Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Meningiomas are intracranial, extra-axial lesions that represent about one-third of all primary brain tumors, and over a half of noncancerous central nervous system (CNS) neoplasms. Falcine meningiomas are relatively rare. They arise from the falx cerebri and make up approximately 5% to 9% of all intracranial meningiomas. , Falcine meningiomas differ from parasagittal meningiomas in that they arise from the dense fibrous tissue of the falx, rather than the dura mater enclosing the superior sagittal sinus (SSS); the latter being up to 5 to 7 times more common that falcine lesions. , Cushing further distinguished falcine meningiomas from parasagittal tumors by how, from the surgeon’s perspective, falcine tumors are often concealed by overlying cerebral cortex ; however, large falcine meningiomas can grow superiorly to secondarily invade the SSS.
Falcine meningiomas and convexity meningiomas are more often found to have atypical features than skull base or posterior fossa meningiomas, and are more likely to present peritumoral edema. These tumors were first divided by Yasagril into outer tumors, arising from the main body of the falx, and inner tumors, arising in conjunction with the inferior sagittal sinus. Like parasagittal tumors, falcine meningiomas can also be classified for surgical considerations based on their relationship to the SSS and with respect to the coronal and lambdoid sutures. The anterior third of the SSS extends from the foramen cecum to the coronal suture, the middle third extends from the coronal suture to the lambdoid suture, and the posterior third extends from the lambdoid suture to the torcular herophili. A more recent classification based on magnetic resonance (MR) imaging was described by Zhuo and colleagues. This classification scheme divides falcine meningiomas based on their growth pattern as follows: type I, hemispheroid tumor invaginating deeply into one hemisphere without shifting the falx; type II, olive-shaped tumor, shifting the falx to the contralateral side; type IIIA, globular/dumbbell-shaped tumor extending into both hemispheres to different extents; and type IIIB, globular/dumbbell-shaped tumor extending into both hemispheres equally. , These preoperative characterizations and classifications of the tumors may allow for better procedure planning and surgical approach.
Meningiomas are known to be slow-growing lesions that clinically manifest depending on their location. Falcine meningiomas located at the level of the anterior third of the SSS often present somewhat insidiously and result in a frontal lobe syndrome ( Figs. 24.1A–F and 24.2A–H ). Slowly progressive symptoms include short attention span, poor short-term memory, personality changes, apathy, and emotional instability; often this presentation is confused with age-related dementia, delaying diagnosis. Consequently, anterior third tumors are frequently larger in size at the time of presentation than tumors located in other regions. In extreme cases, patients can also present with signs of increased intracranial pressure, including headache, papilledema, or optic atrophy. Of patients with anterior third falcine meningiomas, approximately 25% present with seizures, but they are less frequent and less localizing than when tumors abut motor cortex.
