Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Rhabdomyosarcoma (RMS) is the most common form of soft tissue sarcoma, accounting for 5% of all childhood cancers. It is the third most common pediatric extracranial solid tumor after Wilms tumor and neuroblastoma. RMS is a malignant tumor of mesenchymal origin and along with neuroblastoma, primitive neuroectodermal tumors (PNETs), and lymphoma form the group of small, round blue cell tumors of childhood. Its incidence is estimated at 350 cases each year in the United States with approximately 6 cases per 1,000,000 population per year. RMS has a Caucasian male predominance and presents with a bimodal age of distribution with peaks between ages 2 and 6 years and again between 10 and 18 years. However, more than 80% of cases are diagnosed before 14 years of age. This bimodal incidence correlates with the occurrence of the two major histologic subtypes of RMS: embryonal rhabdomyosarcoma (ERMS) for early childhood, which typically presents in the head, neck, and genitourinary (GU) regions, and alveolar RMS (ARMS) for the later childhood and adolescent years and is commonly located in the trunk and extremities. The overall incidence of the two subtypes is approximately 65–75% (ERMS) and 25–32% (ARMS). In terms of site, greater than one-third (35%) of RMS occurs in the head and neck, followed by GU and extremity presentations.
Most cases of RMS are sporadic; however, RMS has been associated with familial syndromes, including Li Fraumeni and neurofibromatosis type 1 ( NF1 ). Li Fraumeni patients with RMS are linked to a germline mutation of the tumor suppressor gene p53 and activation of the RAS oncogene. These patients present with disease at an early age and may develop other malignancies, including premenopausal breast cancer, leukemia, and adrenocortical carcinoma. NF1 patients also have an increased risk of malignant non-neurofibroma tumors, including RMS, and, like Li Fraumeni patients, they have an early age at presentation. Other syndromes associated with RMS include Beckwith–Wiedemann, Noonan, Costello, and hereditary retinoblastoma. The use of marijuana or cocaine during pregnancy has been linked to the development of RMS. Finally, up to one-third of children with RMS have congenital abnormalities found during autopsy.
RMS is thought to arise from pluripotent mesenchymal cells with disrupted cell growth and differentiation. However, its exact pathogenesis remains unclear. Causal relationships have been suggested for the MET proto-oncogene, macrophage migration inhibitory factor (MIF), and p53 with regard to oncogenic transformation and tumor progression. The immunohistochemical (IHC) stains used to identify RMS include desmin, myogenin, MyoD1, and muscle specific actin. ERMS is characterized by a loss of heterozygosity at the 11p15 locus in up to 80% of patients. The insulin growth factor 2 ( IGF-2 ) gene lies within this locus and can be overexpressed due to paternal allele duplication. ERMS has been associated with other genetic aberrations, including fibroblast growth factor receptor 1 ( FGFR1 ) and neuroblastoma RAS viral oncogene homolog ( NRAS ), and has high cytologic variability representing progressive states of muscle morphogenesis. Mutations of MYCN and CDK-4 are more common in ARMS, and its appearance resembles pulmonary parenchyma in IHC stains.
The molecular diagnosis of chromosomal translocations has transformed tumor diagnostics. Specifically, reverse-transcription polymerase chain reaction (RT-PCR) and fluorescence in situ hybridization (FISH) for the detection of fusion proteins have been essential in progressing beyond simple IHC. Patients with ARMS are noted to express fusion proteins arising from the fusion of the FOXO transcription factor gene with either PAX3 (55%) or PAX7 (23%) transcription factors. In these fusions, the DNA binding domain of PAX is combined with the regulatory domain of FOXO , resulting in increased PAX activity leading to dedifferentiation and proliferation of myogenic cells. The PAX7-FOXO fusion results in worse overall survival (OS). Twenty percent of ARMS cases that do not express fusion status are known as fusion-negative and have similar biologic behavior to ERMS tumors with the same loss of heterozygosity at 11p15.5, and also have comparable OS and event-free survival (EFS). Studies have shown that these translocations, rather than what is seen on histopathologic examination, appear to determine the poorer outcome noted for patients with alveolar subtype. Fusion-positive ARMS patients have been shown to have a higher rate of metastasis compared with those who are fusion-negative. Myogenin expression is also associated with alveolar histology and thus carries a worse prognosis. Fusion status will replace tumor histology for the classification of RMS in future treatment protocols.
