Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Infants and children are fasted before sedation and anesthesia to minimize the risk of pulmonary aspiration of gastric contents. In a fasted child, only the basal secretions of gastric juice should be present in the stomach. In 1948, Digby Leigh recommended a 1-hour preoperative fast after clear fluids. Subsequently, Mendelson reported a number of maternal deaths that were attributed to aspiration at induction of anesthesia. During the intervening 20 years, the fasting interval before elective surgery increased to 8 hours after all solids and fluids. In the late 1980s and early 1990s, an evidence-based approach to the effects of fasting intervals on gastric fluid pH and volume concluded that fasting more than 2 hours after clear fluids neither increased nor decreased the risk of pneumonitis should aspiration occur. In the past, the risk for pneumonitis was reported to be based on two parameters: gastric fluid volume greater than 0.4 mL/kg and pH less than 2.5; however, these data were never published in a peer-reviewed journal. In a monkey, 0.4 mL/kg of acid instilled endobronchially, equivalent to 0.8 mL/kg aspirated tracheally, resulted in pneumonitis. Using these corrected criteria for acute pneumonitis (gastric residual fluid volume >0.8 mL/kg and pH <2.5), studies in children demonstrated no additional risk for pneumonitis when children fast for only 2 hours after clear fluids.
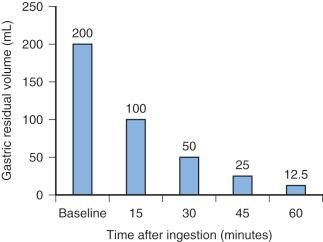
The incidence of pulmonary aspiration in modern routine elective pediatric or adult cases without known risk factors is small. This small risk is the result of a number of factors including the preoperative fasting schedule. The half-life to empty clear fluids from the stomach is approximately 15 minutes ( Fig. 4.1 ); as a result, 98% of clear fluids exit the stomach in children by 1 hour. Clear liquids include water, fruit juices without pulp, carbonated beverages, clear tea, and black coffee. Although fasting for 2 hours after clear fluids ensures nearly complete emptying of the residual volume, extending the fasting interval to 3 hours introduces flexibility in the operative schedule. The potential benefits of a 2-hour fasting interval after clear fluids include a reduced risk of hypoglycemia, which is a real possibility in children who are debilitated, have chronic disease, are poorly nourished, have metabolic dysfunction, or are preterm or formerly preterm infants. Additional benefits include decreased thirst, decreased hunger (and thus reduced temptation that the fasting child will “steal” another child's food), decreased risk for hypotension during induction, and improved child cooperation.
A scheduled operation on a preterm infant or neonate may occasionally be delayed, thus extending the period of fasting to a point that could be potentially dangerous (i.e., from hypoglycemia or hypovolemia). In this circumstance, the infants should be given glucose-containing intravenous (IV) maintenance fluids before induction of anesthesia. Alternatively, if the period may be protracted, the infant should be offered clear fluids orally until 2 hours before induction.
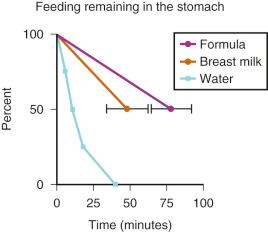
Breast milk, which can cause significant pulmonary injury if aspirated, has a very high and variable fat content (determined by maternal diet), which will delay gastric emptying. Breast milk should not be considered a clear liquid. Two studies estimated the gastric emptying times after clear fluids, breast milk, or formula in full-term and preterm neonates. The emptying times for breast milk in both age groups were substantively greater than for clear fluids, and the gastric emptying times for formula were even greater than those for breast milk. With half-life emptying times for breast milk of 50 minutes and for formula of 75 minutes, fasting intervals of at least 3.3 hours for breast milk and 5 hours for formula are required. More importantly, perhaps, was the large (15%) variability in gastric emptying times for breast milk and formula in full-term infants ( E-Fig. 4.1 ). Based in part on these data, the Task Force on Fasting of the American Society of Anesthesiologists (ASA) issued the following guidelines: for breast milk, 4 hours; and for formula, 6 hours ( Table 4.1 ).
| Clear liquids a | 2 hours |
| Breast milk | 4 hours |
| Infant formula | 6 hours b |
| Solids (fatty or fried foods) | 8 hours |
a Include only fluids without pulp, clear tea, or coffee without milk products.
b Some centers allow plain toast (no dairy products) up to 6 hours prior to induction.
Children who have been chewing gum must dispose of the gum by expectorating it, not swallowing it. Hansen and Rune reported a 70% increase in gastric fluid volume in the first 15 minutes after initiating gum chewing, almost all from swallowing saliva. Chewing gum also increases gastric pH in children, leaving no clear evidence that it affects the risk of pneumonitis should aspiration occur. A systemic review and meta-analysis of preoperative gum chewing in both children and adults concluded that chewing gum is unlikely to lead to a meaningful increase in the risk of major morbidity from pulmonary aspiration. Consequently, we recommend that if the gum is discarded, then elective anesthesia can proceed without additional delay. If, however, the child swallows the gum, then surgery should be canceled, because aspirated gum at body temperature may be very difficult to extract from a bronchus or trachea.
Children can never be trusted to fast. Therefore, anesthesiologists must always be suspicious and question children just before induction as to whether they have eaten or drunk anything (although the veracity of the answer may always be questioned as well). It is not unusual to find bubble gum, candy, or other food in a child's mouth. This is another reason to ask children to open their mouth fully and stick out their tongue during the preoperative examination of the airway.
When the anesthesiologist suspects that the child has a full stomach, induction of anesthesia should be adjusted appropriately. The incidence of pulmonary aspiration of gastric contents during elective surgery in children ranges from 1 : 1163 to 1 : 10,000, depending on the study. In contrast, the frequency of pulmonary aspiration in children undergoing emergency procedures is several times greater, 1 : 373 to 1 : 4544. Risk factors for perianesthetic aspiration included neurologic or esophagogastric abnormality, emergency surgery (especially at night), ASA physical status 3 to 5, intestinal obstruction, increased intracranial pressure, increased abdominal pressure, light anesthesia, obesity, and the skill and experience of the anesthesiologist.
The majority of aspirations in children occur during induction of anesthesia, with only 13% occurring during emergence and extubation. In contrast, 30% of the aspirations in adults occur during emergence. Bowel obstruction or ileus was present in the majority of infants and children who aspirated during the perioperative period in one study, with the risk increasing in children younger than 3 years of age. A combination of factors predisposes the infant and young child to regurgitation and aspiration, including decreased competence of the lower esophageal sphincter, excessive air swallowing while crying during the preinduction period, strenuous diaphragmatic breathing, and a shorter esophagus. In one study, almost all cases of pulmonary aspiration occurred either when the child gagged or coughed during airway manipulation or during induction of anesthesia when neuromuscular blocking drugs were not provided or before the child was completely paralyzed.
