Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
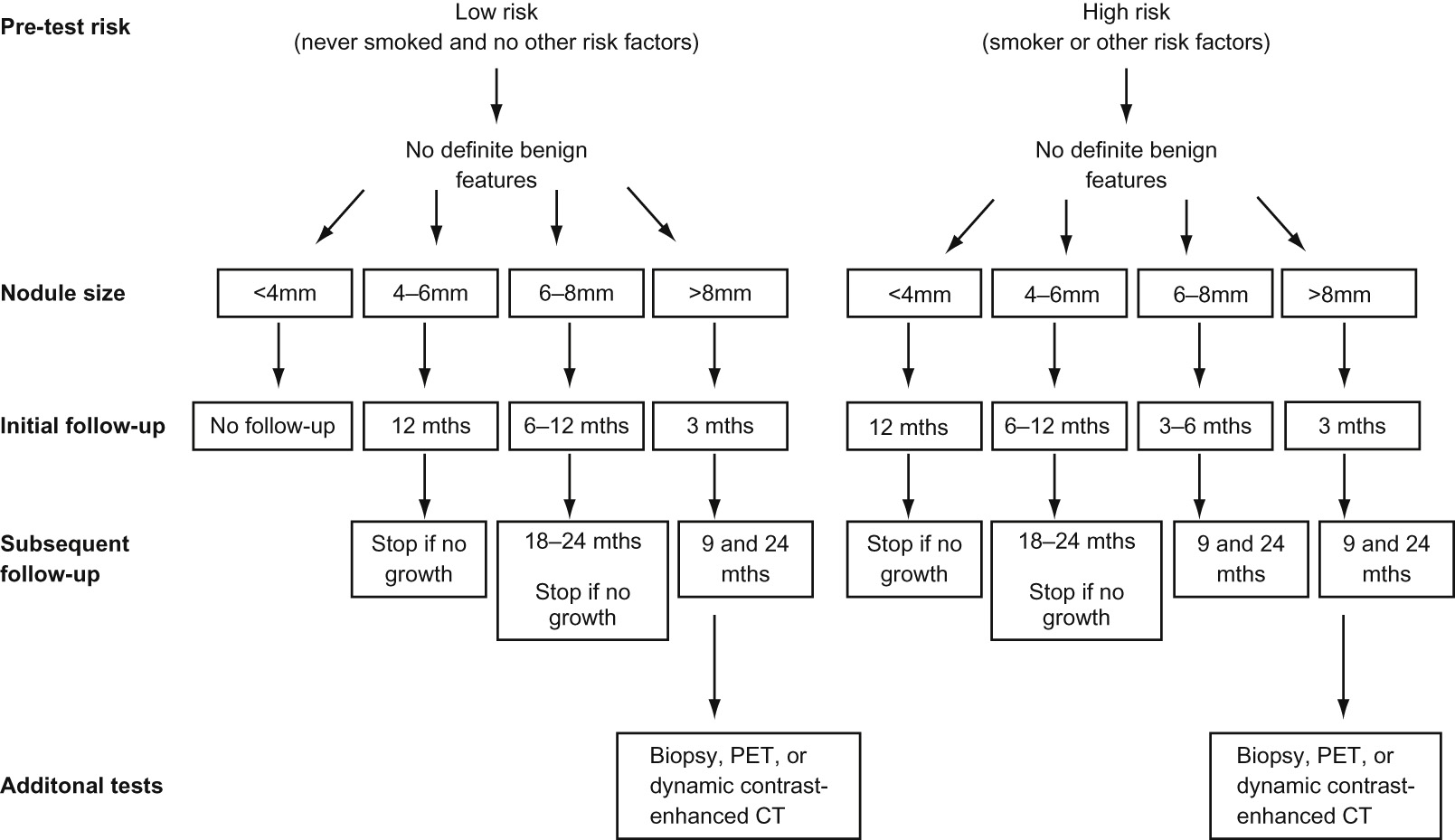
This is defined as a solitary circumscribed pulmonary opacity with no associated pulmonary, pleural, or mediastinal abnormality ▸ it measures < 3 cm in diameter
Many are discovered incidentally (but up to 40% may be malignant)
<10% are due to a solitary pulmonary metastasis
Benign intrapulmonary lymph nodes: nodules <15 mm from the pleural surface ▸ ellipsoid in shape, usually connected to the pleural surface by a fine linear opacity
Ground-glass nodules :
Ground-glass density: focal area of increased lung attenuation (well or poorly defined) through which normal structures can be seen
Pure ground-glass nodule: no soft tissue component
Part solid ground-glass nodule: a solid component obscuring lung architecture
The two primary criteria are the rate of growth (or stability over time) and the attenuation of the nodule ▸ the patient's age is also a significant distinguishing feature (a carcinoma is only seen in < 1% of patients < 35 years old)
Rate of growth/stability over time: benign lesions invariably have a doubling time of < 1 month or > 18 months (bronchoalveolar carcinomas are an exception in that they may have very slow growth rates) ▸ bronchial carcinomas usually have a doubling time of between 1 and 18 months
Attenuation/enhancement: a dense central nidus or lamellated calcification indicates a granulomatous process (e.g. tuberculosis, histoplasmosis) ▸ irregular ‘popcorn’ calcification suggests a hamartoma ▸ fat is virtually diagnostic of a hamartoma ▸ a lack of enhancement (<15HU) following IV contrast medium is indicative of benignity
Granular calcification is seen on CT in up to 7% of carcinomas (and can represent either tumour calcification or a granuloma engulfed by tumour) ▸ eccentric or stippled calcification within soft tissue may indicate malignancy
A mixture of soft tissue and ground-glass attenuation nodules is more likely to be malignant than soft tissue nodules alone
Size: this is of little diagnostic value ▸ most benign nodules are <2 cm ▸ round, flat or tubular nodules tend to be benign
