Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
The WHO classification of gestational trophoblastic disease (GTD) and the TNM and FIGO classifications of gestational trophoblastic tumors are presented in Tables 10.1–10.4 .
| With villi |
|
| Without villi |
|
|
|
a FIGO stages are subdivided into A (no risk factors), B (one risk factor), C (two risk factors). The risk factors are: (1) Serum Hcg >100,000 iu/ML, and (2) duration of disease >6months from termination of antecedent pregnancy.
| Prognostic factors | Score | |||
|---|---|---|---|---|
| 0 | 1 | 2 | 4 | |
| Age (year) | <40 | >39 | - | - |
| Antecedent pregnancy | Mole | Abortion | Term | - |
| Interval (months)* | <4 | >3, <7 >6, <13 | >12 | |
| Pretreatment serum hCG (mIU/ml) | <10 3 | 10 3 to <10 4 | 10 4 to <10 5 | >10 5 |
| Largest tumor, including uterine (cm) | - | 3 to <5 | >4 | |
| Site of metastases | Lung | Spleen GI tract | Brain | |
| Kidney | Liver | |||
| Number of metastases | – | 1–4 | 5–8 | >8 |
| Prior failed chemotherapy | – | - | Single drug | Two drugs |
Hydatidiform moles (HMs), which may be partial (PHM) or complete (CHM), almost always occur in the reproductive age group, although rare cases occur in postmenopausal women.
Risk factors include personal or family history of GTD, two or more previous spontaneous abortions, infertility, smoking, and increased maternal age.
Interobserver and intraobserver variability exists in the histologic diagnosis of hydropic abortus (HA), PHM, and CHM.
In difficult cases, examination of remaining tissue, review of any recent curettage specimens, and staining for p57 (a surrogate marker for the maternal genome) aid diagnosis.
When the diagnosis is not resolvable by p57 staining, molecular genotyping by fluorescence in situ hybridization (FISH) can be diagnostic, showing biparental diploidy in nonmolar specimens, diandric triploidy in PHMs, and androgenetic diploidy in CHMs. have found that chromogenic in situ hybridization can be diagnostic when FISH is unavailable.
found a higher incidence of HMs (3.3/1000 deliveries) than in previous studies that was attributable to improved detection of PMs by molecular genotyping. In their study PMs were almost twice as common as CMs.
have developed an algorithmic approach to diagnosing hydatidiform moles ( Fig. 10.1 ).
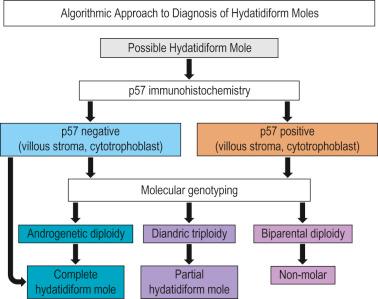
CHMs have been considered the most common molar pregnancy, but as already noted, have found them to be only half as common as PHMs. They occur in 1 : 1500 deliveries in the USA, but are much more common in other parts of the world, particularly the Far East. found an increased risk for them in adolescents and those of advanced maternal age.
The classic presentation of CHMs was second trimester vaginal bleeding, a large-for-dates uterus, and in some cases, passage of molar vesicles. High levels of hCG (>100,000 mlU/ml) and ultrasound findings also suggest the diagnosis. Uncommon manifestations have included pre-eclampsia in early gestation, hyperemesis gravidarum, hyperthyroidism, hyperreactio luteinalis ( Chapter 12 ), and metastases (lung, vagina).
With routine first-trimester ultrasound examinations, most CHMs are now evacuated late in the first trimester, the sonographic diagnosis being suspected mole or, more commonly, a missed abortion.
Most CHMs are diploid and diandric with two sets of paternal chromosomes (androgenetic diploidy) or tetraploid. Rare examples are characterized by biparental diploidy; these may be familial, recurrent, and related to maternal mutations in NLRP7 and C6orf221 ( ).
Grossly evident hydropic villi (villous vesicles) are characteristic, but may not be appreciable in an early CHM or if there has been curettage-related fragmentation and collapse of the villi.
The cardinal histologic features of well-developed CHMs are diffuse villous hydrops of variable degree and trophoblastic hyperplasia.
The hydropic villi are irregular in shape and exhibit a spectrum of sizes. Particularly in the first trimester, the villi exhibit a distinctive budding architecture with secondary club-like or toe-like extensions. Cisterns (cavities) are present within the larger villi and increase in number with gestational age, but may be absent in early CHMs.
The hyperplastic villous trophoblast tends to be circumferential (ensheathing the entire villus) and usually involves many villi in mature CHMs. The degree and extent of trophoblastic hyperplasia, however, can be quite variable, and degenerative changes can result in focal loss of the villous trophoblast.
Cytotrophoblastic cells (CT) may exhibit marked nuclear pleomorphism. The syncytiotrophoblast cells (ST) usually contain cytoplasmic vacuoles and form lacy protrusions from the villous surface.
Intravillous trophoblastic inclusions can occur, but they are not as common as in PHMs.
Intermediate trophoblast in the intervillous space and the implantation site is also hyperplastic, often with marked nuclear atypia.
The villi in well-developed CHMs usually appear avascular, but CD34 staining reveals vessels in numbers similar to those of normal villi of 8–12 weeks gestational age. As fetal tissues (intravascular nucleated erythrocytes, amnion) occur in <2% of CHMs, their presence does not exclude the diagnosis but should suggest a PHM or possibly twin gestation with one gestation being a CHM.
Karyorrhectic debris within the villous stroma is characteristic of CHMs and is more common in early CHMs than in normal placentas of similar gestational age; similar debris within vessels is not diagnostically helpful as it also occurs in PHMs and nonmolar abortuses.
Early CHMs (<12 weeks gestational age).
These can be difficult to diagnose given the presence of only mild focal villous hydrops, rare to absent cisterns, and only focal villous hyperplasia, features that can incorrectly suggest a PHM or even HA. Villous vessels and nucleated erythrocytes may be more commonly seen than in typical CHMs.
Diagnostic clues include a distinctive bluish, myxoid hypercellular villous stroma with small stellate stromal cells, a labyrinthine network of small stromal vessels, prominent club-like extensions of the villi (as noted above), circumferential trophoblastic hyperplasia in at least some villi, villous karyorrhexis, and striking hyperplasia and atypia of intermediate trophoblast.