When children do aspirate, the morbidity and mortality are exceedingly small for elective surgical procedures and generally reflect their ASA physical status; most patients with clinical status of ASA 1 or 2 who aspirate clear gastric contents have minimal to no sequelae. If clinical signs of sequelae from an aspiration in a child are going to occur, they will be apparent within 2 hours ; mortality is exceedingly low and estimated to be between 0 and 1 : 50,000.
Body piercing is common practice in adolescents and young adults. Single or multiple piercings may appear anywhere on the body. To minimize the liability and risk of complications from metal piercings, they should be removed before surgery. Complications that may occur if they are left in situ during anesthesia are listed in E-Table 4.1 .
| Electrocautery Burns |
| All tissues adjacent to metal piercings are at risk; to avoid burns, use bipolar cautery or a harmonic knife; the cautery grounding pad should be remote from the piercing. |
| Airway Difficulty With Intubation |
| Laryngospasm and hypoxia |
| Pulmonary aspiration |
| Tissue Damage |
| Necrosis |
| Bleeding |
Unfortunately, cigarette smoking is not only limited to adults. Each day, 3800 American adolescents smoke their first cigarette and of these, more than 50% will become regular smokers. The annual burden of smoking-attributable mortality remains high with 5.6 million youth currently 0 to 17 years of age projected to die prematurely from a smoking-related illness. Even though the rate of new cigarette smokers in North America has declined over the past 2 decades, this has been offset with an increase in use of other nicotine products such as electronic cigarettes (e-cigarettes). Many factors, including lack of regulation at the federal level, “harmless water vapor” messaging, a strong social media presence with celebrity endorsements, and flavors targeting a young audience (“Cupcake,” “Alien Blood,” “Cherry Crush,” “Chocolate Treat,” etc.) play a significant role in influencing initiation of e-cigarette use in this age group. A national survey indicated that e-cigarette use was at an all-time high among high school students at 13.4%, which was more prevalent than conventional cigarette use at 9.2%. Youth e-cigarette users are more likely than nonusers to report initiation of conventional cigarettes within a year.
Smoking is known to increase blood carboxyhemoglobin concentrations, decrease ciliary function, decrease functional vital capacity (FVC) and the forced expiratory flow in midphase (FEF 25%-75% ), and increase sputum production. There is extensive evidence that smokers undergoing surgery are more likely to develop wound infections and postoperative respiratory complications. Although stopping smoking for 2 days decreases carboxyhemoglobin levels and shifts the oxyhemoglobin dissociation curve to the right, stopping for at least 6 to 8 weeks is necessary to reduce the rate of postoperative pulmonary complications.
Current harmful effects of e-cigarette use focus on nicotine exposure, the potential for these products to be a gateway to cigarette use, and the possibility of exposure to harmful flavoring chemicals such as diacetyl (2,3-butanedione). This chemical is used to give foods a buttery or creamy flavor and has been shown to cause acute-onset bronchiolitis obliterans. Data are lacking on long-term exposure and health effects of the use of e-cigarettes given the limited time span these products have been in the marketplace.
The perioperative period is the ideal time to abandon the smoking habit permanently, and anesthesiologists can perhaps play a more active role in facilitating this process. Physician communication with adolescents regarding smoking cessation has been shown to positively impact their attitudes, knowledge, intentions to smoke, and quitting behaviors. In summary, during the preoperative visit with adolescents, anesthesiologists should inquire about cigarette smoking and emphasize the need to stop the habit by offering measures to ameliorate the withdrawal (e.g., nicotine patch).
A national survey in the United States revealed the percentage of children 3 to 19 years of age without asthma exposed to environmental tobacco smoke (ETS) decreased from 57.3% to 44.2% from 1999 to 2010 while a higher percentage of children with asthma (54.0%) were exposed to ETS.
Studies have shown that children exposed to ETS are more likely to have asthma, otitis media, atopic eczema, hay fever, and dental caries. There is also an increased rate of lower respiratory tract illness in infants with ETS exposure. A recent systemic review and meta-analysis revealed a nearly 2-fold increase in risk of hospitalization for an asthma exacerbation in children with asthma and ETS in westernized countries. This is the first time this risk has been quantified and will enable health care providers to convey the harmful effects of ETS to parents about aggravation of asthma.
Several authors have demonstrated that ETS results in increased perioperative airway complications in children. In one study, urinary cotinine, the major metabolite of nicotine, was used as a surrogate marker of ETS. A strong association was found between passive inhalation of tobacco smoke and airway complications on induction and emergence from anesthesia. A prospective investigation in Australia provided further insight on the effect of the smoking habits of different family members; the risk for perioperative adverse respiratory events was higher when children were exposed to maternal or both parents smoking than when only the father smoked. Respiratory adverse events include laryngospasm, bronchospasm, airway obstruction, oxygen desaturation (<95%), severe or sustained cough, and stridor in the postanesthesia care unit (PACU).
The evidence for adverse perioperative events in children from ETS is clearly overwhelming. During the preoperative visit, the anesthesiologist should ascertain the child's exposure to ETS by asking parents or guardians about smoking within the household. This is an opportune time to educate parents and guardians about the dangers of ETS for their children.
The perioperative period is stressful and anxiety-provoking for the child and family; many parents express more concern about the risks of anesthesia than those of the surgery. The factors that influence the ability of the child and family to cope with the stress of surgery include family dynamics, the child's developmental and behavioral status and cultural biases, and our ability to explain away misperceptions and misinformation. Because of logistics and today's practice constraints, there is limited time to evaluate family dynamics and establish rapport. It is therefore vital for the anesthesiologist to interact directly with the child in a manner consistent with the child's level of development. A specific child-oriented approach by the anesthesiologist, surgeon, nurses, and hospital staff is required.
Although the preoperative evaluation and preparation of children are similar to those of adults from a physiologic standpoint, the psychological preparation of infants and children is very different (see also Chapter 3 ). Many hospitals have an open house or a brochure to describe the preoperative programs available to parents before the day of admission. However, printed material should not replace verbal communication with nursing and medical staff. Anesthesiologists are encouraged to participate in the design of these programs so that they accurately reflect the anesthetic practice of the institution. The preoperative anesthetic experience begins when parents are first informed that the child is to have surgery or a procedure that requires general anesthesia. Parental satisfaction correlates with the comfort of the environment and the trust established between the anesthesiologist, the child, and the parents. If parental presence during induction is deemed to be in the child's best interest, a parental educational program that describes what the parent can expect to happen if he or she accompanies the child to the operating room (OR) can significantly decrease parents' anxiety and increase their satisfaction. The greater the understanding and amount of information the parents have, the less anxious they will be, and this attitude, in turn, will be reflected in the child.
