-mm Hg difference in the pressure at one limb, when compared with the contralateral limb at the same level, is suggestive of significant PAD proximal to the cuff in that limb.
Duplex ultrasound uses a 5- to 7.5-MHz transducer to assess and characterize suprainguinal and infrainguinal PAD with a high sensitivity and specificity (over 90%). Doppler velocities are obtained (60-degree Doppler angle) to complement two-dimensional ultrasonography. Traditionally, arteries are classified into five categories based on the degree of stenosis: category 1 is considered normal, 2 indicates stenosis of 1% to 19%, 3 is stenosis of 20% to 49%, 4 indicates 50% to 99% stenosis, and 5 signifies total occlusion. Duplex ultrasonography may be useful to operators in planning access to a lesion that is amenable to endovascular therapy. It is also very helpful in identifying iatrogenic traumatic lesions and pseudoaneurysms. Direct ultrasound-guided compression or thrombin injection to repair femoral artery pseudoaneurysms is widely used in treating such lesions without the need for surgical procedures. One important limitation of Duplex ultrasonography is that it may overestimate residual stenosis following interventions, which limits its usefulness as a follow-up tool in this setting. Duplex ultrasound is recommended for routine surveillance after femoral-popliteal or femoral-tibial-pedal bypass with a venous conduit. Minimum surveillance intervals are approximately 3, 6, and 12 months and then yearly after graft placement.
The use of spiral computed tomography angiography (CTA) in assessing lower extremity PAD has 93% sensitivity and 96% specificity in detecting greater than 50% stenosis with high accuracy when compared with digital subtraction angiography (DSA). A systematic review and meta-analysis suggested that CTA is highly accurate for assessment of PAD in detecting stenosis of the aortoiliac and femoral arteries. However, diagnostic accuracy of CTA in below-knee vessels is poor and contrast angiography should be considered where below-knee disease needs to be accurately identified. CTA correctly identified hemodynamically significant lesions and also accurately distinguished between greater than 50% stenoses and occlusions. Ninety-four percent of occlusions and 87% of nonoccluded segments with more than 50% stenosis detected by contrast angiography were correctly identified by CTA. The accuracy of CTA may potentially be even higher than reported in this study because all studies used contrast angiography as the reference standard but did not report whether biplanar views were used routinely. Because CTA reconstructions allow three-dimensional (3-D) assessment, significant disease may be detected by CTA but may not be recognized by angiography, which may lead to the underestimation of specificity. CTA has advantages over magnetic resonance angiography (MRA) in patients with pacemakers and defibrillators and in those with metal clips, stents, or prosthesis (no significant artifact is seen in CTA as opposed to MRA), and it is significantly faster to perform than MRA. However, CTA requires the use of iodinated contrast, and it entails exposure to ionizing radiation. Dose-saving algorithms are very effective in reducing radiation exposure and should be used whenever possible.
The use of gadolinium-enhanced magnetic resonance angiography (GEMRA; Fig. 39.3 ) in the assessment of lower extremity PAD has been compared with standard catheter angiography, with a reported sensitivity and specificity for detecting stenosis greater than 50% of about 90% and 100%, respectively. Most contemporary studies report an agreement of 91% to 97% between MRA and catheter angiography. GEMRA is superior to duplex ultrasound in detecting greater than 50% stenotic lesions (sensitivity of 98% vs. 88% and specificity of 96% vs. 95%).
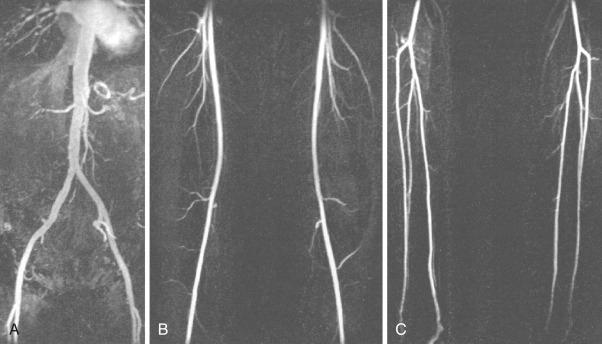
Limitations of this technology include the tendency to overestimate the severity of stenosis and the fact that metal clips may give the impression of total occlusion, and metal stents can obscure vascular flow as well. In addition to these limitations, certain subsets of patients may not be able to be studied with MRA, such as those with pacemakers and defibrillators and those with certain types of cerebral aneurysm clips. The principle role of MRA is in the initial evaluation for PAD, especially in patients with inflow disease, using “bolus chase” 3-D imaging, in which a single bolus of contrast is followed to the foot.