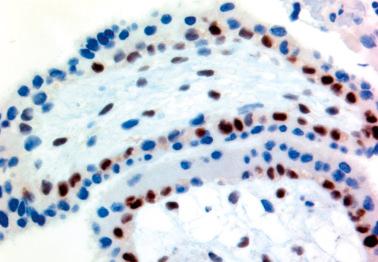
In contrast to PHMs and HA, the cytotrophoblast and villous stromal cells in most CHMs (including early CHMs) have absent or limited (<10% of cells) nuclear positivity for p57. The nuclei of the decidual cells and extravillous trophoblast, however, stain positively and serve as internal controls.
have found equivocal, aberrant, divergent, or discordant p57 expression in ~10% of suspected molar pregnancies requiring molecular genotyping for a definitive diagnosis.
Equivocal p57 expression was defined as >10% to <50% p57 staining of cytotrophoblast/villous stromal cells; all were confirmed as PHMs.
Aberrant p57 staining conflicted with morphologic and genotyping results. These were rare CHMs with diffuse p57 staining found to be due to retention of maternal chromosome 11.
Discordant staining was defined as an admixture of negative or positive results in individual villi: p57 expression was found in either villous stromal cells or cytotrophoblast but not both, presumably due to the presence or absence of maternal genetic material in the cells. These were found to be mosaic/chimeric conceptuses with admixed androgenetic diploid and biparental diploid cells within individual villi (see also Partial Mole, Differential Diagnosis ).
Divergent staining was defined as two populations of villi, one with negative staining and the other with positive staining, such as in a twin gestation composed of a CHM and a nonmolar abortus.
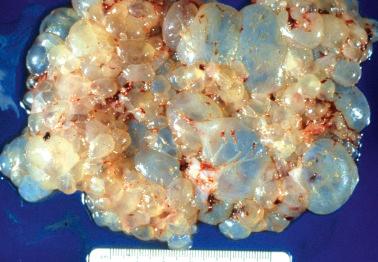
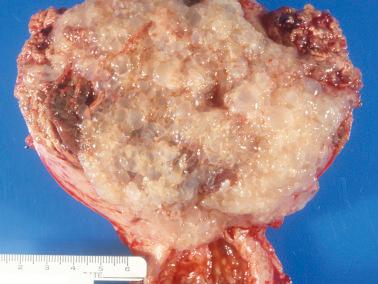
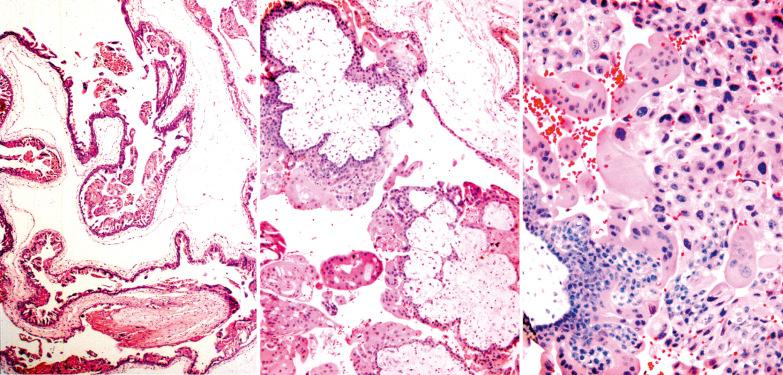
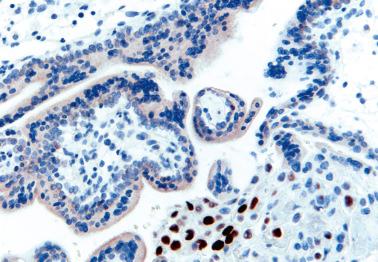
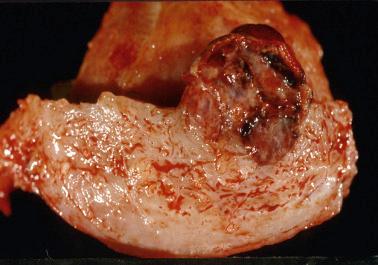
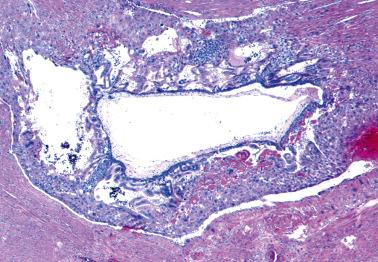
PHM and hydropic abortus. The differential features are indicated in Table 10.5 . Villous club-like projections and stromal karyorrhexis are the most useful histologic features that distinguish early CHM from PHM.
| Complete mole | Partial mole | Hydropic abortus | |
|---|---|---|---|
| Amount of placental tissue compared to normal for gestational age | Voluminous (5- to 10-fold increase); diffuse hydropic change b | Moderately increased (2-fold); focal hydropic change | Scanty tissue, far less than normal |
| Villous population | Spectrum of sizes; (large and small) | Two populations | Mostly of similar size and balloon-like |
| Villous shape | Round to bulbous | Very irregular | Round and smooth |
| Club-like projections from the villi | Common in early CHMs | Rare | Rare |
| Scalloped villous contours | Uncommon | Common | Rare |
| Trophoblastic pseudoinclusions | Common, irregular in shape | Common, round | Rare except for single cell inclusions |
| Villous trophoblastic hyperplasia | Moderate to marked; often circumferential; often mild in early CHM | Mild, circumferential in only occasional villi | Absent except for polar trophoblast |
| Cisterns | Common, especially in second trimester | Focal; may not be obvious in first trimester | Absent (or rare and <3 mm) |
| Karyorrhexis in villous stroma | Common in first trimester | Absent or inconspicuous | Absent or inconspicuous |
| Villous stroma | Mucoid, hydropic, no fibrosis | Some villi fibrotic; angiomatous vessels in 20% of second trimester PHMs | Mostly hydropic; some villi fibrotic |
| Fetal tissue | Usually none | Present in most cases and abnormal | Usually none |
| Amnion; fetal erythrocytes | Rare | Common | May be present |
| Extravillous trophoblast | Usually hyperplastic and atypical; exaggerated placental site | Usually normal | Usually normal |
| p57 nuclear staining of cytotrophoblast and villous stromal cells | Absent or sparse (<10% of cells) | Prominent | Prominent |
| Ki-67 staining of cytotrophoblast c | High (>70%) | High (>70%) | Low (<25%) |
| DNA content | Diploid d (diandric) | Triploid e (diandric, monogynic) | Diploid (biparental) |
| Chromosome number | 46 f | 69 g | 46 ± a few |
a As no single morphologic feature is specific, diagnosis usually rests on a constellation of features. Many of the findings listed for complete moles are less obvious in early moles (<12 weeks' gestation), as noted in the text. The distinction between early PHMs and hydropic abortions can be especially difficult and may be impossible in scanty material.
b Excluding first trimester complete moles.
c Based on the examination of a medium-sized villus (one that just spans a 200x field). Schammel DP, Bocklage T. p53, PCNA, and Ki-67 staining in hydropic molar and nonmolar placentas: An immunohistochemical study. Int J Gynecol Pathol 1996;15:158-66.
d A minority are haploid, triploid, tetraploid, or polyploid. Rare CHMs are biparental.
e Rare PHMs are haploid, diploid, tetraploid, aneuploid, or polyploid.