Informed consent should include a detailed description of what the family can anticipate and our role to protect the welfare of the child. Before surgery, the anesthetic risks should be discussed in clear terms but in a reassuring manner by describing the measures that will be taken to carefully and closely monitor the safety of the child. Mentioning specific details and the purpose of the various monitoring devices may help diminish the parents' anxiety by demonstrating to them that the child will be anesthetized with the utmost safety and care. A blood pressure cuff will “check the blood pressure,” an electrocardiographic monitor will “watch the heartbeat,” a stethoscope will help us “to continuously listen to the heart sounds,” a pulse oximeter will “measure the oxygen in the bloodstream,” a carbon dioxide analyzer will “monitor the breathing,” an anesthetic agent monitor will “accurately measure the level of anesthesia,” and an IV catheter will be placed “to administer fluid and medications as needed.” Children who are capable and their parents should be given ample opportunity to ask questions preoperatively. Finally, they should be assured that our “anesthetic prescription” will be designed specifically for their child's needs, taking into account the child's underlying medical conditions and the needs of surgery to ensure optimal conditions for surgery, the safety of the child, and analgesia.
It has been shown that parents desire comprehensive perioperative information, and that discussion of highly detailed anesthetic risk information does not increase parent's anxiety level. Inadequate preparation of children and their families may lead to a traumatic anesthetic induction and difficulty for both the child and the anesthesiologist, with the possibility of postoperative psychological disturbances. Numerous preoperative educational programs for children and adults have evolved to alleviate some of these fears and anxiety. They include preoperative tours of the ORs, educational videos, play therapy, magical distractions, puppet shows, anesthesia consultations, and child-life preparation. The timing of the preoperative preparation has been found to be an important determinant of whether the intervention will be effective. For example, children older than 6 years of age who participated in a preparation program more than 5 to 7 days before surgery were least anxious during separation from their parents, those who participated in no preoperative preparation were moderately anxious, and those who received the information 1 day before surgery were the most anxious. The predictors of anxiety correlated also with the child's baseline temperament and history of previous hospitalizations. Children of different ages vary in their response to the anesthetic experience (see also Chapter 3 ). Even more important may be the child's trait anxiety when confronted with a stressful medical procedure.
Understanding age-appropriate behavior in response to external situations is essential. Age-specific perioperative anxieties are outlined in E-Table 4.2 (see also Chapter 3 for further discussion of risk factors for preoperative anxiety). Special aspects of a child's perception of anesthesia should be anticipated; children often have the same fears as adults but are unable to articulate them. The reason and need for a surgical procedure should also be carefully explained to the child. It is important to reassure children that anesthesia is not the same as the usual nightly sleep, but rather a special sleep caused by the medicines we give during which they cannot be awakened and no matter what the surgeon does, they cannot feel pain. Many children fear the possibility that they will wake up in the middle of the anesthetic and during surgery. They should be reassured that they will awaken only after the surgery is completed.
| Age | Specific Type of Perioperative Anxiety |
|---|---|
| 0–6 months |
|
| 6 months–4 years |
|
| 4–8 years |
|
| 8 years–adolescence |
|
| Adolescence |
|
The words the anesthesiologist uses to describe to the child what can be anticipated must be carefully chosen, because children think concretely and tend to interpret the facts literally. Examples of this are presented by the following anecdotes:
Example 1: A 4-year-old child was informed that in the morning she would receive a “shot” that would “put her to sleep.” That night, a frantic call was received from the mother, describing a very upset child; the child thought she was going to be “put to sleep” like the veterinarian had permanently “put to sleep” her sick pet.
Example 2: A 5-year-old child admitted for elective inguinal herniorrhaphy received a heavy premedication and was deeply sedated on arrival in the OR. After discharge, the parents frequently discovered him wandering about the house at night. On questioning, the child stated that he was “protecting” his family. He stated: “I don't want anyone sneaking up on you and operating while you are sleeping.”
In the first example, the child's concrete thought processes misunderstood the anesthesiologist's choice of words. The second case represents a problem of communication: the child was never told he would have an operation.
The importance of proper psychological preparation for surgery should not be underestimated. Often, little has been explained to both patient and parents before the day of surgery. Anesthesiologists have a key role in defusing fear of the unknown if they understand a child's age-related perception of anesthesia and surgery (see Chapter 3 ). They can convey their understanding by presenting a calm and friendly face (smiling, looking at the child, and making eye contact), offering a warm introduction, touching the patient in a reassuring manner (holding a child's or parent's hand), and being completely honest. Children respond positively to an honest description of exactly what they can anticipate. This includes informing them of the slight discomfort of starting an IV line or giving an intramuscular premedication, the possible bitter taste of an oral premedication, or breathing our magic laughing gas through the flavored mask.
The postoperative process, from the OR to the recovery room, and the onset of postoperative pain should be described. Encourage the child and family to ask questions. Strategies to maintain analgesia should be discussed, including the use of long-acting local anesthetics; nerve blocks; neuraxial blocks; patient-controlled, nurse-controlled, or parent-controlled analgesia or epidural analgesia; or intermittent opioids (see also Chapter 42, Chapter 43, Chapter 44 ).
As children age, they become more aware of their bodies and may develop a fear of mutilation. Adolescents frequently appear quite independent and self-confident, but as a group, they have unique problems. In a moment their mood can change from an intelligent, mature adult to a very immature child who needs support and reassurance. Coping with a disability or illness is often very difficult for adolescents. Because they are often comparing their physical appearance with that of their peers, they may become especially anxious when they have a physical problem. In general, they want to know exactly what will transpire during the course of anesthesia. Adolescents are usually cooperative, preferring to be in control and unpremedicated preoperatively. The occasional overly anxious or rambunctious adolescent, however, may benefit from preanesthetic medication.
Monitoring the attitude and behavior of a child is very useful. A child who clings to the parents, avoids eye contact, and does not speak is very anxious. A self-assured, cocky child who “knows it all” may also be apprehensive or frightened. This know-it-all behavior may mask the child's true emotions, and he or she may decompensate just when cooperation is most needed. In some cases, nonpharmacologic supportive measures may be effective. In the extremely anxious child, supportive measures alone may be insufficient to reduce anxiety, and premedication is indicated.
Identifying a difficult parent or child preoperatively is not always easy, especially if the anesthesiologist first meets the child or family on the day of surgery and has limited time to assess the situation. Occasionally, we receive a warning regarding a difficult parent or child from the surgeon or nursing staff, based on their encounters with the family. With experience, some anesthesiologists are able to identify difficult parents and children during the short preoperative assessment and make appropriate adjustments to the anesthetic management plan.
The “veterans” or “frequent flyers” of anesthesia can also be difficult in the perioperative period. They have played the anesthesia and surgical game before and are not interested in participating again, especially if their previous experiences were negative. These children may benefit the most from a relatively heavy premedication; reviewing previous responses to premedication will aid in the adjustment of the current planned premedication (e.g., adding ketamine and atropine to oral midazolam to achieve a greater depth of sedation).
