Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
The diagnosis of periprosthetic fractures is based on the history, physical examination, and radiographic evaluation.
Attempts should be made to identify underlying causes of the fracture, if any.
Use of a classification system helps the surgeon better understand the problem, construct a treatment algorithm, and maximize the chances of success.
It is essential to classify the type of fracture and stability of the prosthesis correctly, as failure to identify an unstable implant will lead to treatment failure.
Current standard treatment for most periprosthetic fractures is surgical, with osteosynthesis or revision arthroplasty.
Preoperative planning is essential in the management of these fractures, but the surgeon must have the flexibility to alter the plan should circumstances change during the course of surgery.
Poor bone quality is a common finding among patients presenting with periprosthetic fractures.
Periprosthetic fracture in association with total hip arthroplasty (THA) is an uncommon but potentially devastating complication. It can occur during or after the procedure. The reported intraoperative risk is 0.3% to 1.7% and postoperative risk is up to 1.6% in primary total hip replacements at 10 years. The cumulative probability of suffering a postoperative fracture at 20 years is 7.7%, which makes it one of the most common long-term complications of THA. The risk of a periprosthetic fracture in revision hip arthroplasty is even higher for intraoperative fractures at 12% and 20-year cumulative probability of 11% for postoperative fractures. These fractures can lead to considerable morbidity due to loss of fixation of the components, loss of bone stock, and subsequent functional deficit. Most patients with these fractures require surgical management either in the form of open reduction internal fixation or a revision arthroplasty. Overall, 9.5% of reoperations after THA are attributed to periprosthetic fractures. These procedures have high surgical complication and readmission rates and compromised functional outcomes. The reoperation rate following stem revision for periprosthetic fracture has been recorded to be as high as 24%. Periprosthetic fractures are also associated with a 30-day mortality rate of 10% and considerable costs. This chapter focuses on principles of assessment, classification, and treatment of these challenging complications of arthroplasty.
The number of THAs performed has steadily risen over the years. This is due to the excellent results from THA, which has led to extended indications in the young as well as in the elderly and to an increase in life expectancy. Hence, there are higher numbers of elderly patients with THA in situ with ongoing risk of postoperative fracture due to poor bone quality, periprosthetic bone loss, and risk of falls.
A comprehensive review of the all-payer data from the National Inpatient Sample from the states participating in the Healthcare Cost and Utilization Project in the United States revealed a total of 4571 periprosthetic hip fractures between 2006 and 2010. Total hospital charges steadily increased through the study period from $66,531 in 2006 to $94,214 in 2010.
Periprosthetic fractures of the acetabulum are less common than those of femur, but they are often severe and warrant careful evaluation of the posterior column for its integrity. Intraoperative acetabular fractures can occur during preparation of the acetabulum but more commonly during impaction of a cementless acetabular component. The risk is highest with shells of elliptical shape.
A single-center series of 32,644 patients who underwent primary hip arthroplasties over 40 years with a median follow-up of 7.9 years has reported a rate of intraoperative periprosthetic femoral fractures of 1.7% (564/32,644), with statistically greater risk in female patients (2.0% vs. 1.4%, P < .001) and those older than 65 years (2.5% vs. 1.0%, P < .001). Of note, 93.8% of these fractures were undergoing cementless hip replacement and accounted for a complication rate of 3% versus 0.2% in cemented hip arthroplasty. In the same series, the cumulative probability of postoperative periprosthetic femoral fracture was 0.4% at 1 year, 0.8% at 5 years, 1.6% at 10 years, and 3.5% at 20 years. At 20 years, the cumulative probability of suffering a postoperative fracture after experiencing an intraoperative fracture was 7.2%. Of the 557 postoperative periprosthetic femur fractures, 335 of 17,466 (1.9%) occurred after uncemented implants and 222 of 15,178 (1.5%) after cemented implants ( P < .001; Fig. 107.1 ). The 20-year cumulative probabilities of femur fractures after cementless and cemented stems were 7.7% (95% confidence interval [CI] 6.2–9.1) and 2.1% (95% CI 1.8–2.0), respectively, which makes it one of the most common long-term complications of THA. When stratified based by gender, there was no increased risk of postoperative femur fractures in females (hazard ratio [HR] = 1.0; 95% CI 0.8–1.2; P = .98). Likewise, patients greater than 65 years old at operation also did not have an increased risk when compared with a patient less than or equal to 65 years of age (HR = 1.0; 95% CI 0.8–1.1; P = .59).