Very early nonmolar abortus with prominent trophoblastic proliferation. These cases can have circumferential trophoblastic proliferation, but in at least some villi the trophoblast is polar (confined to the villous tips) and the cytotrophoblastic cells are typically more uniform without the nuclear atypia of those in CHMs.
Some of the placental abnormalities listed in the differential of PHMs (see corresponding heading) are also in the differential of CHMs, such as mosaic/chimeric (androgenetic/biparental diploid) conceptuses that can exhibit trophoblastic hyperplasia and loss of p57 expression in both cytotrophoblast and villous stromal cells.
Egg donor pregnancy. described a case in which chorionic villi in an egg donor pregnancy showed unique alleles not present in maternal decidual tissue, suggesting a diandric paternal-only genome, but the p57 showed a normal staining pattern, excluding a CHM.
Persistent GTD occurs in 15–20% of CHMs. Evacuation of CHMs in the first trimester has not reduced the frequency of gestational trophoblastic neoplasia ( ).
Persistent mole.
Hallmarks are failure of hCG levels to normalize after initial evacuation and residual molar villi in a repeat curettage.
The risk increases with maternal age >40 years, previous molar pregnancy, pre-evacuation hCG levels of >100,000 mIU/ml, uterine size large for dates, and the presence of hyperreactio luteinalis, pre-eclampsia, hyperthyroidism, or trophoblastic emboli.
Invasive mole (chorioadenoma destruens).
Invasion, which occurs in 5–10% of CHMs, is suggested by high post-evacuation hCG levels and absence of molar villi on repeat curettage.
Although invasive PHMs have been reported in the older literature, we are unaware of any invasive PHMs proved to be triploid by molecular testing.
Diagnosis requires the presence of myometrial and/or vascular invasion by the molar villi (findings which usually require a hysterectomy) or the presence of metastases (lungs, vagina, vulva, broad ligament) containing molar villi. The differential includes very rare cases of embolization of villi following termination of a normal pregnancy.
found that some invasive CHMs were associated with synchronous atypical trophoblastic proliferations that were suspicious for or indistinguishable from choriocarcinoma. They also found a higher frequency of heterozygous/dispermic CHMs in invasive than in noninvasive moles.
Postmolar choriocarcinoma occurs in 2–3% of cases (see Choriocarcinoma ).
Recurrent GTD (a new episode of GTD after complete postchemotherapy remission) as evidenced by re-elevation of serum hCG after three consecutive weekly normal values.
100% cure rates are achieved in patients who have nonmetastatic disease or ‘good-prognosis’ metastatic disease (pulmonary or vaginal metastases, hCG levels <40,000 mIU/ml, short duration of disease).
Cure rates of >80% have been achieved even in patients with ‘bad-prognosis’ metastatic disease (brain, liver, kidney); hCG levels >40,000 mIU/ml; and those appearing after a term gestation.
The frequency of PHMs in the literature has varied from similar to that of CHMs to three times more common.
Almost all PHMs have diandric triploidy (one maternal and two paternal sets of chromosomes). Rarely they are tetraploid (3 sets of paternal chromosomes and one maternal set). About 80% of triploid gestations are diandric, most or all of which are PHMs. Cases reported as diploid PHMs are probably misclassified CHMs or HAs.
The usual presentation is bleeding in the late first or early second trimester as a missed or incomplete abortion, a uterus small or normal for dates, and an hCG level that is usually low to normal for gestational age, although rarely it is as high as in CHMs.
Gross examination reveals normal-appearing immature placental tissue admixed with vesicles that tend to be smaller and less numerous than those of a CHM. The volume of tissue is intermediate between that of a hydropic abortion and a CHM. A gestational sac and/or fetal parts may be present.
PHMs typically have all or almost all of the following features:
Two distinct populations of villi: enlarged hydropic villi and small to normal-sized villi that are often fibrous. Early PHMs may have more of a spectrum of villous sizes, only occasional large hydropic villi, or even an absence of hydropic villi.
Markedly irregular villi with prominent scalloping of their borders and trophoblastic invaginations (pseudoinclusions) within the villous stroma
Villi with central cavities (cisterns) that are ≥3–4 mm, but which are less common than in CHMs.
Hyperplastic villous trophoblast that is usually more mild and focal than in CHMs. At least some villi usually have circumferential trophoblastic hyperplasia, but rare early PHMs may lack trophoblastic hyperplasia. ST tends to predominate, with sprouts or knuckles of ST arranged haphazardly on the villous surface and as seemingly detached intervillous papillae. Prominent vacuolation of the ST can produce a lace-like appearance.
Absence of the hyperplastic and atypical IT usually present in CHMs.
Evidence of fetal development is common and includes stromal blood vessels with nucleated erythrocytes. Ectatic anastomosing ‘angiomatoid’ villous vessels occur in 20% of second trimester PHMs. Other fetal tissues may include chorionic plate, amnion, umbilical cord, and fetal parts that may be malformed.
In contrast to CHMs, most or all of the villous cytotrophoblast and the villous stromal cells show nuclear staining for p57. Rare PHMs with loss of maternal chromosome 11 may be p57−.
As noted earlier, molecular genotyping can confirm diandric triploidy in problematic cases.
performed molecular phenotyping in 251 cases with nondiagnostic villous abnormalities and found that 49% were triploid, including 85% of those in which PHM was initially favored and 32% of those in which nonmolar miscarriage was favored.
The authors concluded that while molecular genotyping is diagnostic, it is more expensive than a pragmatic approach of hCG surveillance.
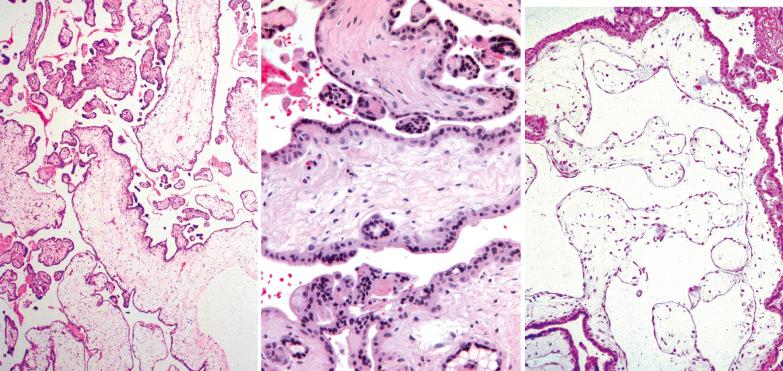
CHM and hydropic abortus. These are the most common considerations; their differential features are listed in Table 10.5 . found that the presence of both cisterns and villous size ≥2.5 mm had a 90% predictive value for PHM. Either of the latter two features or two populations of villi or round to oval pseudoinclusions should prompt DNA genotyping.