Despite recent advances in noninvasive evaluation of lower extremity PAD, contrast angiography remains the gold standard. Traditionally, a pelvic/abdominal aortogram in the anteroposterior projection is done using a straight pigtail catheter (5 or 6 Fr) placed at the level of the L1-L2 vertebrae. Approximately 10 to 15 mL of isoosmolar contrast is injected at a rate of 15 mL/s with DSA technology. This allows an excellent view of the distal aorta, the origin of the common iliac arteries, and the external iliac and common femoral arteries. Angulated views (left anterior oblique [LAO] of 30 degrees) can then be used to visualize the iliac and femoral bifurcations without overlap. Next, the pigtail catheter is placed above the aortic bifurcation (L3 to L4) and DSA with bolus chase, 8 mL/s for 10 seconds, is used to assess the outflow and distal runoff. Selective injections and sheath injections can then be used to further define the territory of interest as needed. The use of “road map” technology can be used subsequently to help operators in their intervention and placement of balloons and stents. Contrast angiography remains the most readily used and widely available imaging technique in patients with PAD of the lower extremity when revascularization is contemplated. The use of noninvasive technologies, such as MRA or CTA, may be used prior to contrast angiography to help identify the potential culprit lesion and plan the best approach (access point, catheter selection, etc.) to study such a lesion invasively. Contrast angiography may be associated with vascular access complications (bleeding, infections, pseudoaneurysms, and vascular disruption), atheroembolism, contrast nephropathy, and contrast-induced anaphylactoid reactions. These complications, although rare, should be considered in the decision-making process with regard to the assessment of PAD.
Overall, contrast angiography provides detailed information about arterial anatomy and is recommended for evaluation of patients with lower extremity PAD when revascularization is contemplated. When conducting a diagnostic lower extremity arteriogram in which the significance of an obstructive lesion is ambiguous, transstenotic pressure gradients and supplementary angulated views should be considered.
Percutaneous or surgical interventions are indicated for severe lifestyle-limiting symptoms, to reduce tissue breakdown in the context of critical limb ischemia (CLI), or for salvage in the context of acute limb ischemia (ALI). In general, endovascular procedures are indicated for individuals with a vocational or lifestyle-limiting disability due to IC when clinical features suggest a reasonable likelihood of symptomatic improvement with endovascular intervention and (1) the response to exercise or pharmacologic therapy has been inadequate or (2) the risk-benefit ratio is a very favorable (e.g., focal aortoiliac occlusive disease). The Society of Vascular Surgery (SVS) recommends objective performance goals to assess efficacy of interventions in a patient-centric manner to include major adverse cardiovascular events (MACE), 30-day major adverse limb events (MALE), 30-day amputation rate, amputation-free survival, and freedom from MALE, and can objectively assess efficacy of interventions on hard outcomes.
The indications for iliac artery (or aortoiliac) percutaneous intervention include (1) symptom relief in patients with IC who failed medical therapy; (2) management of CLI (rest pain, ulceration, or gangrene) prior to a planned distal lower extremity bypass surgery to restore or to preserve the inflow to the lower extremity; (3) in preparation for other invasive procedures, such as the placement of an intraaortic balloon pump (IABP); and (4) for treatment of flow-limiting dissection following invasive catheterization-based procedures.
Occlusive disease confined to the iliac arteries appears to occur in relatively young patients and may therefore have a greater impact on productivity and lifestyle. For instance, the mean age of the cohort in the Dutch Iliac Stent Trial was approximately 59 years. These patients, over 90% of whom were smokers, were otherwise healthy compared with those with infrainguinal disease or more diffuse PAD. In general, any type of revascularization for this subset of patients can offer satisfactory long-term results. Historically, aortobifemoral bypass surgery has been the gold standard for PAD that involves the iliac arteries, because this procedure is associated with excellent long-term patency rates (85% to 90% at 5 years, 75% to 80% at 10 years, and 60% at 20 years); however, it may be associated with an intraoperative mortality of roughly 1% to 3% and a major complication rate of 5% to 10%. This, combined with the excellent intermediate- to long-term patency rates following percutaneous intervention, has led to the emergence of percutaneous revascularization as an attractive alternative to surgery in patients with suitable lesions for such intervention. In a Swedish randomized controlled trial (RCT) in which 37% of randomized patients had iliac artery stenosis, equivalence in outcomes was evident between percutaneous transluminal angioplasty (PTA) and surgery. In the iliac disease subgroup, the patency rate at 1 year was 90% in the PTA arm versus 94% in the surgical arm. Table 39.3 summarizes the recommendation of the Trans-Atlantic Inter-Society Consensus (TASC) working group for the revascularization strategy of iliac lesions. The TASC group classifies peripheral arterial lesions into Type A, B, C, or D according to anatomic distribution, number and nature of lesions (stenosis, occlusion), and according to the overall success rates of treating the lesion percutaneously or surgically.