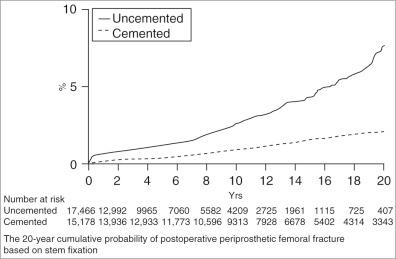
In a separate series from the same institute over 40 years, 668 intraoperative periprosthetic fractures of the femur were reported during revision arthroplasty, with an incidence of 12%. Fractures were 3 times more common with cementless stems (19%) than with cemented stems (6%, P < .001). They also reported 281 postoperative periprosthetic fractures of the femur with a 20-year probability of 11%.
Periprosthetic fractures have an overall mortality risk within 12 months of the complication of 11% to 25%. In a retrospective study of 151 periprosthetic fractures of the femur, 25.2% died within the study period. A significant effect was seen in patients with increasing age, cardiovascular problems, hypothermia, and renal failure.
The risk factors for periprosthetic fractures can be classified as patient-related or surgery-related (summarized in Box 107.1 ).
Increasing age
Young and/or active patients
Female gender
Osteoporosis
Thin cortices
Smaller stem size
Champagne flute–type proximal femur
Rheumatoid arthritis
Dysplasia
Neck of femur fracture
Hardware that required removal in the perioperative period
Radiation therapy
High body mass index
Cervical myelopathy and cervical spinal decompression
Surgical approach: Hardinge and anterior approaches
Cortical defects
Cementless femoral components
Cementless implants for fracture of neck of femur
Cementless implants in revision hip arthroplasty
Implant design
Implant position
Revision surgery
The most commonly implicated patient-dependent risk factor is increasing age, as shown by multiple researchers. THA in young patients for secondary arthritis can also predispose patients to intraoperative fractures due to abnormal anatomy or postoperative fractures due to high activity level. Female gender has been associated with a higher incidence of periprosthetic fractures. While being over 65 years of age and female gender were statistically significantly associated with an increase in intraoperative periprosthetic fracture rates, these findings were not consistently associated with an increased risk in postoperative periprosthetic fractures. This suggests that other variables—such as patient activities, proximal femoral geometry, cortical thickness, and surgical technique—may also influence fracture risk.
Osteoporosis, a history of fragility fractures, and THA for hip fracture has been shown to increase the relative risk of fracture by 4.4. Rheumatoid arthritis also increases the risk of fracture due to the reduced bone density that often coexists with this condition. Preoperative radiation to the region may also compromise the bone quality, hence predisposing patients to fractures. Blizzard and coworkers showed that patients with cervical myelopathy are at increased risk of periprosthetic hip fractures due to gait disturbances, spasticity, and falls (relative risk [RR] of 1.99 [CI, 1.61–2.45]). Risk persists, even after cervical spinal decompression (RR 1.72 [CI, 1.48–2.00]).
Anatomic variations—such as developmental dysplasia, narrow femoral canal, and champagne flute–type proximal femur —can predispose to intraoperative fractures. Patients for whom hardware removal is required in the perioperative period are at increased risk of both intraoperative and postoperative fractures. Fractures may be prevented by careful planning and thoughtful sequence of events during surgery (see Prevention section ).
The risk of periprosthetic fractures can be attributed to the surgical approaches and intraoperative findings such as cortical defects. In recent years, there has been a rise in the number of surgeons using the direct anterior approach, which has been implicated in intraoperative femoral fracture, particularly in elderly female patients. In a case-controlled study, the Hardinge approach was also found to be associated with calcar fractures.
Cementless implants have been associated with a significantly elevated risk of intraoperative periprosthetic fractures in multiple series. Cementless implants were an independent risk factor in patients with hip fractures; 7.4% Corail (DePuy, Warsaw, IN) versus 0.9% Spectron (Smith & Nephew, Memphis, TN) cemented and 12.2% Bi-Metric (Biomet, Bridgend, UK) versus 0% Exeter (Stryker, Mahwah, NJ).
Cemented stems yielded significantly lower rates of periprosthetic fractures overall. However, among the cemented femoral implants, higher risk of fractures was recorded with taper-loaded compared to composite beam designs (0.36% vs. 0.27%). Exeter femoral stems were associated with a 5-fold risk of fractures when compared to composite beam stems. The majority of cemented stem articles identified that the fractures occurred 5 or more years from the index arthroplasty The risk of fracture was also seen to rise for every year of its survival ( P < .001) .
In the revision setting, fractures were 3-fold more common with cementless implants when compared to cemented implants. Additional contributing risk factors in this group of patients were reduced bone stock, osteoporotic bone in older patients, and difficulties with removal of well-fixed implants, particularly the acetabular components.