It is important to observe the family dynamics to better understand the child and determine who is in control, the parent or the child. Families many times are in a state of stress, particularly if the child has a chronic illness; these parents are often angry, guilt ridden, or simply exhausted. Ultimately, the manner in which a family copes with an illness largely determines how the child will cope. The well-organized, open, and communicative family tends to be supportive and resourceful, whereas the disorganized, noncommunicative, and dysfunctional family tends to be angry and frustrated. Dealing with a family and child from the latter category can be challenging. There is the occasional parent who is overbearing and demands total control of the situation. It is important to be empathetic and understanding but to set limits and clearly define the parent's role. He or she must be told that the anesthesiologist determines when the parent must leave the OR; this is particularly true if an unexpected development occurs during the induction.
One controversial area in pediatric anesthesia is parental presence during induction. Some anesthesiologists encourage parents to be present at induction, whereas others are uncomfortable with the process and do not allow parents to be present. Inviting a parent to accompany the child to the OR has been interpreted by some courts as an implicit contract on the part of the caregiver who invited the parent to participate in the child's care; in one case, the institution was found to have assumed responsibility for a mother who suffered an injury when she fainted. Each child and family must be evaluated individually; what is good for one child and family may not be good for the next. (See Chapter 3 for a full discussion of this and other anxiolytic strategies in children.)
If the practice of having parents present at induction is to work well, then the anesthesiologist must be comfortable with such an arrangement. No parent should ever be forced to be present for the induction of anesthesia, nor should any anesthesiologist be forced into a situation that compromises the quality of care he or she affords a child in need.
Parents must be informed about what to anticipate in terms of the OR itself (e.g., equipment, surgical devices), in terms of what they may observe during induction (e.g., eyes rolling back, laryngeal noises, anesthetic monitor alarms, excitation), and when they will be asked to leave. They must also be instructed regarding their ability to assist during the induction process, such as by comforting the child, encouraging the child to trust the anesthesiologist, distracting the child, and consoling the child ( and ). Personnel should be immediately available to escort parents back to the waiting area at the appropriate time. Someone should also be available to care for a parent who wishes to leave the induction area or who becomes lightheaded or faints. An anesthesiologist's anxiety about parents' presence during induction decreases significantly with experience.
Explaining what parents might see or hear is essential. We generally tell parents the following:
As you see your child fall asleep today, there are several things you might observe that you are not used to seeing. First, when anyone falls asleep, the eyes roll up, but since we are sleeping we do not generally see it. You may see your child do that today, and I do not want you to be frightened by that—it is expected and normal. The second thing is that as children go to sleep from the anesthesia medications, the tone of the structures in the neck decreases, so that some children will begin to snore or make vibrating noises. Again, I do not want you to be frightened or think that something is wrong. We expect this, and it is normal. The third thing you might see is what we call “excitement.” As the brain begins to go to sleep, it can actually get excited first. About 30 to 60 seconds after breathing the anesthesia medications, your child might suddenly look around or suddenly move his or her arms and legs. To you it appears that he or she is awakening from anesthesia or that he or she is upset. In reality, this is a good sign, because it indicates to us that your child is falling asleep and that 15 to 30 seconds later he or she will be completely anesthetized. Also, you should know that even though your child appears to be awake to you, in reality he or she will not remember any of that. As soon as your child loses consciousness, we will ask you to give your child a kiss and step out of the operating room.
This kind of careful preparation provides to the parents the confidence that the anesthesiologist really knows what he or she is talking about, and it avoids frightening the parents. In general, the more information provided, the lower the parental anxiety levels. A child-life specialist in surgical services may also prove valuable to the anesthesiologist by calming and preparing both children and parents for the OR experience (see “ Inhalation Induction ” later in this chapter for further details).
Occasionally, the best efforts to relieve a child's anxiety by parental presence or administration of a sedative premedication (or both) are not successful, and an anticipated smooth induction may not go as planned. There are three options that may be used depending on the age of the child: (1) renegotiating (which is seldom successful), (2) holding the mask farther away from the child's face, or (3) suggesting an IV or intramuscular induction. If an intramuscular shot or IV induction is proposed, the child will usually choose the mask. If the situation is totally out of control, either elective surgery can be rescheduled or intramuscular ketamine can be used if the parents choose to proceed. These situations are particularly difficult for the parents and the caregivers but must be handled on an individual basis.
The medical history of a child obtained during the preanesthetic visit allows the anesthesiologist to determine whether the child is optimized for the planned surgery, to anticipate potential problems due to coexisting disease, to determine whether appropriate laboratory or other tests are available or needed, to select optimal premedication, to formulate the appropriate anesthetic plan including perioperative monitoring, and to anticipate postoperative concerns including pain management and postoperative ventilatory needs. The history of the present illness is described to the physicians by the parents and verified by the referring or consultant surgeon's notes. If the child is old enough, it is helpful to obtain the child's input. The history should focus on the following aspects:
A review of all organ systems ( Table 4.2 ) with special emphasis on the organ system involved in the surgery
| System | Factors to Assess | Possible Anesthetic Implications |
|---|---|---|
| Respiratory | Cough, asthma, recent cold | Irritable airway, bronchospasm, medication history, atelectasis, infiltrate |
| Croup | Subglottic narrowing | |
| Apnea/bradycardia | Postoperative apnea/bradycardia | |
| Cardiovascular | Murmur | Septal defect, avoid air bubbles in IV line |
| Cyanosis | Right-to-left shunt | |
| History of squatting | Tetralogy of Fallot | |
| Hypertension | Coarctation, renal disease | |
| Rheumatic fever | Valvular heart disease | |
| Exercise intolerance | Congestive heart failure, cyanosis | |
| Neurologic | Seizures | Medications, metabolic derangement |
| Head trauma | Intracranial hypertension | |
| Swallowing incoordination | Aspiration, esophageal reflux, hiatus hernia | |
| Neuromuscular disease | Neuromuscular relaxant drug sensitivity, malignant hyperpyrexia | |
| Gastrointestinal/hepatic | Vomiting, diarrhea | Electrolyte imbalance, dehydration, full stomach |
| Malabsorption | Anemia | |
| Black stools | Anemia, hypovolemia | |
| Reflux | Possible need for full-stomach precautions | |
| Jaundice | Drug metabolism/hypoglycemia | |
| Genitourinary | Frequency | Urinary tract infection, diabetes, hypercalcemia |
| Time of last urination | State of hydration | |
| Frequent urinary tract infections | Evaluate renal function | |
| Endocrine/metabolic | Abnormal development | Endocrinopathy, hypothyroid, diabetes |
| Hypoglycemia, steroid therapy | Hypoglycemia, adrenal insufficiency | |
| Hematologic | Anemia | Need for transfusion |
| Bruising, excessive bleeding | Coagulopathy, thrombocytopenia, thrombocytopathy | |
| Sickle cell disease | Hydration, possible transfusion | |
| Allergies | Medications | Possible drug interaction |
| Dental | Loose or carious teeth | Aspiration of loose teeth, bacterial endocarditis prophylaxis |
A review of patient and parental smoking history
Medications (over-the-counter and prescribed) related to and taken before the present illness, including herbals and vitamins, and when the last dose was taken
Medication allergies with specific details of the nature of the allergy and whether immunologic testing was performed
Previous surgical and hospital experiences, including those related to the current problem
Timing of the last oral intake, last urination (wet diaper), and vomiting and diarrhea. It is essential to recognize that decreased gastrointestinal motility often occurs with an illness or injury.