The two major groups of RMS are further subdivided according to histologic findings. ERMS includes botryoid, spindle cell, and dense patterns. Having the botryoid and spindle cell histology portends a better prognosis. Spindle cell histology is commonly found in paratesticular lesions, and botryoid tumors are often found in hollow viscus such as the bladder, vagina, and biliary tree. ERMS is characterized by regions of loose myxoid mesenchymal tissue alternating with dense cellular regions with rhabdomyoblasts in various stages of differentiation ( Fig. 69.1 ). In contrast, in the dense subtype without myogenic differentiation, sheets of primitive cells with scant cytoplasm and ovoid nucleus are seen.
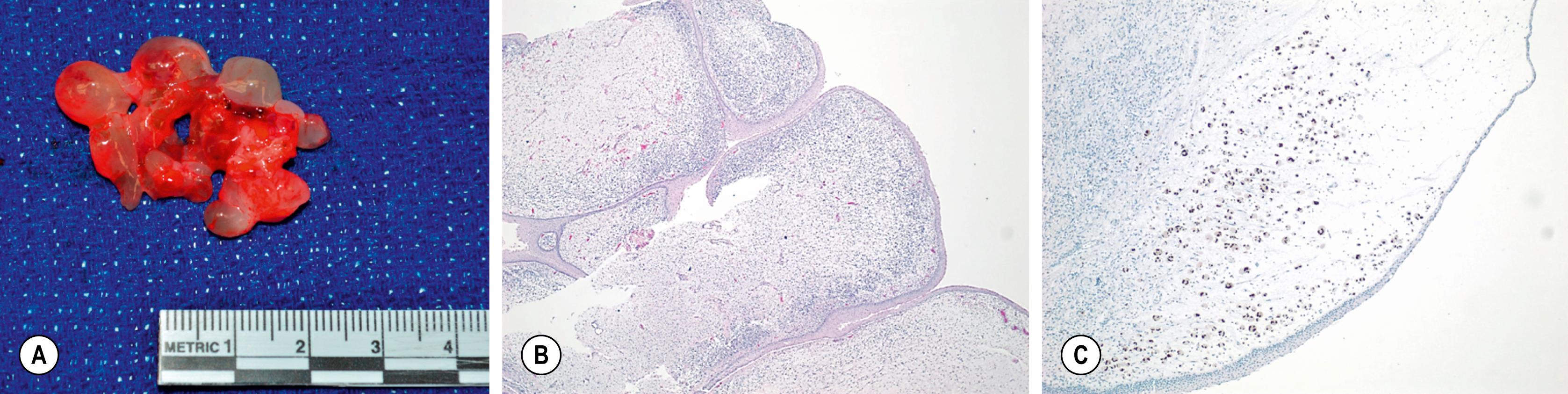
ARMS is subdivided into classic and solid patterns, and requires more than 50% of the specimen to be alveolar in nature to be classified as such. Classic ARMS cells contain eosinophilic cytoplasm arranged in nests separated by fibrous septae with islands of tumor cells, whereas the solid pattern lacks the dividing septae and is characterized by sheets of monomorphic cells with round nuclei ( Fig. 69.2 ).
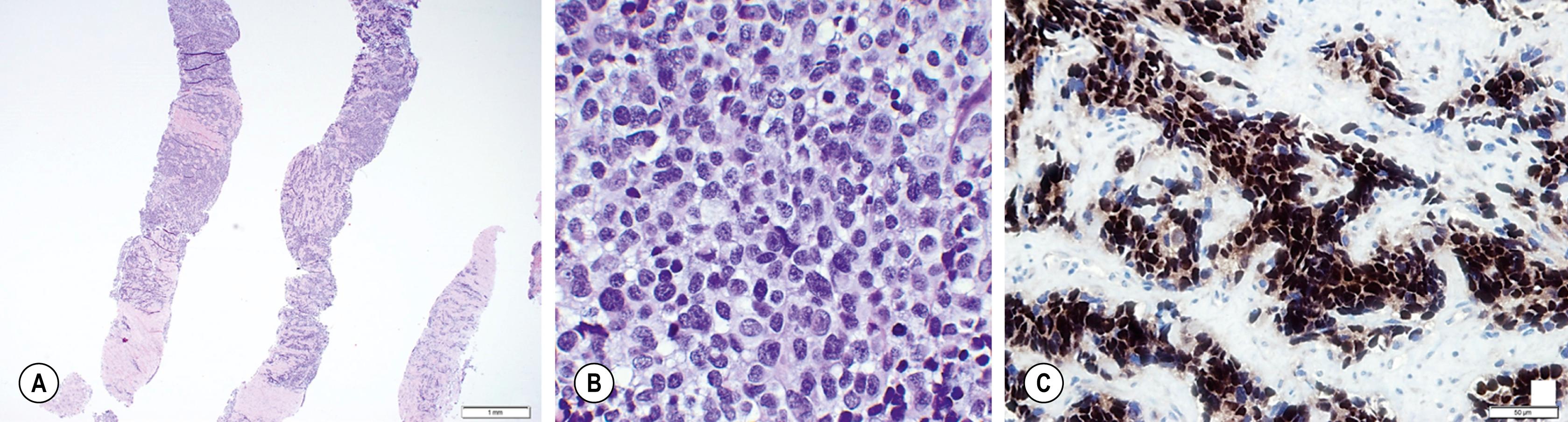
Symptoms of RMS will depend on the location and size of the primary disease. However, most patients present with an asymptomatic mass. Patients can also have signs and symptoms secondary to mass effect on adjacent structures and complications due to compression. RMS involving the head and neck are most commonly embryonal subtype and can present with proptosis, ophthalmoplegia, cranial nerve palsies, and meningeal symptoms.