The diagnosis of periprosthetic fracture is generally not difficult and is based primarily on radiographic examination ( Fig. 107.2A ). Intraoperative periprosthetic fractures can be diagnosed by direct observation, but this requires circumferential exposure of the calcar or on intraoperative radiographs if a fracture is suspected and, more rarely, on postoperative radiographs if a fracture was not suspected intraoperatively. These are likely to occur during 1 of the 3 stages of reconstruction: removal of fixation hardware or previous implants, bone preparation, or implant insertion. Signs that alert the surgeon to this possibility include a change in pitch while using instruments, a sudden reduction in the force required when a broach or rasp is used, or advancement of the implant beyond the planned level of placement, either in the socket or in the canal. In acetabular fractures, a sudden loss of cup stability should alert the surgeon to this possibility. Exposure of the superior portion of the posterior column is necessary to ensure safe reaming and the lack of an intraoperative fracture.
Postoperative periprosthetic fractures are typically associated with acute onset of pain and deformity that may or may be associated with a fall. Radiographs prior to the fracture should be examined for evidence of osteolysis and loosening. The authors of 2 large data reports attributed many of these fractures to implant loosening or osteolysis and highlighted the importance of regular radiographic follow-up of all patients with primary and revision THAs.
Radiographic evaluation of all suspected fractures should be comprehensive, including full-length views of the femur in 2 planes, an anteroposterior (AP) pelvic radiograph, and Judet views when required to evaluate the floor, roof, and columns of the acetabulum. Comparison with previous radiographs is valuable when available. This comparison also allows assessment of implant migration. More specialized imaging, such as computed tomography (CT), may be helpful in selected circumstances of unusual complexity, especially around the acetabulum, and can be combined with 3-dimensional reconstruction to assist with preoperative planning. Metal artifact reduction sequences (MARS) in CT and magnetic resonance imaging scanners help to improve image quality. If the acetabular component is intrapelvic, CT angiography is recommended for preoperative planning to evaluate the risk of potential vascular injuries. Sporadic cases of death from vascular injury have been reported with acute protrusion of the acetabular component.
Confirming stability of the implant is critical in the management of periprosthetic fractures, using implant exposure and interface assessment when needed. Failure to appreciate implant loosening and inadvertent management of the fracture without implant revision has been attributed to a high failure rate.
A number of classification systems have been described for periprosthetic fractures around the hip. The Vancouver Classification System (VCS) for fractures involving the femur described by Duncan and Masri in 1995 has been widely adopted. It is based on the location of the fracture, stability of the stem, and quality of the surrounding bone. Its reliability and validity have been established ( Table 107.1 ). The classification was subsequently expanded so that it could be applied to intraoperative femoral fractures. The anatomic types A, B, and C are subdivided into 3 types: subtype 1 represents a simple cortical perforation; subtype 2, an undisplaced linear fracture; and subtype 3, a displaced or unstable fracture.
| Type | Location | Subtype |
|---|---|---|
| Type A | Trochanteric region | A–G: Greater trochanter |
| A–L: Lesser trochanter | ||
| Type B | Around or just distal to the stem | B 1 : Prosthesis stable (well fixed) |
| B 2 : Prosthesis unstable (loose) | ||
| B 3 : Implant loose and bone stock inadequate | ||
| Type C | Well distal to the stem |
The principles of the VCS have been more recently expanded so that they can be applied to other joint replacements, as the Unified Classification System (UCS). The UCS is now applied to fractures following replacement of the shoulder, elbow, wrist, hip, knee, and ankle. A simple mnemonic has been proposed to assist with recall of the fracture type ( Table 107.2 ). The high interobserver and intraobserver reliability of the UCS has been established.
| Type | General Description | Pelvis and Acetabulum | Femur |
|---|---|---|---|
| A | Apophyseal | Anterior superior or inferior iliac spine, or ischial tuberosity | Greater or lesser trochanter |
| B | Bed of the implant | Acetabular fracture: B 1 : Fracture of the acetabular rim, good bone B 2 : Loose cup, good bone B 3 : Loose cup, poor bone; pelvic discontinuity |
Femoral fracture: B 1 : Stem stable, good bone B 2 : Loose stem, good bone B 3 : Loose stem, poor bone stock |
| C | Clear of the implant bed | Pelvic/acetabular fractures distal to the implant | Distal to the implant and the cement mantle |
| D | Dividing the bone that supports 2 joint replacements (interprosthetic) | Pelvic fracture between bilateral total hip arthroplasties | Between hip and knee arthroplasties |
| E | Each of 2 bones supporting joint replacement (polyprosthetic) | Pelvis and femur | Pelvis and femur |
| F | Facing or articulating with an implant | Fracture of the acetabulum articulating with the femoral hemiarthroplasty | Not applicable |
A classification for acetabular periprosthetic fractures was proposed by Della Valle and colleagues in 2003 ( Table 107.3 ). The intraoperative fracture classification has been simplified further by Davidson and coworkers. The authors believe that the UCS of the pelvis is reliable and easier to use. Like the VCS, the type B fractures, which are those fractures involving the surface of the acetabular component, have been further divided to subtype 1 when the cup is well fixed, subtype 2 when the cup is loose with good remaining bone stock, and subtype 3 when the cup is loose with poor remaining bone stock ( Table 107.4 ).