In the case of a neonate, problems that may have been present during gestation and birth may still be relevant in the neonatal period and beyond ( E-Table 4.3 ). The maternal medical and pharmacologic history (both therapeutic and drug abuse) may also provide valuable information for the management of a neonate requiring surgery.
| Maternal History | Commonly Expected Problems With Neonate |
|---|---|
| Rh-ABO incompatibility | Hemolytic anemia, hyperbilirubinemia, kernicterus |
| Toxemia | SGA and its associated problems, a muscle relaxant interaction with magnesium therapy |
| Hypertension | SGA and its associated problems a |
| Drug addiction | Withdrawal, SGA |
| Infection | Sepsis, thrombocytopenia, viral infection |
| Hemorrhage | Anemia, shock |
| Diabetes | Hypoglycemia, birth trauma, LGA, SGA, and associated problems a |
| Polyhydramnios | Tracheoesophageal fistula, anencephaly, multiple anomalies |
| Oligohydramnios | Renal hypoplasia, pulmonary hypoplasia |
| Cephalopelvic disproportion | Birth trauma, hyperbilirubinemia, fractures |
| Alcoholism | Hypoglycemia, congenital malformation, fetal alcohol syndrome, SGA, and associated problems a |
a See Table 2.2 .
The past medical history should include a history of all past medical illnesses with a review of organ systems, previous hospitalizations (medical or surgical), childhood syndromes with associated anomalies, medication list, herbal remedies, and any allergies, especially to antibiotics and latex. Whether the child was full-term or preterm at birth should be discerned; if preterm, any associated problems should be noted, including admission to a neonatal intensive care unit, duration of tracheal intubation, history of apnea or bradycardia (including oxygen treatment, home apnea monitor, intraventricular hemorrhage), and congenital defects.
Examination of previous surgical and anesthesia records greatly assists in planning the anesthesia. Particular attention should be paid to any difficulties encountered with airway management, venous access, or emergence. The response to or need for premedication and the route of administration used should be noted.
The use of herbal medicinal products has become increasingly popular, likely driven by the notion that “natural” substances have fewer side effects. A survey in five geographically diverse centers in the United States found that 3.5% of pediatric surgical patients had been given herbal supplements or homeopathic remedies 2 weeks prior to surgery. The findings of the National Health Interview Survey confirm a similar prevalence rate in which natural product usage among children age 0 to 17 years amounts to 3.9%. Herbal medicine use is more common in adults; 32% of adult surgical patients take one or more herb-related compounds. Nearly 70% of adults failed to disclose their use of herbal remedies when asked about medications during routine perioperative assessment. Herbal medicines are regulated as food supplements under the Dietary Supplement Health and Education Act of 1994 and as such manufacturers are not required to demonstrate safety or efficacy before placing a product on the market. Without the Food and Drug Administration (FDA) regulation, there are no quality assurance requirements for manufacturing and labeling and much variation can occur in each preparation. Anesthesiologists should include specific inquiries regarding the use of these medications because of the potential for adverse effects and drug interactions.
Herbal medicines are associated with cardiovascular instability, coagulation disturbances, potentiation of sedation, and immunosuppression. The most commonly used herbal medications reported are garlic, ginseng, Ginkgo biloba , St. John's wort, and Echinacea, with Echinacea and other herbal medicines for the treatment of coughs and colds taking the lead in the pediatric population. The three “g” herbals, together with feverfew (Tanacetum parthenium), potentially increase the risk of bleeding during surgery. The amount of active ingredient in each preparation and the dose taken may vary, thus making detection of a change in platelet function and other subtle coagulation disturbances difficult. St. John's wort is the herb that most commonly interacts with anesthetics and other medications, usually via a change in drug metabolism, because of potent inducing effects on the cytochrome P-450 enzymes (e.g., CYP3A4) and P-glycoprotein. A potentially fatal interaction between cyclosporine and St. John's wort has been well documented. Heart, kidney, or liver transplant recipients who were stabilized on a dose of cyclosporine experienced decreased plasma concentrations of cyclosporine and, in some cases, acute rejection episodes after taking St. John's wort. A summary of the most commonly used herbal remedies and their potential perioperative complications is shown in E-Table 4.4 .
| Name of Herb | Common Uses | Pharmacologic Effects | Potential Perioperative Complications |
|---|---|---|---|
| Echinacea, purple coneflower root | Prophylaxis and treatment of viral, bacterial, and fungal infection | Stimulation of the immune system. With long-term use may be immunosuppressive. | Reduced effectiveness of immunosuppressants. Potential for wound infection. May cause hepatotoxicity. |
| Ephedra, ma huang | Diet aid | Indirect- and direct-acting sympathomimetic | Dose-dependent increase in heart rate and blood pressure with potential for perioperative myocardial infarction and stroke. Arrhythmias with halothane. Tachyphylaxis with intraoperative ephedrine. |
| Garlic, ajo | Antihypertensive, lipid-lowering agent, anti-thrombus forming | Inhibits platelet aggregation (partially irreversibly) in a dose-dependent manner. Lowers serum lipid and cholesterol levels. | May potentiate other platelet inhibitors. Concerns for perioperative bleeding. |
| Ginkgo, maidenhair; fossil tree | Circulatory stimulant. Used to treat Alzheimer disease, peripheral vascular disease, and erectile dysfunction. | Inhibits platelet-activating factor. Antioxidant. Modulates neurotransmitter activity. | Concerns for perioperative bleeding. May potentiate other platelet inhibitors. |
| Ginseng | Used to protect the body against stress and restore homeostasis | Poorly understood. Inhibits platelet aggregation (partly irreversibly). | Potential to increase perioperative bleeding. Potential for hypoglycemia. |
| Kava, ava pepper | Anxiolytic | Possible potentiation of γ-aminobutyric acid (GABA) transmission | Potentiates sedative effects of anesthetic agents. Possible withdrawal syndrome after sudden abstinence. Kava-induced hepatotoxicity. |
| St. John's wort, goat weed, amber, hard hay | Treatment of depression and anxiety | Central inhibition of serotonin, norepinephrine, and dopamine. Induction of cytochrome P-450 3A4. | Decreased effectiveness of drugs metabolized by CYP3A4 such as cyclosporine, alfentanil, midazolam, lidocaine, calcium-channel blockers, and digoxin |
| Valerian, vandal root, all heal, Jacob's ladder | Anxiolytic and sleep aid | Potentiation of GABA neurotransmission | Potentiates sedative effects of anesthetic agents. Withdrawal-type syndrome with sudden abstinence. |
To avoid potential perioperative complications, the ASA has encouraged the discontinuation of all herbal medicines 2 weeks before surgery, although this recommendation is not evidence based. Recognizing that this is not always feasible, the ASA further recommends that anesthesiologists have knowledge of herbal medications and their potential interactions. Each herb should be carefully evaluated using standard resource texts, and a decision should be made regarding the timing of or need for discontinuation as determined on a case-by-case basis.