Paratesticular RMS can present with painless swelling in the scrotum and is known to have a high rate of retroperitoneal lymph node metastases particularly among boys >10 years of age. Tumors involving the GU tract can present with obstruction, constipation, or urinary frequency. Patients with vaginal RMS are usually younger and present with bleeding, discharge, or fullness secondary to the mass effect ( Fig. 69.3 ). Uterine involvement usually has extensive disease at diagnosis. Perineal/perianal RMS most commonly has alveolar histology, frequently involves regional lymph nodes, and has a poor prognosis with a 5-year OS around 45%. Extremity RMS usually presents as a painless mass. These tumors are more aggressive and are usually ARMS. Almost half of them will involve regional lymph nodes at presentation. Neonatal presentation of RMS is extremely rare, with most cases presenting with the embryonal botryoid subtype.
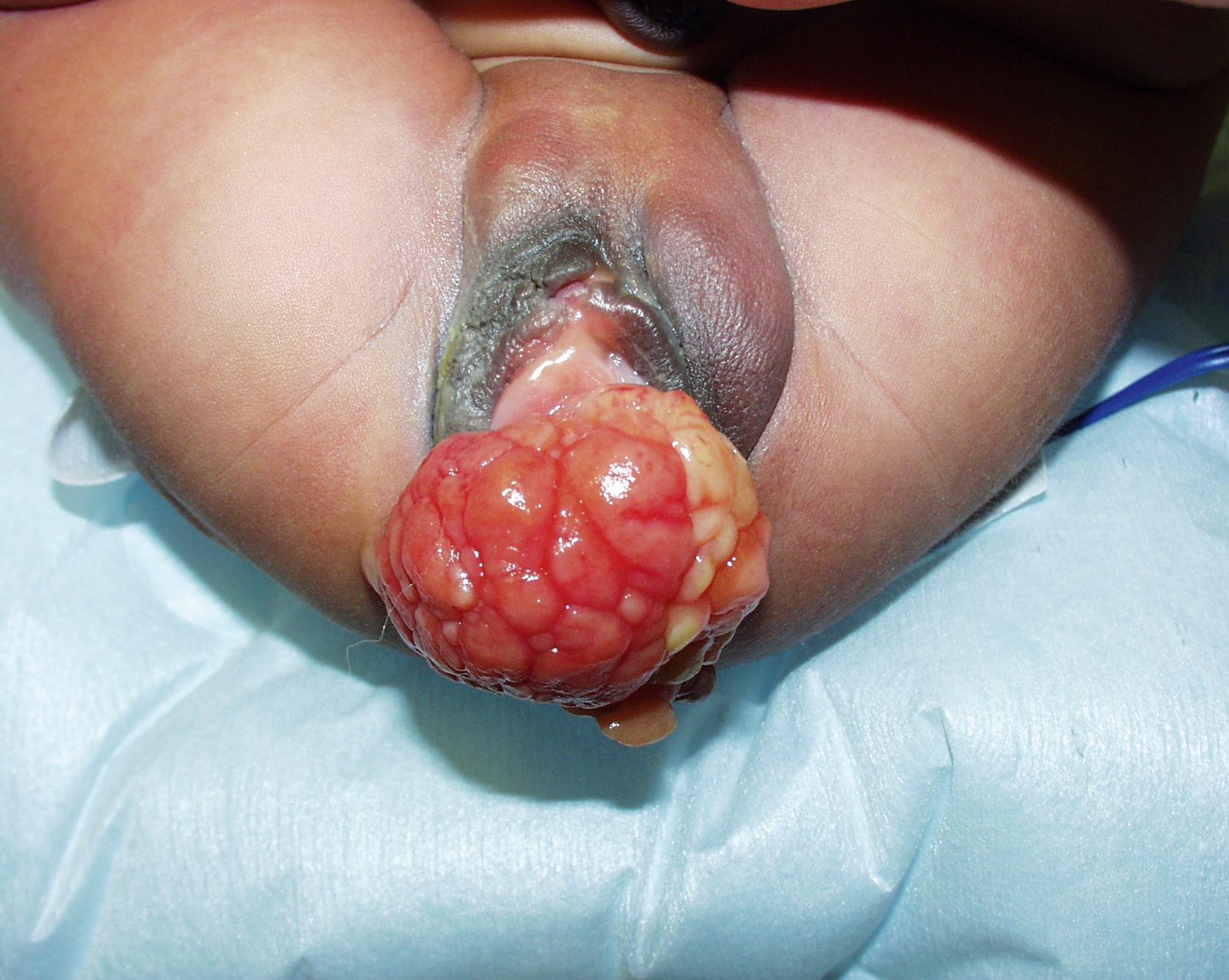
All patients with suspected RMS require a complete workup prior to initiation of treatment, including laboratory examination with complete blood count, electrolytes, renal and liver function tests, coagulation panel, and urinalysis. Cross-sectional imaging studies should be performed on the primary tumor with computed tomography (CT) or magnetic resonance imaging (MRI) to assess its true size and involvement of surrounding structures or vital organs. For most patients, staging generally includes bone marrow biopsy; whole-body bone scan; CT of the brain, chest for lung evaluation, and abdomen with triple-phase contrast for liver assessment; and lumbar puncture for cerebrospinal fluid evaluation. However, recent studies have shown that RMS without evidence of local invasion has a low rate of metastatic disease, and bone marrow biopsy and bone scan are unnecessary in these patients. The use of metabolic imaging with 18 F-fluorodeoxyglucose positron emission tomography ( 18 FDG-PET) in the pediatric RMS population has limited experience and is not yet part of the first-line imaging. Evaluation of occult