Margins: a well-defined mass with a smooth pencil sharp margin is likely to be benign ▸ carcinomas typically have ill-defined margins which are irregular, spiculated, or lobulated and may exhibit umbilication or a notch – unfortunately all these features can be seen with benign disease
This is useful for nodules >1 cm ▸ a positive result is 97% sensitive and 82% specific for malignancy
False-positive result: this can be secondary to an infectious or inflammatory processes (e.g. TB, sarcoid or rheumatoid nodules)
False-negative result: this can occur if a nodule is <1 cm ▸ it can also occur if the nodule is due to a carcinoid tumour or a slow-growing adenocarcinoma subtype
| Nodule type | Management recommendations | Additional remarks |
|---|---|---|
| Solitary pure GGNs | ||
| ≤5 mm | No CT follow-up required | Obtain contiguous 1-mm-thick sections to confirm that nodule is truly a pure GGN |
| >5 mm | Initial follow-up CT at 3 months to confirm persistence then annual surveillance CT for a minimum of 3 years | FDG PET is of limited value, potentially misleading, and therefore not recommended |
| Solitary part-solid nodules | Initial follow-up CT at 3 months to confirm persistence. If persistent and solid component <5 mm, then yearly surveillance CT for a minimum of 3 years. If persistent and solid component ≥5 mm, then biopsy or surgical resection | Consider PET/CT for part-solid nodules >10 mm |
| Multiple subsolid nodules | ||
| Pure GGNs ≤5 mm | Obtain follow-up CT at 2 and 4 years | Consider alternate causes for multiple GGNs ≤5 mm |
| Pure GGNs >5 mm without a dominant lesion(s) | Initial follow-up CT at 3 months to confirm persistence and then annual surveillance CT for a minimum of 3 years | FDG PET is of limited value, potentially misleading, and therefore not recommended |
| Dominant nodule(s) with part-solid or solid component | Initial follow-up CT at 3 months to confirm persistence. If persistent, biopsy or surgical resection is recommended, especially for lesions with >5 mm solid component | Consider lung-sparing surgery for patients with dominant lesion(s) suspicious for lung cancer |
SCLC: small (oat) cell carcinoma
This originates from submucosal neuroendocrine cells ▸ it rapidly spreads haematogenously and to the lymph nodes ▸ it behaves as a systemic disease and is usually disseminated at presentation