Children may present for surgery after having been recently immunized. The anesthesiologist and surgeon must then consider (1) whether the immunomodulatory effects of anesthesia and surgery might affect the efficacy and safety of the vaccine and (2) whether the inflammatory responses to the vaccine will alter the perioperative course.
What do anesthesiologists think about anesthesia and vaccination? An international survey revealed that only one-third of responding anesthesiologists had the benefit of a hospital policy, ranging from a formal decision to delay surgery to an independent choice by the anesthesiologist. Sixty percent of respondents would anesthetize a child for elective surgery within 1 week of receiving a live attenuated vaccine (such as oral polio vaccine or measles, mumps, and rubella [MMR] vaccine), whereas 40% would not. The survey also revealed that 28% of anesthesiologists would delay immunization for 2 to 30 days after surgery.
A scientific review of the literature associating anesthesia and vaccination in children resulted in recommendations for the care of children under these circumstances. The review demonstrated a brief and reversible influence of vaccination on lymphoproliferative responses that generally returned to preoperative values within 2 days. Vaccine-driven adverse events (e.g., fever, pain, irritability) might occur but should not be confused with perioperative complications. Adverse events to inactivated vaccines such as diphtheria-tetanus-pertussis (DPT) become apparent from 2 days and to live attenuated vaccines such as MMR from 7 to 21 days after immunization. Therefore appropriate delays between immunization and anesthesia are recommended by type of vaccine to avoid misinterpretation of vaccine-associated adverse events as perioperative complications. Because children remain at risk of contracting vaccine-preventable diseases, the minimum delay seems prudent, especially in the first year of life. Likewise, it seems reasonable to delay vaccination after surgery until the child is fully recovered. These recommendations were adopted in a consensus guideline by the Association of Paediatric Anaesthetists of Great Britain and Ireland, *
* See http://www.apagbi.org.uk/sites/default/files/images/Final%20Immunisation%20apa.pdf (accessed March 27, 2016).
though to date, the US Centers for Disease Control and Prevention does not have a policy regarding the timing of vaccinations and surgery. Other immunocompromised patients, such as human immunodeficiency virus (HIV)-positive children, cancer patients, and transplant recipients, have distinct underlying immune impairments, and the influence of anesthesia on vaccine responses has not been comprehensively investigated.
The details pertaining to all allergies to medications and materials should be described in the child's record. These include the age of onset, frequency, severity, investigations, and treatments. The vast majority of reported allergies on children's charts are either nonimmunologic reactions or known (or unknown) drug adverse effects. The most common medication- and hospital-related allergies in children are penicillin and latex allergy.
Most cases of reported penicillin allergy consist of a maculopapular rash after oral penicillin. This occurs in 1% to 4% of children receiving penicillin or in 3% to 7% of those taking ampicillin, usually during treatment. Rarely are signs or symptoms that suggest an acute (immunoglobulin E–mediated) allergic reaction present (i.e., angioedema), and even less frequently is skin testing conducted to establish penicillin allergy. Given the frequency of penicillin allergy, most of these unverified allergies in fact are not allergies to penicillin but rather minor allergies to the dye in the liquid vehicle or a consequence of the (viral) infection. If the child has not received penicillin for at least 5 years since the initial exposure and has not been diagnosed with a penicillin allergy by an immunologist or allergist, then a reexposure is warranted. If the child has been tested immunologically for penicillin allergy, then it is best to avoid this class of antibiotics. Although there is a 5% to 10% cross-reactivity between first-generation cephalosporins and penicillin, there is no similar cross-reactivity with second- and third-generation cephalosporins. To date, there have been no fatal anaphylactic reactions in penicillin-allergic children from a cephalosporin.
Latex allergy is an acquired immunologic sensitivity resulting from repeated exposure to latex, usually on mucous membranes (e.g., children with spina bifida or congenital urologic abnormalities who have undergone repeated bladder catheterizations with latex catheters, those with more than four surgeries, those requiring home ventilation). It occurs more frequently in atopic individuals and in those with certain fruit and vegetable allergies (e.g., banana, chestnut, avocado, kiwi, pineapple). For a diagnosis of latex anaphylaxis, the child should have personally experienced an anaphylactic reaction to latex, skin-tested positive for anaphylaxis to latex, or experienced swelling of the lips after touching a toy balloon to the lips or a swollen tongue after a dentist inserted a rubber dam into the mouth. The avoidance of latex within the hospital will prevent acute anaphylactic reactions to latex in children who are at risk. Latex gloves and other latex-containing products should be removed from the immediate vicinity of the child. Prophylactic therapy with histamine H 1 - and H 2 -receptor antagonists and steroids do not prevent latex anaphylaxis. Latex anaphylaxis should be treated by removal of the source of latex, administration of 100% oxygen, acute volume loading with balanced salt solution (10 to 20 mL/kg repeated until the systolic blood pressure stabilizes), and administration of IV epinephrine (1 to 10 µg/kg according to the severity of the anaphylaxis). In some severe reactions, a continuous infusion of epinephrine alone (0.01 to 0.2 µg/kg per minute) or combined with other vasoactive medications may be required for several hours.
It is important to inquire about a family history, particularly focusing on a number of conditions, including malignant hyperthermia, muscular dystrophy, prolonged paralysis associated with anesthesia (pseudocholinesterase deficiency), sickle cell disease, bleeding (and bruising) tendencies, and drug addiction (drug withdrawal, HIV infection). The precise relationship to the proband must be documented.
The laboratory data obtained preoperatively should be appropriate to the history, illness, and surgical procedure. Routine hemoglobin testing or urinalysis is not indicated for most elective procedures; the value of these tests is questionable when the surgical procedure will not involve clinically important blood loss. There are insufficient data in the literature to make strict hemoglobin testing recommendations in healthy children. A preoperative hemoglobin value is usually determined only for those who will undergo procedures with the potential for blood loss, those with specific risk factors for a hemoglobinopathy, former preterm infants, and those younger than 6 months of age. Coagulation studies (platelet count, international normalized ratio [INR], and partial thromboplastin time [PTT]) may be indicated if major reconstructive surgery is contemplated, especially if warranted by the medical history, and in some centers before tonsillectomy. In addition, collection of a preoperative type-and-screen or type-and-crossmatch sample is indicated in preparation for potential blood transfusions depending on the nature of the planned surgery and the anticipated blood loss.
In general, routine chest radiography is not necessary; studies have confirmed that routine chest radiographs are not cost-effective in children. The oxygen saturation of children who are breathing room air is very helpful. Baseline saturations of 95% or less suggest clinically important pulmonary or cardiac compromise and warrant further investigation.