| Type I | Intraoperative during component insertion | A. Recognized, stable component, undisplaced fracture |
| B. Recognized, unstable cup, displaced fracture | ||
| Type II | Intraoperative during removal | A. Less than 50% bone stock loss |
| B. Greater than 50% bone stock loss | ||
| Type III | Traumatic | A. Component stable |
| B. Component unstable | ||
| Type IV | Spontaneous | A. Less than 50% bone stock loss |
| B. Greater than 50% bone stock loss | ||
| Type V | Pelvic discontinuity | A. Less than 50% bone stock loss |
| B. Greater than 50% bone stock loss | ||
| C. Associated with pelvic irradiation |
| Type A | Apophyseal : Anterior superior or inferior iliac spine, or ischial tuberosity |
| Type B | Bed of the implant: The acetabulum, with subtypes, see below |
| Type C | Clear of the implant: Ilium, superior, and/or inferior pubic rami |
| Type D | Dividing the pelvis between two implants (interprosthetic)—a pelvic fracture complicating bilateral hip replacements |
| Type E | Each of 2 bones supporting 1 arthroplasty (polyprosthetic): acetabulum and femur |
| Type F | Facing and articulating with a hemiarthroplasty: acetabular fracture after hip hemiarthroplasty |
The subtyping of type B acetabular fractures is thus identical to that involving the femur, and emphasizes the importance of implant loosening and degree of bone loss (volume or strength) when planning treatment ( Table 107.5 ). While each fracture pattern is more common at differing times, they can occur early or late, at the time of implantation or following injury, and during primary or revision surgery. Type B 2 and type B 3 fractures as a late or delayed presentation secondary to implant migration and osteolysis, particularly in the presence of chronic pelvic discontinuity or dissociation, are more challenging to treat successfully because the potential for fracture healing is compromised. However, it is believed that the quantity and quality of the remaining bone stock is dominant in choosing the most appropriate treatment plan, hence the distinction between B 2 and B 3 .
| Type B 1 | Fracture of the acetabular lip, wall, or floor; does not affect the stability of the component, which is still well fixed |
| Type B 2 | Fracture of the acetabulum (such as the posterior column) or pelvic discontinuity, with a loose acetabular component but adequate bone stock to support an uncomplicated acetabular component revision; with or without bone graft; with or without plate fixation of the fracture |
| Type B 3 | Fracture of the acetabulum, with a loose component and severe bone loss such that complex reconstruction or a salvage procedure is required |
Pelvic discontinuity is important to recognize because standard acetabular revision techniques may not be adequate for reconstruction. Berry described the diagnostic features of pelvic discontinuity in a review of 27 patients. On the AP radiograph, 3 cardinal features were an obvious fracture line, malalignment of the upper and lower halves of the hemipelvis (usually due to medial displacement of the inferior half), and rotation of the inferior half of the hemipelvis in comparison with the opposite side (observed by comparing the size and shape of the obturator foramina ( Fig. 107.2B ). Judet views can be helpful when implants obscure the normal acetabular anatomy, especially the columns. In some cases, in which it is still difficult to diagnose a pelvic discontinuity on plain radiographs, MARS CT scan may be helpful. Furthermore, in every case, it is important to confirm the continuity of the hemipelvis during the revision procedure, regardless of preoperative radiographic findings.
The objective of the VCS was successful assessment and management of these complex complications of THA (see Table 107.1 ). It takes into consideration 3 core principles: the location of the fracture (whether the fracture involves the bone supporting the implant or is distant to it); stability of the implant (whether the bone-implant interface was stable prior to fracture and remained so after the injury); and the adequacy of the bone stock supporting the implant (whether it is sufficient to permit revision without requiring additional major reconstruction).