NSCLC: non-small cell lung cancer
Squamous cell carcinomas: arises from the proximal airway epithelium
Large cell carcinomas: atypical cells that appear ‘large’ under the microscope
Adenocarcinomas: arising from the bronchial glands
Risks: tobacco smoke (20- to 30-fold increased risk) – has the greatest association with squamous cell carcinomas and weakest association with bronchoalveolar carcinomas ▸ asbestos exposure, interstitial pulmonary fibrosis and radiotherapy are additional risks
Asymptomatic (25%): asymptomatic peripheral tumours are more likely to be incidental findings and surgically resectable
Symptomatic: recurrent pneumonia ▸ cough ▸ wheeze ▸ haemoptysis
Paraneoplastic syndromes: inappropriate ADH secretion ▸ Cushing's syndrome (ACTH) ▸ carcinoid syndrome ▸ hypercalcaemia (PTH)
Poor prognostic features: hoarseness ▸ chest pain ▸ brachial plexus neuropathy or Horner's syndrome (due to a Pancoast's tumour) ▸ SVC obstruction ▸ dysphagia
The majority are spherical or oval in shape ▸ lobulated masses can occur due to uneven growth rates ▸ there may be a ‘corona radiata’ due to numerous fine strands radiating into the lung ▸ a bronchocele or mucoid impaction can be seen distal to an obstructing carcinoma ▸ collapse and consolidation is less commonly seen than with central tumours
Cavitation with irregular thick walls (≥8 mm) ± fluid levels (particularly squamous tumours)
Calcification is rare (6–10%) and may represent engulfed granulomatous disease ▸ amorphous or cloud-like calcification can be seen with dystrophic tumour calcification (10%)
Air bronchograms are rare but can be seen with adenocarcinoma
Ground-glass attenuation is associated with a higher risk of malignancy (and commonly seen with lepidic or invasive mucinous adenocarcinoma)
Collapse or consolidation can be seen peripheral to the tumour (due to bronchial narrowing ± hilar enlargement) ▸ any peripheral collapsed lung will enhance more than a central tumour ▸ collateral air drift may prevent some post-obstructive changes
‘ Golden S’ sign : a fissure may show a central bulge due to collapse around a central tumour