Triploid conceptus with two sets of maternal chromosomes. These digynic monoandric conceptuses, which account for up to a third of triploid gestations, lack the typical gross and microscopic features of PHM, but may exhibit abnormal villous morphology, with some villous hydrops irregular in size and shape and with pseudoinclusions and ST knuckles. The placentas tend to be small and the fetus is often growth-retarded.
Nontriploid genetically abnormal placentas with abnormal villous morphology. A definite diagnosis of these disorders will usually require molecular genotyping.
Trisomy gestations. In a group of PHMs and trisomy gestations, found that ≥2 of: cisterns, multifocal trophoblastic proliferation, and large trophoblastic inclusions correctly classified PHMs in 93% of cases. There was no difference in villous enlargement or scalloped villous contours between the two groups. found a ~20% frequency of aneuploidy in nonmolar gestations with abnormal villous morphology of which 11 of 15 were trisomy and 4 were monosomy X. Such findings may assist subsequent counseling or those undergoing in vitro fertilization.
Mosaic/chimeric conceptuses, which are androgenetic/biparental diploid, can have variable morphology suggesting a PHM or CHM, including some with features of placental mesenchymal dysplasia (see next bullet). There may be an admixture of hydropically enlarged villi without trophoblastic hyperplasia with p57+ cytotrophoblast and p57− villous stromal cells and villi with trophoblastic hyperplasia that are uniformly p57− in both cell types . Some of these shown to have a component of CHM by FISH have had persistent GTD ( )
Placental mesenchymal dysplasia (pseudopartial moles) ( ). These placentas may show villous hydrops and aneurysmal villous vessels with chorioangiomatoid change, but usually no pseudoinclusions or trophoblastic hyperplasia. The fetus may be normal, show intrauterine growth retardation, or features of the Beckwith–Wiedemann syndrome.
found that molecular analysis of nonmolar abortuses (that included some spontaneous abortions) with atypical villus morphology yielded a 59% detection rate of abnormal chromosomal alterations including complex karyotypes and aneuploidy.
CHM associated with a twin pregnancy. These cases have an admixture of normal villi (with positive p57 staining) and molar villi with the features of a CHM (with negative p57 staining). All the villi are diploid (some diandric, others biparental). Genotyping may be inconclusive unless microdissection with analysis of the abnormal villi is performed.
Persistence occurs in <1% of cases. found that none of 196 PHMs (all proven triploid) had persistent disease. Studies indicating higher figures of persistence likely included misdiagnosed CHMs.
Rare reported PHM-associated complications include invasive mole, metastatic mole, and the subsequent development of choriocarcinoma or placental site trophoblastic tumor (see corresponding headings).
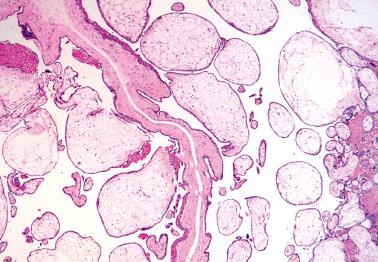
The evacuated tissue in a hydropic abortus (HA) is typically scanty, with complete examination of the chorionic tissue (which should be performed in any suspected case) requiring only one or two blocks.
The villi show variable degrees of hydropic change; smaller villi may be fibrotic. Villi only rarely reach the size of those in PHMs or CHMs and tend to have a more uniform, balloon-like shape than those of PHMs and CHMs. Cisterns are rare to absent.
Most villi are hypovascular to avascular. The vessels may be empty and collapsed or contain fetal erythrocytes.
The trophoblast is usually diffusely attenuated, although some villi may show normal polar or apical cytotrophoblast and/or intermediate trophoblast. Villous trophoblastic hyperplasia may occur in abortions with an abnormal karyotype, especially triploid digynic abortuses and trisomy.
The differential diagnosis with a PHM ( Table 10.5 ) is associated with considerable interobserver variation. In cases in which a definite histologic distinction between the two lesions cannot be made, molecular genotyping can be diagnostic; most HAs, in contrast to PHMs, show biparental diploidy or rarely, digynic triploidy.
Choriocarcinomas in the USA complicate about 1 : 25,000 pregnancies; they are twice as common in blacks and other races compared to whites. The incidence is 20–40 times higher in Asia, Africa, and Latin America. A SEER study found that its annual incidence in the USA decreased by 50% between 1973 and 1999.
About 50% of choriocarcinomas are preceded by a CHM, 25% by an abortion, 22.5% by a normal pregnancy, 2.5% by an ectopic pregnancy; a prior PHM is rare. The interval between the choriocarcinoma and the antecedent pregnancy ranges from several months to many years. Rare choriocarcinomas are nongestational (see Chapter 8 ).
The most common presentation is vaginal bleeding and an elevated serum hCG. Patients may be asymptomatic if the tumor is confined to the myometrium. Some patients have manifestations related to hematogenous spread and have very high hCG levels.
Rare presentations and complications have included:
Choriocarcinoma in a term placenta, which is often macroscopically normal ( ). An undiagnosed tumor in this setting may account for some choriocarcinomas that follow a ‘normal’ term pregnancy.
Microscopic choriocarcinomas in curettage specimens performed at 8–11 weeks of gestation for vaginal bleeding; one such patient had pulmonary metastases. In this study, Fukunaga proposed that choriocarcinomas can arise from stem villi at any stage of pregnancy.
Presentation in an infant. In some such cases, no maternal tumor is found presumably due to tumor regression.
found an increased frequency of a second primary tumor post-choriocarcinoma, including acute myeloid leukemia, non-Hodgkin's lymphoma, and thyroid cancer.
In a genotyping study, found that 19 of 22 choriocarcinomas were gestational and 3 were nongestational.
14 of the 19 gestational tumors were purely androgenetic/homozygous XX: 6 uterine tumors with a concurrent or prior genetically related CHM; 4 uterine tumors without an accompanying villous component; 1 uterine cornual tumor separate from a genetically distinct second trimester intrauterine placenta; 1 ectopic ovarian tumor separate from a genetically distinct third trimester intrauterine placenta; and 2 ectopic tubal tumors.
Five gestational tumors were biparental: 3 intraplacental choriocarcinomas genetically related to the placenta and 2 uterine tumors without placental tissue after term deliveries. Three tumors were nongestational: all were XX with allelic imbalances.