metastases, regional adenopathy, and persistent or recurrent disease may be improved with PET/CT compared with CT/MRI alone. It is important to note that PET findings during staging or as follow-up imaging after therapy do not correlate with disease burden or outcomes, and can have a positive predictive value up to 90% but a negative predictive value close to 70%.
Clinical and radiographic assessment of regional and distant lymph nodes should be performed in all patients prior to the initiation of treatment as this will guide staging and therapeutic management. Positive regional nodes are irradiated, and positive distant nodes are considered metastatic disease, which upstages the disease and alters therapy. Nodal disease is present in up to 25% of all RMS patients, with a higher incidence in specific primary sites such as the perineum, retroperitoneum, extremity, bladder, and parameningeal and paratesticular regions. Positive lymph node status is also an independent poor prognostic factor for OS and failure-free survival (FFS) in patients with fusion-positive ARMS, but does not appear to be as prognostic for fusion-negative ERMS patients, provided they receive radiation therapy (RT). Indications for nodal evaluation include positive clinical nodes and extremity, trunk, and paratesticular tumors in patients >10 years old, and is recommended in all patients with fusion-positive ARMS given its poor outcome and high incidence of false negative imaging.
Biopsy is necessary to confirm local and disseminated metastatic disease. In the absence of palpable nodes, sentinel lymph node (SLN) biopsy is the technique of choice and should be performed to assess involvement, particularly for patients with extremity or trunk RMS ( Fig. 69.4 ). Completion lymph node dissection is unnecessary and does not improve outcome. Extremity tumors within transit nodes require aggressive evaluation since the incidence of involvement is higher than anticipated, and failure to include these nodal groups in the radiation field increases the chances for local and regional tumor failure.