Selective preoperative laboratory tests, such as electrolyte and blood glucose determinations, renal function tests, blood gas analysis, blood concentrations of seizure medication and digoxin, electrocardiography, echocardiography, liver function tests, computed tomography (CT), magnetic resonance imaging (MRI), or pulmonary function tests, should be performed when appropriate. These tests may be ordered after consideration of specific information obtained from sources such as medical records, patient interview, physical examination, and the type and invasiveness of the planned procedure and anesthesia.
Although pregnancy rates among teenagers in the United States are declining, a small percentage of adolescents may still present for elective surgery with an unsuspected pregnancy. Birth rates for teenagers in the United States declined to historic lows in 2014, to 24.2 births/1000 females aged 15–19 compared with 2003 when the birth rate in that age group was 41.6 births/1000. The birth rate for girls aged 10 to 14 years also declined from 0.6/1000 in 2003 to 0.3/1000 in 2014. However, routine preoperative pregnancy testing in adolescent girls may present ethical and legal dilemmas, including social and confidentiality concerns. This places the anesthesiologist in a predicament when faced with a question of whether to perform routine preoperative pregnancy screening. Each hospital should adopt a policy regarding pregnancy testing to provide a consistent and comprehensive policy for all females who have reached menarche.
A survey of members of the Society for Pediatric Anesthesia practicing in North America revealed that pregnancy testing was routinely required by approximately 45% of the respondents regardless of the practice setting (teaching versus nonteaching facilities). A retrospective review of a 2-year study of mandatory pregnancy testing in 412 adolescent surgical patients revealed that the overall incidence of positive tests was 1.2%. Five of 207 patients aged 15 years and older tested positive, for an incidence of 2.4% in that age group. None of the 205 patients younger than the age of 15 years had a positive pregnancy test. A prospective study of 261 menarcheal patients 10 to 34 years of age revealed 3 pregnancies but none of 107 children <15 years.
The most recent ASA Task Force on Preanesthesia Evaluation recognized that a history and physical examination may not adequately identify early pregnancy and issued the following statement: “The literature is insufficient to inform patients or physicians on whether anesthesia causes harmful effects on early pregnancy. Pregnancy testing may be offered to female patients of childbearing age and for whom the result would alter the patient's management.” Because of the risk of exposing the fetus to potential teratogens and radiation from anesthesia and surgery, the risk of spontaneous abortion, and the risk of apoptosis reported in the rapidly developing fetal animal brain (see Chapter 25 ), elective surgery with general anesthesia is not advised during early pregnancy. Therefore, if the situation is unclear, and when indicated by medical history, it is best to perform a preoperative pregnancy test. If the surgery is required in a patient who might be pregnant, then using an opioid-based anesthetic such as remifentanil and the lowest concentration of inhalational agent or propofol that provides adequate anesthesia is preferred.
The major objectives of preanesthetic medication are to (1) allay anxiety, (2) block autonomic (vagal) reflexes, (3) reduce airway secretions, (4) produce amnesia, (5) provide prophylaxis against pulmonary aspiration of gastric contents, (6) facilitate the induction of anesthesia, and (7) if necessary, provide analgesia. Premedication may also decrease the stress response to anesthesia and prevent cardiac arrhythmias. The goal of premedication for each child must be individualized. Light sedation, even though it may not eliminate anxiety, may adequately calm a child so that the induction of anesthesia will be smooth and a pleasant experience. In contrast, heavy sedation may be needed for the very anxious child who is unwilling to separate from his or her parents.
Factors to consider when selecting a drug or a combination of drugs for premedication include the child's age, ideal body weight, drug history, and allergic status; underlying medical or surgical conditions and how they might affect the response to premedication or how the premedication might alter anesthetic induction; parent and child expectations; and the child's emotional maturity, personality, anxiety level, cooperation, and physiologic and psychological status. The anesthesiologist should also consider the proposed surgical procedure and the attitudes and wishes of the child and the parents.
The route of administration of premedicant drugs is very important. Premedications have been administered by many routes, including the oral, nasal, rectal, buccal, IV, and intramuscular routes. Although a drug may be more effective and have a more reliable onset when given intranasally or intramuscularly, most pediatric anesthesiologists refrain from administering parenteral medication to children without IV access. Many children who are able to verbalize report that receiving a needle puncture was their worst experience in the hospital. In most cases, medication administered without a needle will be more pleasant for children, their parents, and the medical staff. Oral premedications do not increase the risk of aspiration pneumonia unless large volumes of fluids are ingested. In general, the route of delivery of the premedication should depend on the drug, the desired drug effect, and the psychological impact of the route of administration. For example, a small dose of oral medication may be sufficient for a relatively calm child, whereas an intramuscular injection (e.g., ketamine) may be best for an uncooperative, combative, extremely anxious child. Intramuscular administration may be less traumatic for this type of child than forcing him or her to swallow a drug, giving a drug rectally, or forcefully holding an anesthesia mask on the face.
Since Waters' classic work in 1938 on premedication of children, numerous reports have addressed this subject. Despite the wealth of studies, no single drug or combination of drugs has been found to be ideal for all children. Many drugs used for premedication have similar effects, and a specific drug may have various effects in different children or in the same child under different conditions.
Several categories of drugs are available for premedicating children before anesthesia ( Table 4.3 ). Selection of drugs for premedication depends on the goal desired. Drug effects should be weighed against potential side effects, and drug interactions should be considered. Premedicant drugs include tranquilizers, benzodiazepines, barbiturates, nonbarbiturate sedatives, opioids, ketamine, α2 agonists, and drugs that increase gastric motility.
| Drug | Route | Dose (mg/kg) |
|---|---|---|
| Barbiturates | ||
| Methohexital | Rectal | (10% solution) 20–40 |
| Intramuscular | (5% solution) 10 | |
| Thiopental | Rectal | (10% solution) 20–40 |
| Benzodiazepines | ||
| Diazepam | Oral | 0.1–0.5 |
| Rectal | 1 | |
| Midazolam | Oral | 0.25–0.75 |
| Nasal | 0.2 | |
| Rectal | 0.5–1 | |
| Intramuscular | 0.1–0.15 | |
| Lorazepam | Oral | 0.025–0.05 |
| Phencyclidine | ||
| Ketamine a | Oral | 3–6 |
| Nasal | 3 | |
| Rectal | 6–10 | |
| Intramuscular | 2–10 | |
| α 2 -Adrenergic Agonist | ||
| Clonidine | Oral | 0.004 |
| Opioids | ||
| Morphine | Intramuscular | 0.1–0.2 |
| Meperidine b | Intramuscular | 1–2 |
| Fentanyl | Oral | 0.010–0.015 (10–15 µg/kg) |
| Nasal | 0.001–0.002 (1–2 µg/kg) | |
| Sufentanil | Nasal | 0.001–0.003 (1–3 µg/kg) |
b Only a single dose is recommended due to metabolites that may cause seizures.
The major effect of tranquilizers is to allay anxiety, but they also have the potential to produce sedation. This group of drugs includes the benzodiazepines, phenothiazines, and butyrophenones. Benzodiazepines are widely used in children, whereas phenothiazines and butyrophenones are infrequently used.