This study concluded that (1) homozygous XX CHMs are associated with risk of GTD; (2) as intraplacental choriocarcinomas are biparental and genetically related to the placenta, a biparental choriocarcinoma in a postpartum uterine sample suggests an undetected intraplacental choriocarcinoma; (3) eutopic or ectopic androgenetic choriocarcinoma separate from a concurrent intrauterine placenta is not derived from the latter but is a form of dispermic twin gestation or originates from an antecedent molar pregnancy; (4) while tubal tumors are usually gestational, tumors in other sites (ovary, pelvis) can be nongestational and not necessarily metastatic from a regressed or occult intrauterine or intraplacental gestational tumor.
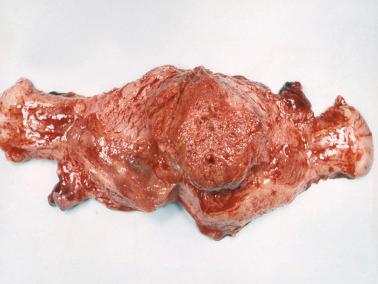
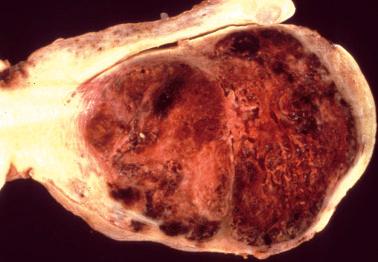
The tumors are typically red to brown and fleshy with extensive hemorrhage, necrosis and destructive myometrial invasion. The superficial part of the tumor may be polypoid. Rare tumors are primary in the cervix. Some patients with metastatic choriocarcinoma have no residual tumor in the uterus, likely due to regression of the primary tumor.
Choriocarcinomas arising in a term placenta are usually undetected on gross examination and found only microscopically. Grossly visible lesions are typically small (<3 cm) and resemble an infarct. Rare placental choriocarcinomas have been associated with a chorangioma (‘chorangiocarcinoma’).
The histologic sine qua non is an admixture of syncytiotrophoblast (ST) and mononucleated trophoblastic cells (cytotrophoblast [CT] and intermediate trophoblast [IT]), often in a plexiform pattern. The proportion of each component can be variable.
ST cells have densely eosinophilic cytoplasm (often with lacunae containing erythrocytes); large, vesicular, multiple nuclei with clumped chromatin; and reactivity for hCG and inhibin.
Mononuclear trophoblast (IT and CT) consists of discrete oval to polygonal cells with clear to eosinophilic cytoplasm, convoluted nuclei, macronucleoli, and numerous mitoses. Compared to CT, IT cells are larger with more abundant cytoplasm and have more pleomorphic nuclei. IT cells have a Mel-CAM+/ß-catenin− immunoprofile, whereas CT cells have the opposite profile.
Unusual findings include:
Chorionic villi. Villi can be seen in choriocarcinomas associated with a synchronous CHM, choriocarcinomas associated with the hyalinized villi of a remote pregnancy or associated with but separate from a term placenta (twin gestation), and choriocarcinomas arising in a term placenta (see above). In the last situation, the lesion may consist of a centrally necrotic zone composed of infarcted villi surrounded by viable choriocarcinoma in intervillous spaces.
A predominant component of CT or IT with scanty ST (‘atypical choriocarcinoma’). This pattern may be found in primary or metastatic tumor, and in some cases following chemotherapy.
A component of placental site trophoblastic tumor or epithelioid trophoblastic tumor (see Lesions of Intermediate Trophoblast ), a finding that should be specified in the report as their treatment may differ from that of pure choriocarcinoma.
There is usually invasion of the myometrium and myometrial vessels. Extensively necrotic and hemorrhagic tumors may require numerous sections to find viable tumor, which is often confined to a thin peripheral zone.
There is usually staining for hCG, HLA-G, Mel-CAM (CD146), SALL4, and inhibin.
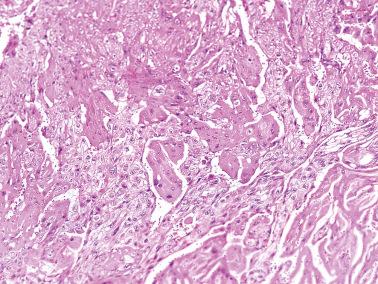
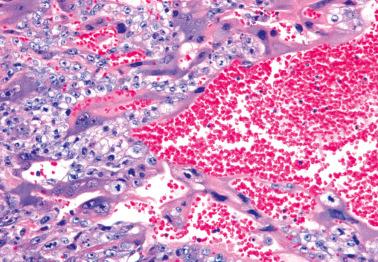
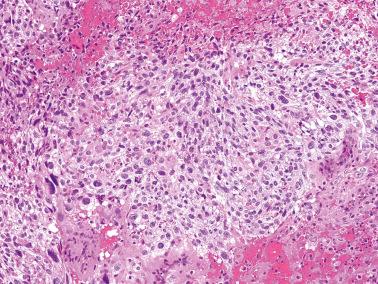
Trophoblastic tissue obtained in curettage specimens other than choriocarcinoma: villous trophoblast, simple trophoblast, trophoblast suspicious for choriocarcinoma, and intermediate trophoblast.
Villous trophoblast. The interpretation rests on evaluation of the associated villi (normal, HA, PHM, or CHM). Evacuation of a spontaneous abortion or a mole may leave residual avillous trophoblast that may be removed at a later procedure. Knowledge of the history and the presence of villi in the prior specimen will avoid a misdiagnosis of choriocarcinoma.
Simple trophoblast. Small amounts of undifferentiated trophoblast are present without villi. Such trophoblast includes the often worrisome-appearing and invasive previllous trophoblast of a very early gestation. The diagnosis of simple trophoblast requires sampling the entire specimen and that the trophoblast be scanty and exhibit only minimal differentiation towards CT or ST. Vascular invasion does not exclude the diagnosis.
Trophoblast suspicious for choriocarcinoma. There is abundant trophoblast with differentiation into ST and CT and/or IT, and invasion; no villi are present. Simple or suspicious trophoblast in a curettage specimen after a normal pregnancy is likely to be followed by malignant trophoblastic disease, in contrast to only 50% of patients with similar findings who have had a previous molar gestation.
Intermediate trophoblast of a normal or exaggerated implantation site, a placental site nodule/plaque, a placental site trophoblastic tumor, or an epithelioid trophoblastic tumor (see corresponding headings). Although proliferations of IT may contain occasional multinucleated cells somewhat resembling syncytiotrophoblast, the distinctive admixture of typical syncytiotrophoblast and cytotrophoblast of choriocarcinoma is absent.