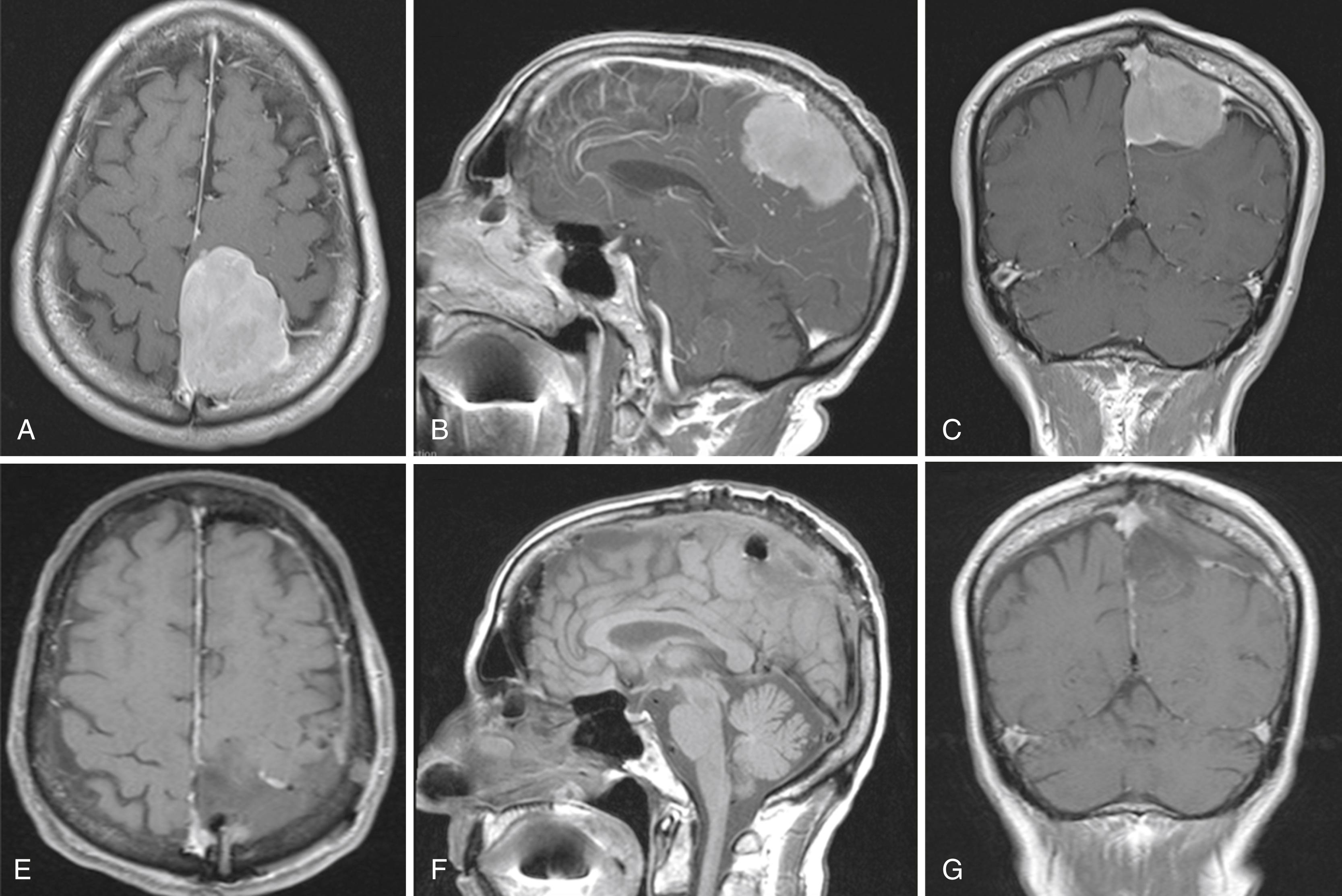
Tumors along the middle third of the SSS are usually located adjacent to the motor and sensory regions. These produce more focal and localizable symptoms that cause patients to seek earlier medical intervention. Earlier presentation of symptoms allows for earlier diagnosis; thus, tumors in this region rarely attain the same bulk as tumors localized in the anterior third. While symptoms of increased intracranial pressure are rare, tumors in this region are more likely to produce spastic weakness and focal seizures that involve the contralateral foot and leg. Both motor cortices may be affected if the tumor extends bilaterally from the midline. Falcine meningiomas located at the middle third confer the highest risk of surgery-related morbidity. ,
Patients with meningiomas involving the posterior third of the sagittal sinus usually present with persistent headache and often are found to have visual problems during examination ( Fig. 24.3A–E ). The degree of visual field involvement depends on the size and location of the tumor. Smaller tumors located above the calcarine fissure can cause anopia to the inferior quadrant. Smaller tumors along the tentorium cerebelli may involve the visual cortex below the calcarine fissure and only produce anopia in the upper quadrants. Larger tumors, on the other hand, can case homonymous hemianopsia with macular sparing. Some patients may present with reports of visual hallucinations. Tumors of the middle and posterior third can occasionally present with bilateral symptoms as the tumor extends across the falx. Meningiomas can also manifest with the patient presenting nonspecific mass effect symptoms.