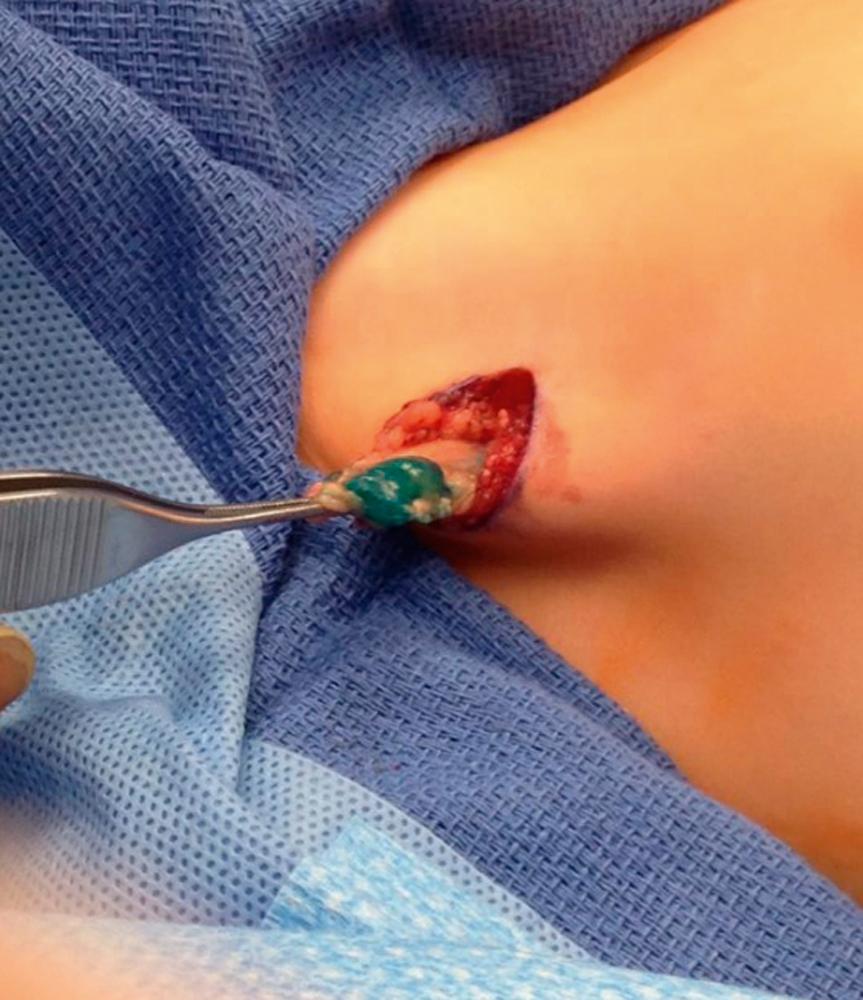
Staging of RMS follows the classic tumor/node/metastasis (TNM) classification and defines a pretreatment system determined by site and size of the primary tumor, degree of invasion, nodal status, and the presence or absence of metastatic disease. It is based on the preoperative physical examination and imaging studies ( Table 69.1 ).
| Stage | Sites | T | Size | N | M |
|---|---|---|---|---|---|
| 1 | Orbit, head and neck, a genitourinary b | T1 or T2 | A or B | N0 or N1 or Nx | M0 |
| 2 | Bladder/prostate, extremity, cranial, parameningeal, other c | T1 or T2 | A | N0 or Nx | M0 |
| 3 | Bladder/prostate, extremity, cranial, parameningeal, other c | T1 or T2 | A B |
N1 N0 or N1 or Nx |
M0 M0 |
| 4 | Any | T1 or T2 | A or B | N0 or N1 | M1 |
a No parameningeal involvement.
One of the most important prognostic factors in RMS is the extent of residual disease after initial resection. After the initial surgical procedure, the patients are assigned to a clinical group according to the pathologic evaluation of the specimen, which encompasses the completeness of excision (residual disease including margin status) and evidence of tumor metastasis to lymph nodes or distant sites. The clinical group assigned refers to the pathologically determined extent of tumor after resection or biopsy of the primary lesion, along with the lymph node evaluation and the patient’s status prior to the initiation of systemic therapy ( Table 69.2 ).
| Group | Definition |
|---|---|
| Group I | Complete resection of localized disease |
| Group II | Regional resection |
| IIA | Regional resection Inadequate microscopic margins |
| IIB | Regional resection Lymph node disease Adequate microscopic margins |
| IIC | Regional resection Lymph node disease Inadequate microscopic margins |
| Group III | Incomplete resection Biopsy only |
| Group IV | Distant metastatic disease |
The Soft Tissue Sarcoma Committee of the Children’s Oncology Group (STS-COG) created the risk stratification system in an effort to tailor therapy to patient outcomes. It incorporates pretreatment staging (including site and TNM status), clinical group, and histology. It also classifies patients into low-, intermediate-, and high-risk groups. This comprehensive stratification process has shown to be an accurate predictor of outcomes ( Table 69.3 ).
| Risk | Stage | Group | Histology | Overall Survival (%) |
|---|---|---|---|---|
| Low, subset 1 | 1 or 2 1 |
I or II III orbit |
ERMS ERMS |
90 |
| Low, subset 2 | 1 3 |
III non orbit I or II |
ERMS ERMS |
|
| Intermediate | 2 or 3 1, 2, or 3 |
II or III I, II, or III |
ERMS ARMS |
60–80 |
| High | 4 | IV | ERMS or ARMS | 20–40 |
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here