This is a common presenting feature and may reflect a proximal central tumour, lymphadenopathy or consolidated lung ▸ extensive hilar and mediastinal lymphadenopathy is typical of small cell tumours ▸ simple pneumonias rarely cause hilar adenopathy
This does not preclude surgical resection (although it adversely affects the prognosis)
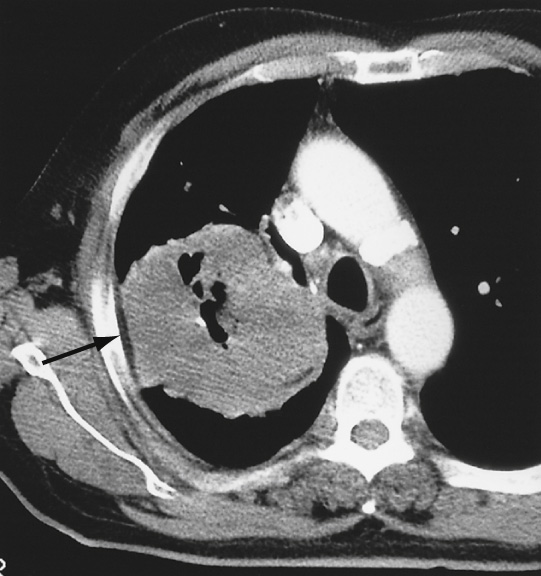
CT: assessment is unreliable (with local chest wall pain remaining the most specific indicator) ▸ contact with or a thickened pleura does not necessarily indicate invasion ▸ a clear extrapleural fat plane is helpful but not definitive ▸ reliable signs include clear-cut bone destruction or a large soft tissue mass
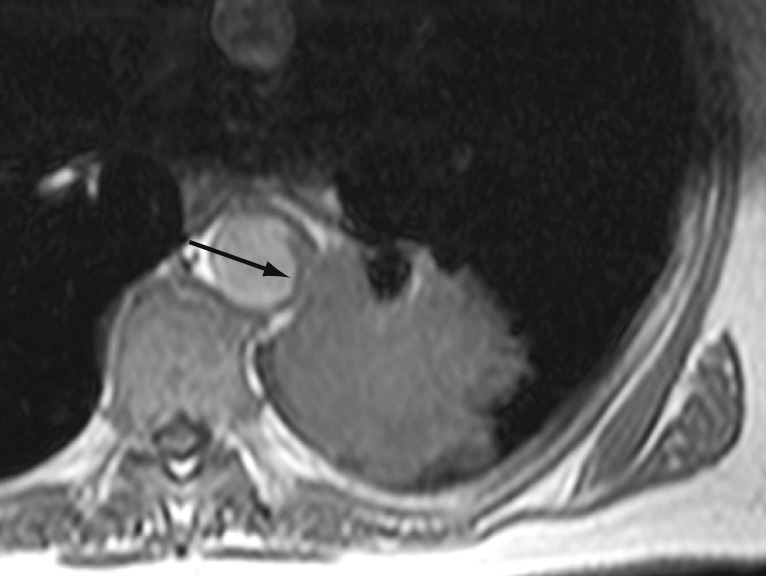
MRI: this is better than CT in selected cases ▸ it is the optimal modality for demonstrating the extent of a superior sulcus tumour
Transthoracic ultrasound: this is an accurate technique
99m Tc radionuclide skeletal scintigraphy: this is sensitive (and may detect bone invasion before plain radiography) ▸ PET-CT is now the technique of choice for distant spread
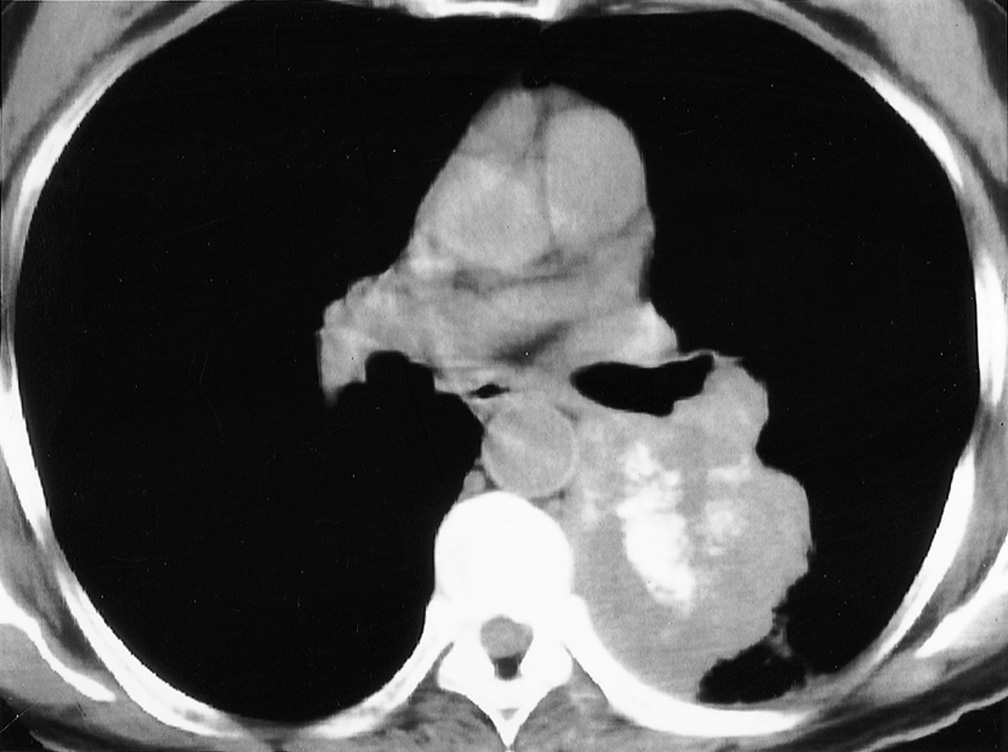
CT/MRI indicators: visible tumour deep within the mediastinal fat (mediastinal contact alone is not enough to diagnose invasion) ▸ encasement of the mediastinal vessels, oesophagus, or proximal mainstem bronchi ▸ SVC obstruction ▸ an elevated hemidiaphragm (indicating phrenic nerve involvement)
Criteria for resectability: < 3 cm of contact with the mediastinum ▸ < 90° of circumferential contact with the aorta ▸ a visible mediastinal fat plane between the mass and any vital mediastinal structure
Irresectability: tumours obliterating fat planes or showing greater contact than that described above (although less certain)
Direct rib or spine invasion ▸ haematogenous osteolytic bone metastases ▸ hypertrophic osteoarthopathy
| Premalignant | Malignant |
|---|---|
| Atypical adenomatous hyperplasia | Minimally invasive adenocarcinoma |
| Adenocarcinoma in situ | predominantly lepidic growth pattern |
| lepidic growth pattern (no solid components) | ≤3 cm |
| pure ground-glass opacity on CT | invasive component <5 mm |
| subsolid nodules on CT | |
| Lepidic predominant adenocarcinoma | |
| >5 mm invasion | |
| previously known as non-mucinous bronchoalveolar carcinoma | |