Preoperative imaging allows neurosurgeons to establish a diagnosis, to evaluate tumor characteristics before surgery, to trace tumor growth over time, to predict tumor grade and behavior, and to determine the course of treatment. Meningiomas are often evident in computed tomography (CT) scans, appearing as well-circumscribed homogeneous and hyperdense lobular lesions. This imaging modality is also useful for evaluation of tumor-related bone changes, such as hyperostosis and osteolysis. However, due to anatomic distribution, falcine meningiomas seldom present with osseous involvement.
MR imaging is the gold standard for preoperative assessment of falcine meningiomas. The majority of meningiomas typically appear iso- or slightly hypointense relative to the cortex on noncontrast T1-weighted MR images, and hyperintense on T2-weighted sequences. These tumors present with a rounded edge and smooth appearance, which compresses the brain parenchyma instead of invading it. The T2-weighted characteristics can be variable and are believed to correlate best with histologic subtype, vascularity, consistency, and extent of dural invasion. , , Tumors that are hypo- to isointense on T2-weight imaging are found to be firmer and are histologically more fibroblastic or transitional (see Figs. 24.1D and 24.3D ). More malignant meningiomas, on the other hand, are more likely to exhibit greater brain edema, which manifests as increased hyperintensity on T2-weighted imaging. , Aggressive meningiomas also manifest with a heterogeneous MR imaging appearance characterized by the presence of intratumoral cyst formation, hemorrhages, or necrosis.
Falcine meningiomas frequently extend bilaterally, acquiring a dumbbell or bilobed shape with invagination into the medial aspects of both left and right hemispheres. Attempts to use MR imaging to differentiate histologic types of meningiomas have thus far been limited. Although efforts to differentiate benign from atypical meningiomas using MR spectroscopy (MRS) have been attempted, noninvasive measures have been unreliable.
For preoperative planning, multiplanar T1-weighted gadolinium-enhanced MR images can provide coronal, sagittal, and axial views that help in defining the tumor’s anatomic location, size, and cortical involvement (see Figs. 24.1A–C and 24.2A–C ). Brain edema adjacent to the tumor can be a sign of pial invasion and can be assessed preoperatively using T2-weighted images and fluid-attenuated inversion recovery (FLAIR) sequences (see Fig. 24.1E ). Three-dimensional (3D) T1-weighted gadolinium-enhanced multiecho, magnetization-prepared, rapid gradient echo images can also be used with neuronavigational tools to provide surgeons with intraoperative guidance. MR imaging can also be used as a tool for disease prognosis. Peritumoral edema, presence of heterogeneity, tumor necrosis, and tumors located in the falx or convexity have been demonstrated to correlate with high-grade meningioma features that can be observed preoperatively on imaging. Cerebral angiography can provide essential information delineating the tumor’s arterial feeding pattern. Additionally, the arterial phase of the angiography can provide crucial information on the course, displacement, and possibly the encasement of the anterior cerebral arteries (ACAs), pericallosal arteries, and callosomarginal arteries. The venous phase of the angiogram is also vital toward determining the patency of the SSS, understanding the anatomy of the draining cortical veins in relation to the tumor, and the degree of collateral circulation. If the sinus is occluded, the anatomy of the draining collateral vessels needs to be clearly identified during preoperative planning and meticulously preserved during surgery. Inadvertent occlusion of collateral vessels may result in severe postoperative complications such as venous infarct and cerebral edema ( Fig. 24.4 ).