Nongestational choriocarcinoma, including those of germ cell origin ( Chapter 15 ) metastatic to the uterus, and endometrial adenocarcinomas with trophoblastic differentiation ( Chapter 8 ).
Chorangiocarcinoma (see below).
Other malignant tumors, both epithelial (such as undifferentiated carcinoma) and mesenchymal (such as epithelioid leiomyosarcoma), that can potentially resemble typical or atypical choriocarcinoma. The diagnosis is facilitated by a history of a recent pregnancy or mole, thorough sampling to demonstrate ST, elevated serum hCG, and immunoreactivity for the markers noted above.
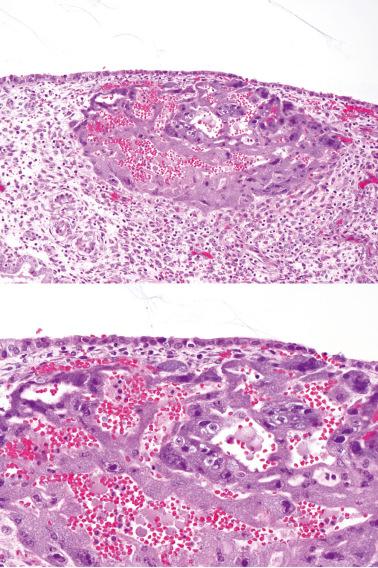
Overall postchemotherapy survival is about 90%. Death is usually due to hemorrhagic events within the metastases or pulmonary insufficiency (tumor burden or effects of treatment).
Choriocarcinomas arising in term placentas may have an uneventful course or be associated with metastatic disease in the mother or infant.
recognize three subpopulations of intermediate trophoblast (IT) with distinctive morphologic and immunohistochemical features: villous IT, implantation site IT, and chorionic-type IT.
The lesional cells of exaggerated placental site and its neoplastic counterpart, placental site trophoblastic tumor, are derived from implantation site IT, whereas placental site nodule and its neoplastic counterpart, epithelioid trophoblastic tumor, are derived from chorionic-type IT.
In the absence of chorionic villi or fetal tissues, identification of IT in a curettage specimen is useful in establishing the diagnosis of an intrauterine pregnancy and thus excluding an ectopic pregnancy. In such cases, it is important that IT cells are distinguished from decidual cells, which they can resemble.
The nuclei of IT cells, unlike those of decidual cells, tend to be variable in size and shape, are more hyperchromatic, and often have convoluted outlines or a cleft. Additionally the cytoplasm of IT is almost always more amphophilic.
Implantation IT, in contrast to decidual cells, is reactive for CK, hPL, inhibin-α, and Mel-CAM (CD146).
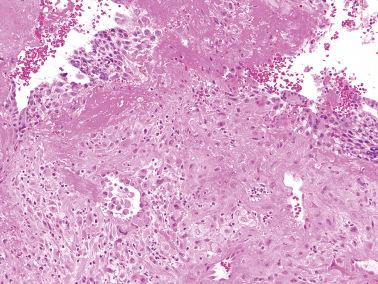
This term (EPS) refers to unusually striking involvement at the placental site by implantation site IT; synonymous terms have included ‘syncytial endometritis’ and ‘exaggerated placental site reaction’. In a review of their files, diagnosed EPS in 1.6% of first trimester spontaneous and elective abortions. EPS is more commonly present in molar gestations.
The histologic distinction between a ‘normal’ placental site and an ‘exaggerated’ placental site is arbitrary given the absence of specific criteria for EPS. EPS is probably a normal physiologic variant, the term being one of convenience when a placental site is unusually striking based on the pathologist's prior experience.
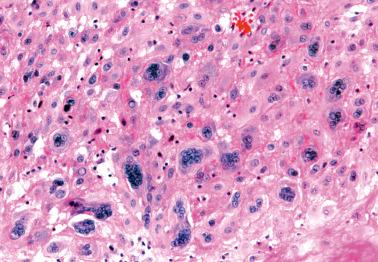
Compared to the normal implantation site, the IT cells within the endometrium and superficial myometrium are increased in number and often show striking degrees of nuclear atypia and multinucleation.
The cells are usually disposed singly or in small nests, although small sheets may be seen. As in the normal placental site, vascular involvement may be seen. Necrosis is absent.
The immunoprofile of the cells is identical to that of normal IT.
EPS is assumed to regress spontaneously and has not been associated with any adverse sequelae. It is not genetically related to the placental site trophoblastic tumor (PSTT), lacking the latter's constant XX genome.
The differential diagnosis is mainly with PSTT.
Features favoring PSTT are absence of a recent pregnancy, the presence of a mass and confluent aggregates of mitotically active cells, destructive myoinvasion, necrosis, and Ki-67 index of >10%. Nuclear atypia is not helpful as it can be striking in EPS. The presence of decidua and villi and a low Ki-67 index (<1%) favor EPS.
In the rare curettage specimens in which a definite distinction between a PSTT and an EPS cannot be made, imaging studies, serum hCG determinations, and a repeat curettage may be informative.
The differential diagnosis with epithelioid trophoblastic tumor is considered under that heading.

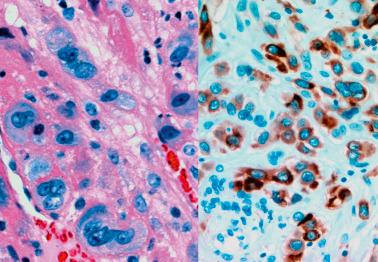
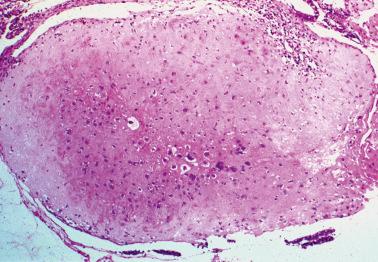
These are nodules or plaques of chorionic-type IT that are usually an incidental finding in the reproductive era but occasionally are discovered in the early postmenopausal years; one was found in a 72-year-old woman. PSNPs are non-neoplastic and have an uneventful follow-up; typical examples have no malignant potential.
The interval from the most recent known pregnancy (which rarely has been a hydatidiform mole) may be many years (average 3 years in one series). Some patients have had a tubal ligation several years before presentation.
PSNPs are usually an incidental finding in a curettage, or occasionally in a hysterectomy, specimen. The most commonly involved sites are the lower uterine segment, the endometrium immediately above this area, or the endocervix. Only about 25% of lesions are grossly visible, usually as a yellow–tan mucosal or superficial myometrial lesion <2.5 cm in size.