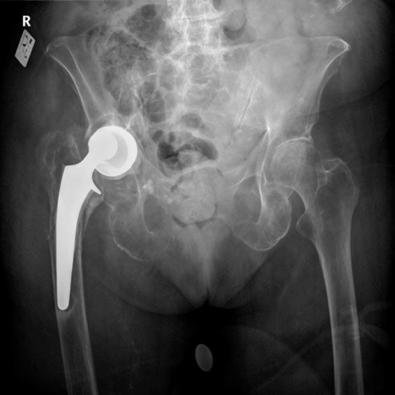
The 3 major anatomic types defined by this classification system depend on the location of the fracture: type A refers to the trochanteric area (one or both trochanters and proximal metaphysis, not extending to the diaphysis) and has little influence on implant fixation. These fractures are subdivided into greater trochanter (A G ; Figs. 107.3 and 107.4 ) and lesser trochanter (A L ; Fig. 107.5 ) depending on the location of the fracture. Type B refers to a fracture around or just distal to the femoral stem (metaphysis and/or diaphysis) and has been subdivided into B 1 , B 2 , and B 3 on the basis of stability of the implant and adequacy of bone. Type C refers to fractures that are sufficiently distal to the tip of the stem that their treatment can be considered independent of the presence of the implant (the diaphysis and/or metaphysis; Fig. 107.6 and see Table 107.1 ). If a fracture occurs through the cement mantle distal to an otherwise well-fixed cemented stem, this would be classified as a B 1 . When the fracture passes through the cement mantle around the stem, it leads to a B 2 fracture, irrespective of whether the stem was well fixed before the fracture. In both of these categories, the bone stock is usually good. In some cases, the distinction between type B 1 and B 2 will require further radiologic investigation, such as MARS CT scan or fluoroscopy), a review of prior radiographs, or examination of the bone-implant interface at the time of the operation. However, it is best to make this differentiation preoperatively given the differences in the surgical approach between a revision procedure and fixation of a femoral fracture in which there is no need to dislocate the hip.
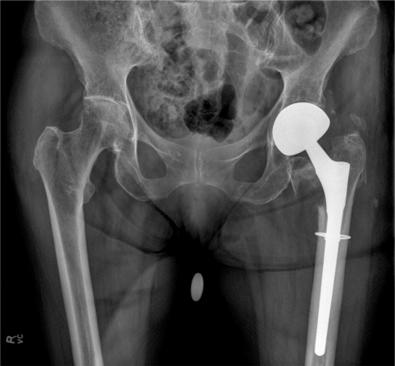
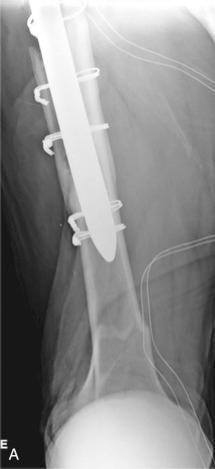
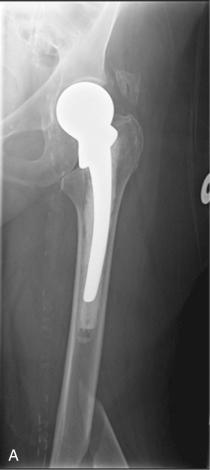
Extending the principles of the VCS, in the UCS for periprosthetic femoral fractures (see Table 107.2 ), type A fractures in the femur are also subdivided into greater trochanter (A G ) and lesser trochanter (A L ). Type B has been subdivided into those in which the implant is stable (B 1 ), those in which the implant is loose (B 2 ), and those in which the bone stock is poor and the implant is loose (B 3 ). In type C, the fracture is distant to the arthroplasty (see Fig. 107.6 ). In type D, the fracture has divided a femur that supports 2 implants (hip and knee replacement; Fig. 107.7 ); these are also known as intraprosthetic fractures. In type E, the fractures involve 2 bones that support 1 joint replacement, such as both the acetabulum and femur after hip replacement ( Fig. 107.8 ).
A single-center series of 32,644 patients over 40 years old with a median follow-up of 7.9 years identified Vancouver A G fractures as the most common, followed by Vancouver B 2 fractures. A more in-depth analysis revealed that Vancouver A G , A L , B 1 , and B 2 fractures were more common with cemented stems, while Vancouver B 3 and C fractures were more common with uncemented stems. In addition, Vancouver A G fractures mostly occurred with no known trauma, were avulsion-type fractures, and were treated nonoperatively. On the other hand, Vancouver B fractures were mostly due to a fall from standing height, were oblique or spiral-pattern fractures, and were treated with open reduction internal fixation or revision.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here