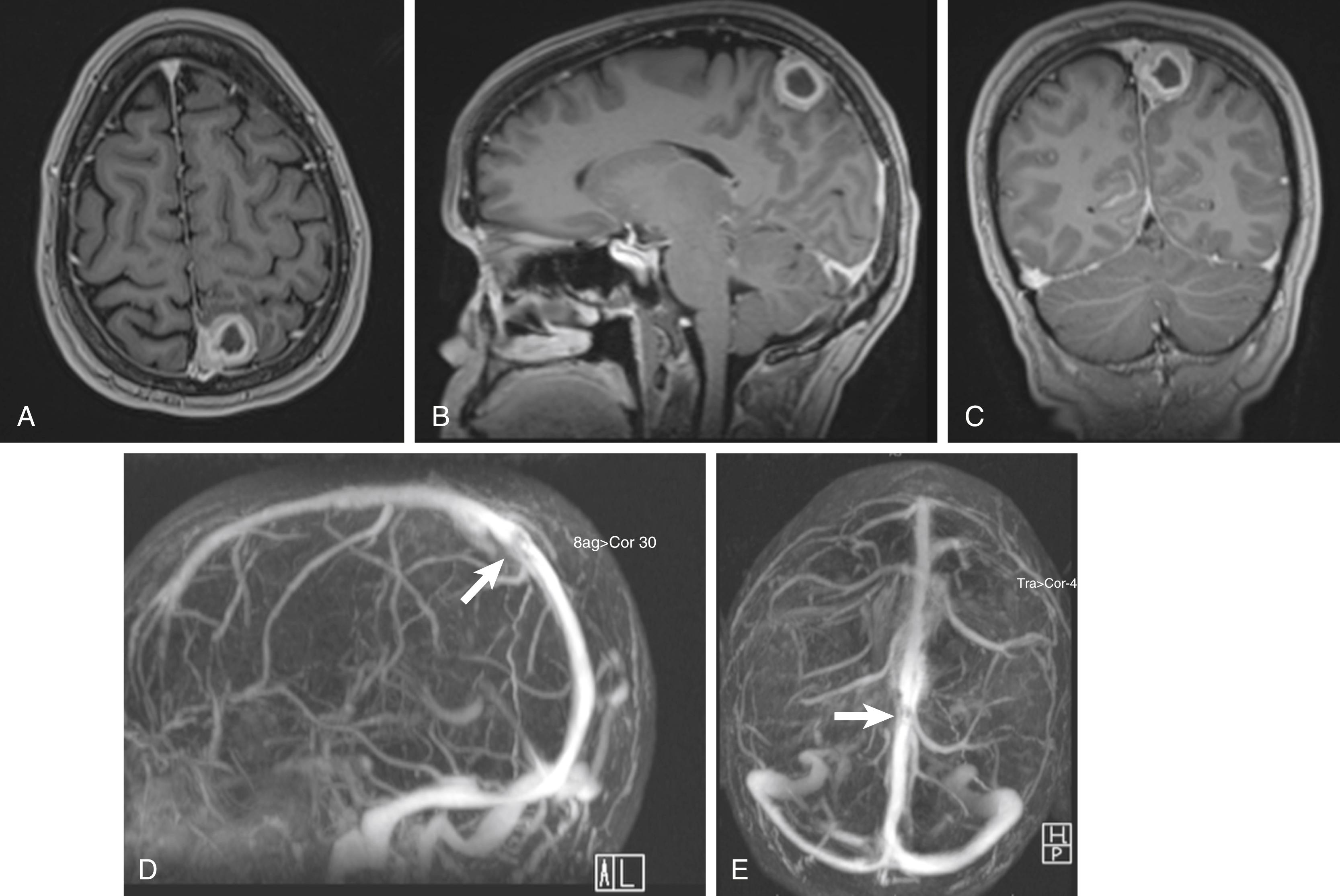
Digital subtraction angiography (DSA) provides a dynamic view of both the arterial and venous phases, better resolution of small vessel feeders, better accuracy in identifying sinus obstruction, and areas with reversal of normal venous flow. However, DSA is invasive and associated with a small, but not insignificant, risk for strokes, vessel injury, post-diagnostic hematoma, and possible renal injury from contrast use. Noninvasive means to evaluate the cerebral vasculature include MR angiography and CT angiography. MR angiography/venography capitalizes on its intrinsic sensitivity to flow to detect the patency of vessels. Vessels with high flow give rise to high signal intensity while the absence of flow is characterized by reduced signal intensity. , The technical limitations to MR angiography include its susceptibility to flow-related artifacts: for example, patent but obstructed regions of the sinus may appear completely occluded due to decrease in flow; intracellular deoxyhemoglobin/methemoglobin found in clot can create false signals to suggest a patent vessel. Other practical limitations include longer image acquisition times and sensitivity to patient movement. CT angiography/venography (CTA/CTV) is acquired using thin 0.625-mm slices on a helical 64-channel multidetector scanner during the peak arterial and venous enhancements. The acquired images can be viewed using maximum intensity projection (MIP) images, multiplanar reconstruction (MPR) images, and Vitrea 3D reconstruction images (Vital Images, Minnetonka, MN) to provide the surgeon a better sense of the tumor and its relationship to the vasculature (see Fig. 24.2F–J ). The faster image acquisition times as well as the ability to maintain high spatial resolution has made CTA/CTV one