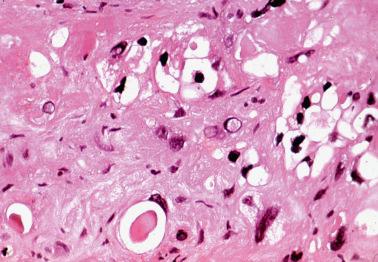
Microscopic examination reveals single or multiple, typically well-circumscribed, paucicellular nodules or plaques, sometimes with lobulated margins, within or on the surface of the endometrium or endocervix, or within the myometrium or endocervical stroma. Small rounded pseudopods may project from their periphery.
Intermediate trophoblastic cells are disposed singly or in irregular clusters, cords, or rounded nests within a usually abundant eosinophilic to hyalinized matrix. Focal necrosis may be seen and rarely there is cystic degeneration or calcification. Necrotic or hyalinized chorionic villi are present in occasional cases.
The lesional cells often appear degenerative with cytoplasm that varies from scanty to abundant and amphophilic to clear or vacuolated; rounded eosinophilic hyaline bodies may be seen. Their nuclei are often irregular and lobated (sometimes multiple) and vary from hyperchromatic to pale and vesicular; mitotic figures are typically absent or rare.
The cells are strongly and diffusely reactive for CK, EMA, p63, p40, CD10, and inhibin-α. Markers of implantation site intermediate trophoblast are less frequently or more focally present, including hPL and Mel-CAM (CD146). The mean Ki-67 labelling index is low (<5%).
‘Atypical placental site nodule’ (APSN).
This designation was applied by to PSNs >5 mm in size and with histologic features intermediate between typical PSNP and epithelioid trophoblastic tumor (see corresponding heading); none of their APSNs recurred.
Subsequently, applied the term APSN to a PSNP with one or more of: cytologic atypia, increased cellularity with small cohesive nests or cords, mitoses, necrosis, and a raised proliferation index. Synchronous or subsequent malignant GTD developed in 3 of 21 patients within 16 months of diagnosis; none had raised hCG levels.
found that 22% of PSNs in their study were atypical as defined by 2–3+ atypia or a borderline proliferation index (~10%). None progressed to ETT.
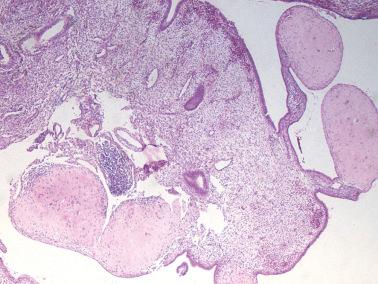
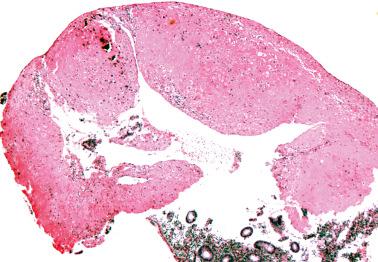
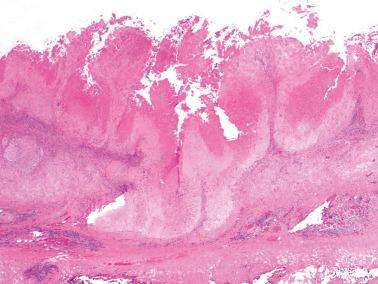
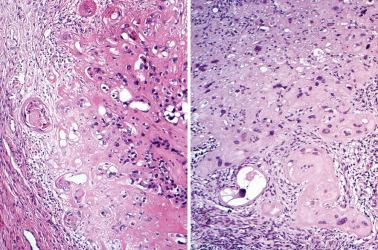
Hyalinized decidua. The distinction between decidual cells and intermediate trophoblast has been considered above (see Normal Intermediate Trophoblast ).
Exaggerated placental site. PSNPs differ in their sharp circumscription, hyalinization, usual lack of association with a current or very recent pregnancy, and usual positivity for p63 and p40.
Placental site trophoblastic tumor. Features favoring or indicating a diagnosis of PSNP include small size, circumscription, multiplicity, striking hyalinization, degenerative appearance of the cells, mitotic inactivity, p63 reactivity, and a Ki-67 index of <5%.
Epithelioid trophoblastic tumor (see corresponding heading).
Postcesarean section PSNP-like lesion. reported two such cases and found two previously reported cases. The lesions were within the anterior uterus at the site of a cesarean section scar and were characterized by a subserosal cyst with a fistulous connection with the endometrial cavity. The cysts and fistulas were lined by intermediate trophoblastic cells similar to those of PSNPs and possibly derived from implantation of intermediate trophoblast within the cesarean section scar.
Hyalinizing squamous cell carcinoma (potentially suggested by PNSP's eosinophilic cytoplasm and the pseudopods noted above). Features indicating a PSNP include typical circumscription, mitotic inactivity, an absence of squamous differentiation, a CD10+/inhibin-α+/CK18+/p16− profile, and a Ki-67 index of <5%.
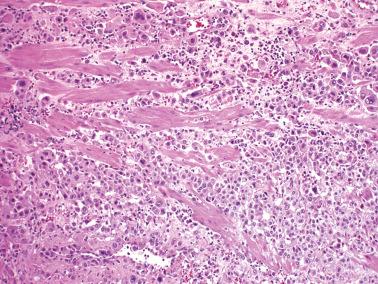
PSTTs typically occur in the reproductive era (mean, 32 years), although rare patients are postmenopausal. Amenorrhea and an enlarged uterus may suggest a normal pregnancy and an episode of abnormal vaginal bleeding in this setting may suggest a missed abortion.
Low-level elevation of the serum hCG is present in ~75% of cases. The free β subunit is the predominant form of hCG in patients with PSTT, unlike other forms of GTD.
There is usually a history of an antecedent normal pregnancy, nonmolar abortion, or hydatidiform mole, with a median interval of 18 months from the gestation. ) found that the antecedent pregnancy was female in 78% of cases.
In a study of 55 cases, found that 84% of PSTTs were stage I vs 67% from a literature review, suggesting that high-stage and fatal cases are more likely reported.
Rare presentations or initial clinical findings have included metastatic disease, spontaneous uterine perforation, virilization secondary to hCG-induced ovarian stromal hyperthecosis, and paraneoplastic phenomena (erythrocytosis, hyperprolactinemia, nephrotic syndrome).
Rare PSTTs have been reported in the vagina ( Chapter 3 ) and fallopian tube ( Chapter 11 ).