| Iliac Disease | Femoral Lesions | |
|---|---|---|
| TASC A | Single, <3 cm of CIA or EIA (unilateral/bilateral) | Single, ≤3 cm in length, does not involve the SFA or popliteal artery |
| TASC B | 1. Single, 3–10 cm, not extending into the CFA 2. Two stenoses <5 cm long in the CIA and/or EIA, does not extend to the CFA 3. Unilateral CIA occlusion |
1. Single stenoses or occlusion 3–5 cm long, does not involve the distal popliteal artery 2. Heavily calcified, ≤3 cm, or multiple stenoses or occlusions each <3 cm 3. Single or multiple lesions in the absence of continuous tibial runoff |
| TASC C | 1. Bilateral 5- to 10-cm stenosis of the CIA and/or EIA, does not extend to CFA 2. Unilateral EIA occlusion or stenosis, does not extend into the CFA 3. Bilateral CIA occlusion |
1. Single stenosis/occlusion >5 cm 2. Multiple stenoses or occlusions 3–5 cm, with or without heavy calcification |
| TASC D |
|
Complete CFA or SFA occlusions |
In patients with a unilateral stenotic iliac lesion that does not involve the CFA, ipsilateral access through the CFA with retrograde PTA is usually preferred because it provides direct access to the diseased segment and provides a coaxial alignment of the equipment ( Fig. 39.4 ). Contralateral access with the use of a crossover sheath is reserved for patients with disease that involves the ipsilateral CFA ( Fig. 39.5 ) when the surgeon plans to intervene on more distal lesions in the same limb or when jeopardizing flow to the affected limb by placement of the sheath in the ipsilateral CFA is a concern. Various crossover sheaths are available; the ArrowFlex sheath (Arrow International, Reading, PA) is more flexible and can be helpful in crossing over acute aortic bifurcation angles, but it provides less support than the Flexor Ansel, Raabe, and Balkin sheaths (Cook Medical, Bloomington, IN); these provide more support but are less compliant. If the iliac artery is occluded, both approaches may be needed. For aortic bifurcation lesions, a bilateral retrograde approach is used; the placement of kissing balloons—and, subsequently, kissing stents—is the optimal approach in this setting.
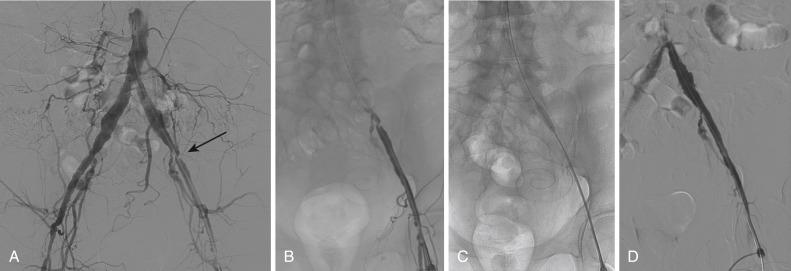
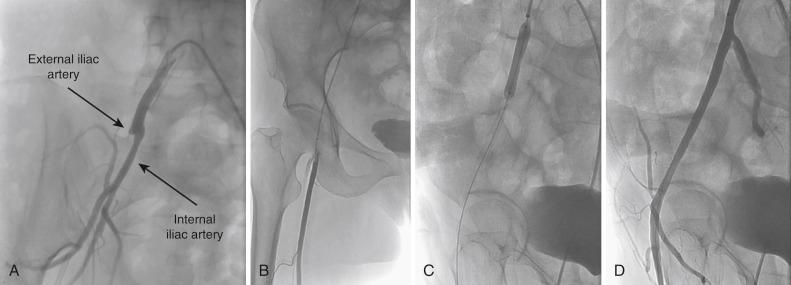
In lesions distal to the aortic bifurcation (in the body of the common iliac artery or the external iliac artery), PTA is attempted, and if satisfactory results are achieved (<5 mm Hg residual gradient and <30% residual stenosis with no flow-limiting dissection), stenting may not be indicated. However, ostial lesions of the common iliac arteries (i.e., aortoiliac bifurcation lesions) are preferably stented with kissing stents. In general, 0.035-inch guidewires are used in PTA and in stenting of the iliac arteries, but 0.018- or 0.014-inch guidewires may be used as well. In nonocclusive lesions, a regular nonhydrophilic guidewire can be used; however, if crossing such lesions is difficult, the use of a hydrophilic wire is indicated.
Both balloon- and self-expandable stents can be used in aortoiliac disease. The balloon-expandable stent is advantageous in the context of an aortic bifurcation lesion, in which kissing stents are usually placed. It is also superior to self-expandable stents when precision in stent placement is needed. The self-expanding stent provides the flexibility in flexion points, which reduces the risk of stent deformity and fracture, and is ideal in the setting of common iliac lesions that do not involve the ostium and those in the external iliac artery. Self-expanding stents were generally favored for the external iliac artery because they reduce the perforation risk.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here