of the preferred methods for preoperative vessel assessment. CTA/CTV has better small-vessel and sinus resolution and fewer motion and flow artifacts than MR venography. , In a study by Wetzel and colleagues, CTV was compared to digital subtraction angiography. CTV was found to have an overall sensitivity of 95% and 91% specificity, while DSA had 90% sensitivity and 100% specificity. Additionally, the shorter image acquisition times allow for fewer motion artifacts, and thus makes CTA/CTV better suited for uncooperative or sick patients.
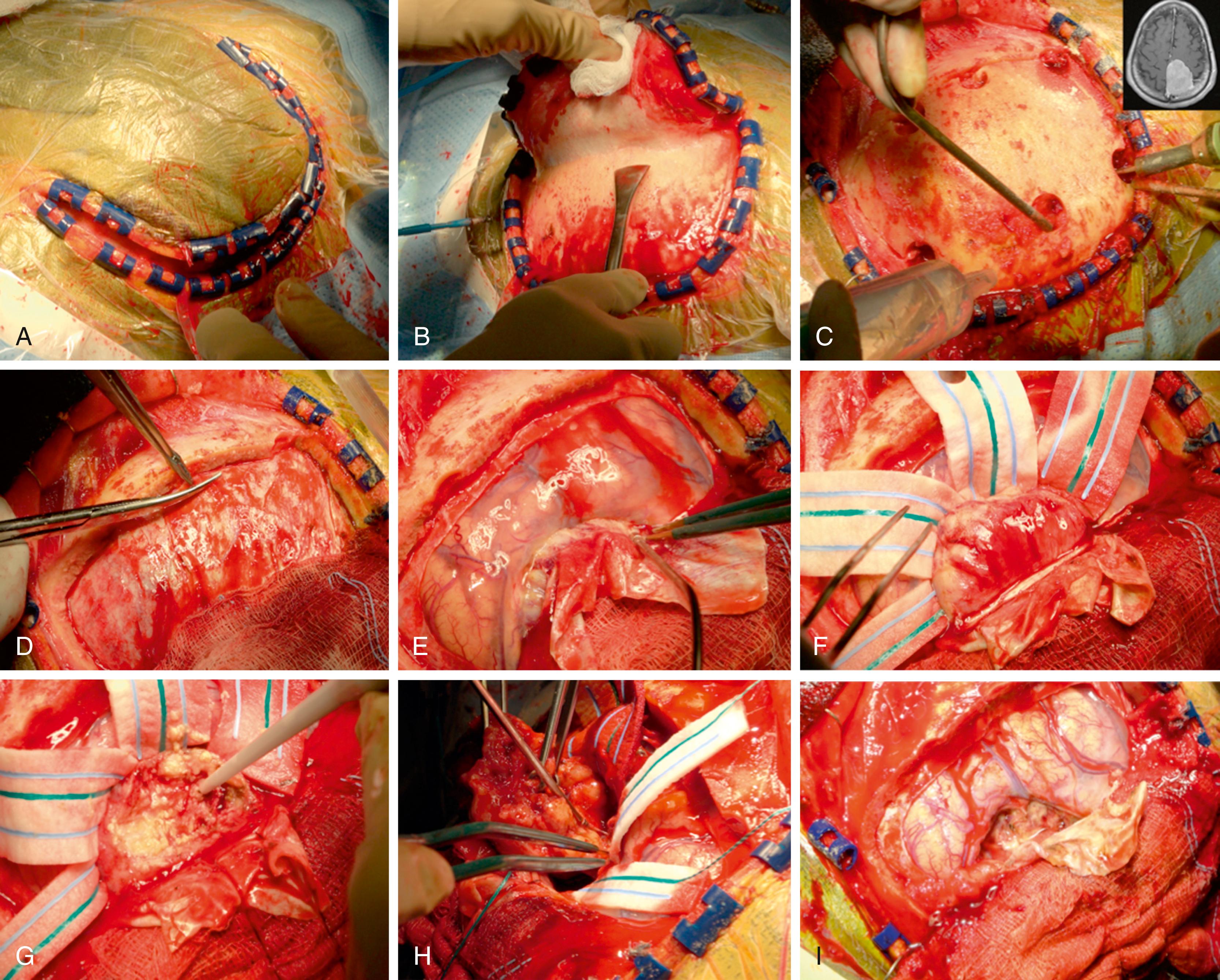
The operative technique is highly influenced by tumor location, vasculature, relationship with adjacent structures, possible surgical corridor—and of course patient characteristics, symptoms, and comorbidities. After diagnosis, meningioma growth can be evaluated over time and treated conservatively. Surgical treatment is usually indicated for patients with neurologic symptoms, associated brain edema, increased intracranial pressure, and large tumor volumes. The goal of surgery should always be to achieve a maximum safe resection of the tumor, while minimizing complications by performing a careful identification and manipulation of blood vessels, protection of adjacent eloquent structures—such as the paracentral lobule in the case of middle falcine meningiomas—and avoiding excessive brain retraction. Postoperative extent of resection in meningioma surgery is determined by the Simpson Scale. Simpson Grade 1 to III are considered to be gross total resections, while Grade IV and V fall into the subtotal resection groups ( ).