Benzodiazepines calm children, allay anxiety, and diminish recall of perianesthetic events. At low doses, minimal drowsiness and cardiovascular or respiratory depression are produced.
Midazolam, a short-acting, water-soluble benzodiazepine with an elimination half-life of approximately 2 hours, is the most widely used premedication for children. The major advantage of midazolam over other drugs in its class is its rapid uptake and elimination. It can be administered intravenously, intramuscularly, nasally, orally, and rectally with minimal irritation, although it leaves a bitter taste in the mouth or nasopharynx after oral or nasal administration. Most children are adequately sedated after receiving a midazolam dose of 0.025 to 0.1 mg/kg intravenously, 0.1 to 0.2 mg/kg intramuscularly, 0.25 to 0.75 mg/kg orally, 0.2 mg/kg nasally, or 1 mg/kg rectally.
Orally administered midazolam is effective in calming most children and does not increase gastric pH or residual volume. Evidence suggests that the required dose of midazolam increases as age decreases in children, similar to that for inhaled agents and IV agents. An increased clearance in younger children contributes to their increased dose requirement. A number of medications that affect the cytochrome oxidase system significantly affect the first-pass metabolism of midazolam, including grapefruit juice, erythromycin, protease inhibitors, and calcium-channel blockers that decrease CYP3A4 activity, which in turn increases the blood concentration of midazolam and prolongs sedation. Conversely, anticonvulsants (phenytoin and carbamazepine), rifampin, St. John's wort, glucocorticoids, and barbiturates induce the CYP3A4 isoenzyme, thereby reducing the blood concentration of midazolam and its duration of action. The dose of oral midazolam should be adjusted in children who are taking these medications.
Concerns have been raised about possible delayed discharge after premedication with oral midazolam. Oral midazolam, 0.5 mg/kg, administered to children 1 to 10 years of age, did not affect awakening times, time to extubation, postanesthesia care unit, or hospital discharge times, after sevoflurane anesthesia. Similar results have been reported in children and adolescents after 20 mg of oral midazolam; however, detectable preoperative sedation in this group of children was predictive of delayed emergence. In children aged 1 to 3 years undergoing adenoidectomy as outpatients, premedication with oral midazolam, 0.5 mg/kg, slightly delayed spontaneous eye opening by 4 minutes and discharge by 10 minutes compared with placebo; children who had been premedicated, however, exhibited a more peaceful sleep at home on the night after surgery.
Likely the greatest effect of oral midazolam on recovery occurs with its use in children undergoing myringotomy and tube insertion, a procedure that normally takes 5 to 7 minutes. After oral midazolam premedication (0.5 mg/kg), induction of anesthesia with propofol, and maintenance with sevoflurane, emergence and early recovery were delayed by 6 and 14 minutes, respectively, in children 1 to 3 years of age compared with unpremedicated children, although discharge times did not differ. Increased postoperative sedation may be attributed to synergism between propofol and midazolam on γ-aminobutyric acid (GABA) receptors.
Although anxiolysis and a mild degree of sedation occur in most children after midazolam, a few develop undesirable adverse effects. Some children become agitated after oral midazolam. If this occurs after IV midazolam (0.1 mg/kg), IV ketamine (0.5 mg/kg) may reverse the agitation.
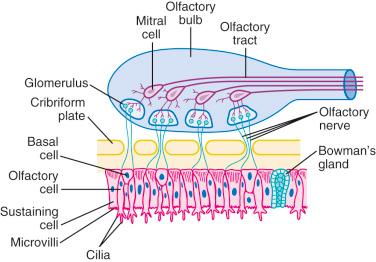
Anxiolysis and sedation usually occur within 10 minutes after intranasal midazolam ; nasal administration is not well accepted because it produces irritation, discomfort, and a burning aftertaste. Another theoretical concern for the nasal route of administration of midazolam is its potential to cause neurotoxicity via the cribriform plate. There are direct connections between the nasal mucosa and the central nervous system (CNS) ( E-Fig. 4.2 ). Medications administered nasally reach high concentrations in the cerebrospinal fluid very quickly. To date, no such sequelae have been reported. Because midazolam with preservative has been shown to cause neurotoxicity in animals, we recommend only preservative-free midazolam for nasal administration.
Sublingual midazolam (0.2 mg/kg) has been reported to be as effective as, and better accepted than, intranasal midazolam. Oral transmucosal midazolam given in three to five small allotments (0.2 mg/kg total dose) placed on a child's tongue (8 months to 6 years of age) was found to provide satisfactory acceptance and separation from parents in 95% of children.
Diazepam is used only for premedication of older children. In infants and especially preterm neonates, the elimination half-life of diazepam is markedly prolonged because of immature hepatic function (see Chapter 7 ). In addition, the active metabolite (desmethyldiazepam) has pharmacologic activity equal to that of the parent compound and a half-life of up to 9 days in adults. The most effective route of administration of diazepam is intravenous, followed by oral and rectal. The intramuscular route is not recommended because it is painful and absorption is erratic. The average oral dose for premedicating healthy children with diazepam ranges from 0.1 to 0.3 mg/kg; however, doses as large as 0.5 mg/kg have been used. The recommended dose of rectal diazepam is 1 mg/kg, and the peak serum concentration is reached after approximately 20 minutes. Compared with rectal midazolam, rectal diazepam is less effective.
Lorazepam (0.05 mg/kg) is reserved primarily for older children. Lorazepam causes less tissue irritation and more reliable amnesia than diazepam. It can be administered orally, intravenously, or intramuscularly and is metabolized in the liver to inactive metabolites. Compared with diazepam, the onset of action of lorazepam is slower and its duration of action is prolonged. The IV formulation of lorazepam is avoided in neonates because it may be neurotoxic.
Barbiturates are infrequently used for premedication as they have been replaced by oral midazolam. The advantages of barbiturates include minimal respiratory or cardiovascular depression, anticonvulsant effects, and a very low incidence of nausea and vomiting.
The relatively short-acting barbiturates thiopental and methohexital may be given rectally as a 10% solution in the presence of the parents who may hold the toddler until he or she is sedated. The usual dose of rectal thiopental or methohexital is 30 mg/kg via a shortened suction catheter, which produces sleep in about two-thirds of the children within 15 minutes. In some cases, the sedation may be profound, resulting in airway obstruction and laryngospasm. Hence, all children should be closely monitored with a source of oxygen, suction, and a means for providing ventilatory support; rectally administered methohexital has been reported to cause apnea in children with meningomyelocele. Children chronically treated with phenobarbital or phenytoin are more resistant to the effects of rectally administered methohexital, probably because of enzyme induction.
Additional disadvantages of rectal methohexital include unpredictable systemic absorption, defecation after administration, and hiccups. Contraindications to methohexital include hypersensitivity, temporal lobe epilepsy, and latent or overt porphyria. Rectal methohexital is also contraindicated in children with rectal mucosal tears or hemorrhoids because large quantities of the drug can be absorbed, resulting in respiratory or cardiac arrest.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here