| Invasive mucinous adenocarcinoma | |
| previously known as mucinous bronchoalveolar carcinoma |
They may occur due to direct spread, lymphatic involvement, or tumour emboli (an adenocarcinoma may cause lobular pleural thickening indistinguishable from a malignant mesothelioma)
A pleural effusion designates a tumour as M1a (unless clinically there is another cause and the pleural cytology is negative)
This can appear as a solitary lobulated or spiculated pulmonary mass with air bronchograms and cavitation ▸ it may also appear as an ill-defined opacity resembling a pneumonia, homogeneous consolidation, multiple ill-defined nodules throughout the lung, or a focal ground-glass opacity
They are often PET negative
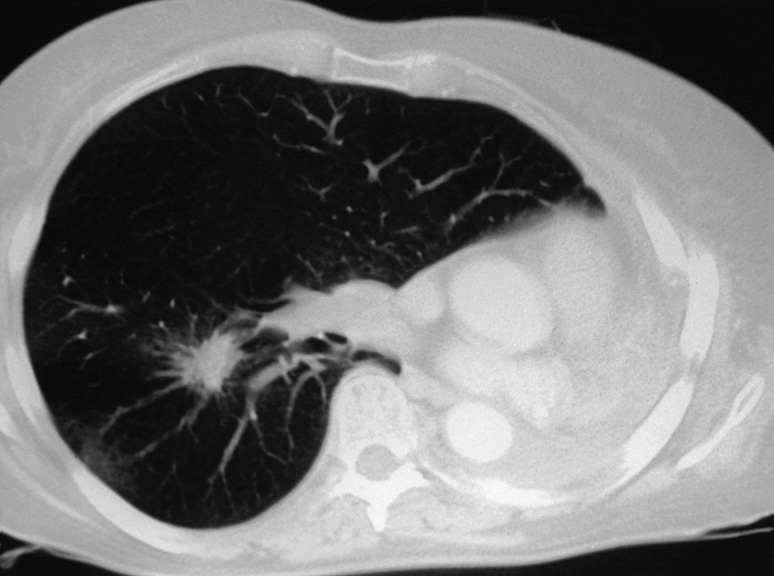
A Pancoast's tumour is an apical lung tumour which can extend into the chest wall and may resemble apical pleural thickening (and is commonly a squamous carcinoma) ▸ it can invade the subclavian vessels, brachial plexus (with pain radiating into the arm) and cervical sympathetic chain (leading to Horner's syndrome)
MRI is the optimal modality for assessment but CT is used for assessing bony involvement
Lung cancers normally spread sequentially to the ipsilateral hilar nodes, ipsilateral mediastinal nodes and then the contralateral mediastinal and supraclavicular nodes
Skip metastases to the mediastinal nodes (in the absence of hilar nodes) can occur in ![]() of patients
of patients
Other causes of enlarged nodes: previous tuberculosis ▸ histoplasmosis ▸ pneumoconiosis ▸ sarcoidosis ▸ reactive hyperplasia to the tumour or an associated pneumonia
This is insensitive
Generally nodes with a short-axis diameter of > 10 mm are considered enlarged (normal subcarinal and lower mediastinal nodes can be up to 10 mm – upper paratracheal nodes rarely exceed 7 mm) ▸ increased signal on STIR imaging in abnormal nodes ▸ however microscopic tumour involvement can be present in normal-sized nodes
This can assess nodal size and morphology, and guide fine needle aspiration of the aortopulmonary, subcarinal and posterior mediastinal nodes ▸ it has greater sensitivity and specificity than CT or PET in some series
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here