The patient is brought to the operating room and general anesthesia is administered via endotracheal intubation. A combination of intravenous furosemide, mannitol, and slight hyperventilation is used to reduce intracranial pressure. Preoperative antibiotics, stress dose steroids, and antiseizure prophylaxis (such as fosphenytoin) are also given prior to skin incision. If the preoperative plan involves resection or manipulation of the sagittal sinus, air emboli precautions are implemented. A precordial Doppler is securely placed over the right atrium to monitor for air emboli and an intra-atrial venous catheter is placed for aspirating the air bubbles if necessary. Intraoperative monitoring with somatosensory evoked potentials or direct cortical stimulation is often used when tumors are located adjacent to the motor or sensory areas, and attaching leads should be completed prior to incision.
Patient positioning depends on the preoperative evaluation and classification of the tumor. For tumors located in the anterior third of the sagittal sinus, the patient is placed in a supine position with the head slightly elevated. Tumors around the middle third of the sagittal sinus can be approached with the patient supine with the head elevated and flexed, or, alternatively, a semiprone or lateral approach can be used, especially if the tumor is located more posteriorly. A semilateral, semisitting position with the head well elevated so that the scalp over the area of the tumor is at the highest point can also be considered for tumors in this area. For tumors involving the posterior third, the patient is placed in the lateral position and the head is elevated and turned to the opposite side so that the center of the tumor is uppermost. , The head is secured with the Mayfield 3-point fixation head rest and the patient’s body is secured to the operative table with tape. Foam padding and gel rolls are used to pad all pressure points.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here