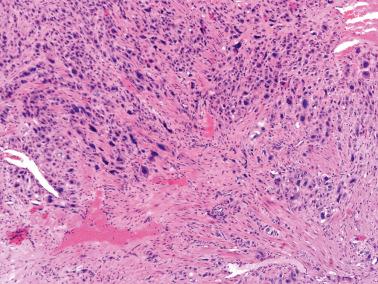
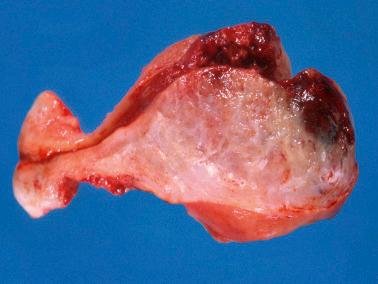
There is usually an endomyometrial polypoid and/or endophytic mass (mean size, 5 cm) unless most of it has been removed by curettage. The tumors vary from circumscribed to ill defined, and usually have a fleshy, yellow or tan sectioned surface, often with focal hemorrhage and necrosis. About 10% of tumors extend into the cervix.
About half of PSTTs invade into the outer third of the myometrium, some extending to the serosa or even into the broad ligament. Spontaneous or curettage-related uterine perforation may complicate such cases.
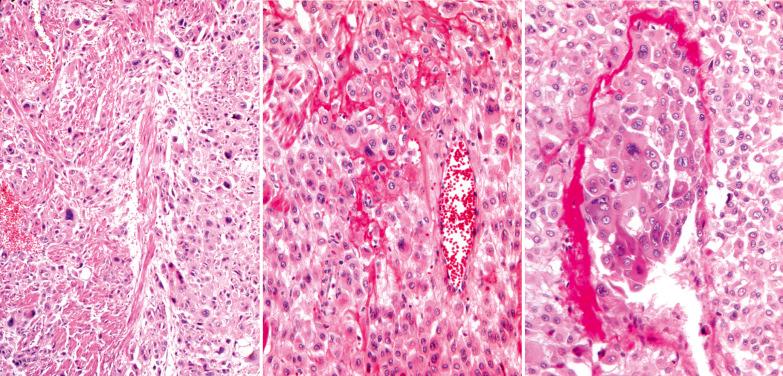
Although occasional tumors are predominantly well circumscribed, most tumors are at least focally infiltrative, with single cells or sheets of cells characteristically separating individual or bundles of myometrial smooth muscle cells.
The tumors typically consist of a relatively monotonous population of polygonal intermediate trophoblastic cells with moderate amounts of amphophilic or eosinophilic cytoplasm, or in occasional tumors, a minor or predominant population of clear cells. Spindle-shaped cells (especially in myoinvasive tumor) may also be present.
Most of the tumor cells are mononucleate but some are binucleate or multinucleate, and rare cells may resemble syncytiotrophoblastic-type giant cells. The variably atypical nuclei are commonly convoluted and have cytoplasmic inclusions, and occasionally, large nucleoli. found a mitotic rate of 0–20 mf/10 hpf (mean 5); atypical mitoses are common.
Fibrinoid material may surround groups of tumor cells and replace vessel walls. There is usually a distinctive pattern of vascular invasion in which tumor cells infiltrate and replace vessel walls and line and fill their lumina. Necrosis is common and may be extensive.
Occasional PSTTs contain a component resembling choriocarcinoma or epithelioid trophoblastic tumor (see corresponding heading).
The endometrium may show a decidual reaction, the Arias-Stella reaction, or both. Normal or molar chorionic villi are only rarely present.
Almost all tumors strongly react for CK, hPL, Mel-CAM (CD146), inhibin-α, HLA-G, and CD10. hCG staining is focal and usually confined to multinucleated cells. The Ki-67 index is usually >10%. Conspicuous membranous staining of PD-L1 has been shown in cases of PSTT, suggesting a possible role for immune checkpoint inhibitor therapy ( , )
PSTTs appear to always contain a paternal X chromosome, that is, are always preceded by a female gestation.
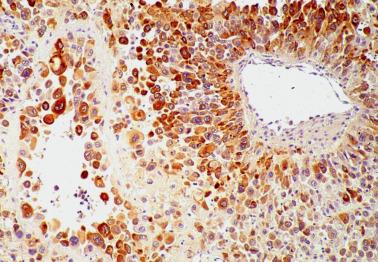
Exaggerated placental site, placental site nodule, and epithelioid trophoblastic tumor (see corresponding headings).
Choriocarcinoma (CCA).
Differential features indicating or favoring CCA include high serum hCG, a hemorrhagic mass, a biphasic growth composed of CT and ST, diffuse staining for hCG, and staining for SALL4. CCAs usually lack the fibrinoid material and the distinctive pattern of vascular invasion of PSTTs. The Ki-67 proliferative index is much higher in CCAs than in PSTTs.
Poorly differentiated PSTTs are not always distinguishable from atypical CCAs, and some CCAs have areas resembling PSTT that may suggest a trophoblastic tumor of mixed type. A diagnosis of pure PSTT should be reserved for well-sampled tumors in a hysterectomy specimen.
Epithelioid leiomyosarcoma ( Chapter 9 ). The clinical setting and associated symptoms of PSTTs usually differ from those of a leiomyosarcoma, as does PSTT's distinctive pattern of myometrial and vascular invasion, fibrinoid material, and staining for hCG, inhibin, and hPL, but not smooth muscle markers.
Carcinomas (clear cell carcinoma, cervical squamous cell carcinoma) and metastatic malignant melanoma.
The clinical features of PSTT (young age, amenorrhea, history of pregnancy, elevated hCG level) and its microscopic features (characteristic myoinvasion and vascular invasion, hPL/hCG/inhibin reactivity, and progestational endometrial changes) differ from those of most uterine carcinomas and metastatic melanoma. Negative staining for melanoma markers help exclude the latter diagnosis.
The presence of more typical invasive squamous cell carcinoma, keratin, and an SIL also facilitate the differential with squamous cell carcinoma. Also, most cervical squamous and adenosquamous carcinomas, in contrast to PSTTs, are CEA+, and strongly and diffusely p16+ (caveat: PSTTs may show weak focal p16 positivity). Also, the aforementioned carcinomas are negative for HLA-G, hCG, and CD10.
found that 30% of PSTTs were clinically malignant, almost half of them fatal. In a more recent large series from China, found a mortality rate of 6.5%.
The most common metastatic sites are lungs, pelvis/vagina, and liver; lymph nodes are involved in 6% of cases. Metastatic and recurrent PSTTs generally respond poorly to chemotherapy.
Most clinically malignant tumors are associated with one or more of: age >35 years, interval since last pregnancy of >2 years, previous term birth (vs another type of antecedent pregnancy), myometrial invasion beyond the inner one-third, stage III or IV, maximum hCG level >1000 mIU/ml, extensive necrosis, >5 mf/10 hpf, and tumor cells with clear cytoplasm.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here