Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
![]() This chapter includes an accompanying lecture presentation that has been prepared by the authors: .
This chapter includes an accompanying lecture presentation that has been prepared by the authors: .
A neuroprosthetic is a device that supplants or supplements the input or output of the nervous system.
The clinical goal of a neuroprosthetic is to restore lost function or augment existing abilities.
The interface a neuroprosthetic uses to link a human with a machine is variable.
Research is rapidly advancing on multiple parallel pathways, many of which will converge to form several different neuroprosthetic systems.
Neuroprosthetics exist on a spectrum of invasiveness with various advantages and disadvantages.
Motor output systems are the most widely studied.
The cochlear implant is the most successfully developed and clinically utilized neuroprosthetic device.
Multiple commercialization efforts are under way and expected to produce clinical devices that will require neurosurgical implants.
Neuroprosthetics continues to be a rapidly expanding field that will affect the practice of neurosurgery through emerging technologies aimed to restore function to patients. A neuroprosthetic in its most simplistic form is a device that supplants or supplements the input and/or output of the nervous system. The fundamental application is to bypass a deficit caused by disease or to augment existing function for improved performance. The technical approaches to accomplish this goal are numerous and typically include research from multiple fields of study. This rich variety of approaches has led to a diversity of form factors to meet different clinical indications and surgical considerations. Generally speaking, neuroprosthetic devices have numerous synonymous names such as brain-computer interface (BCI), brain-machine interface (BMI), neural interface system (NIS), direct neural interface (DNI), mind-machine interface (MMI), and other nomenclature that similarly describes an interface of the nervous system with an external device.
Research and development of neuroprosthetics requires integration across a diversity of technical and scientific disciplines ranging from neuroscience and computer science to engineering. Neuroprosthetic research had its conceptual inception long before the advent of an organized academic field of study. Seminal work was described by Vidal, who upon observing modulations of signals in the electroencephalogram, had the foresight in 1973 to ask “Can these observable electrical brain signals be put to work as carriers of information in man-computer communication or for the purpose of controlling such external apparatus as prosthetic devices or spaceships?”. , Although the proposition of spaceship control has yet to be realized, control of a prosthetic device is now becoming commonplace in the translational research laboratory.
Although computational performance was initially restrictive, advances in microprocessor design and digital signal analysis now outperform neuroprosthetic requirements such that computational speed is no longer a rate-limiting factor. Similar technological advances have provided the necessary tools for device development allowing innovative applications to introduce motor, sensory, visual, auditory, speech, and other modalities to the field. Here we review these applications in neuroprosthetics by presenting the state of the art across a multitude of modalities and emphasize the role of neurosurgeons in its translation from fundamental research to clinical application.
The goal of a neuroprosthetic device is to replace or augment an individual’s function by interfacing an external device with the nervous system. A classic understanding of a neuroprosthetic device is a device that records brain signals from the user (i.e., signal input), computationally analyzes these signals to infer the user’s intentions, and then transforms these intentions to an external effector output. There are multiple modalities to achieve this goal. The source of signals used as input to the device can range from individual neuronal spiking to field potential from cortical ensembles, or action potentials conducted by peripheral nerves. Output effectors are also wide-ranging. Examples include computer cursor movement, robotic arm control, and re-animation of paretic limbs. By reversing the direction of information transfer, one can similarly develop an input device. In this scenario, the device input is recorded from the external environment and converted to an appropriate stimulus delivered to the nervous system. These devices may be complimentary to an output device, such as sensory input to accompany the robotic arm of a prosthesis for tactile or proprioceptive feedback. In contrast, it may be independent in application, such as a cochlear implant in which acoustic information recorded from a microphone is translated to a corresponding electrical stimulus delivered to the acoustic nerve, thereby providing auditory perception to the otherwise deaf ear. In the broadest sense, neuroprosthetics can be categorized as “output neural interfaces” that convert the brain’s intentions to external actions, or as “input neural interfaces” that take information from the environment and convert it to the brain’s perceptions.
Neuroprosthetics may also be broadly characterized by the modality of operation (e.g., motor, somatosensory, speech, auditory) and the source of neural interface (e.g., single-unit neuron, cortical local field potential, peripheral nerve). The most successful neuroprosthesis that has seen widespread adoption and success in clinical application to date is the cochlear implant. This technology has played an ever-expanding role in otology. For neurosurgery, the developing neuroprosthetic research that is poised to affect the clinical field is motor output prosthetics. The general goal of a motor output prosthesis is to develop a device that is capable of interpreting an individual’s movement intentions in real time and direct the output to an effector such as a robotic limb. With these canonical examples in mind, we will explore the various modalities of interfacing with the nervous system and their clinical implications.
Electroencephalography (EEG) is a commonly used means of studying cerebral electrophysiology because of its noninvasive nature and clinical familiarity. Through electrodes placed on a subject’s scalp, an investigator is able to record the neural rhythms manifested in electrical potentials resulting from complex interactions of the neurons and glial cells that make up the cerebrum’s functional systems. The electroencephalogram, therefore, represents measurement of summated electrical potentials from the electrochemical interaction of a vast number of cells, both neuronal and glial in origin.
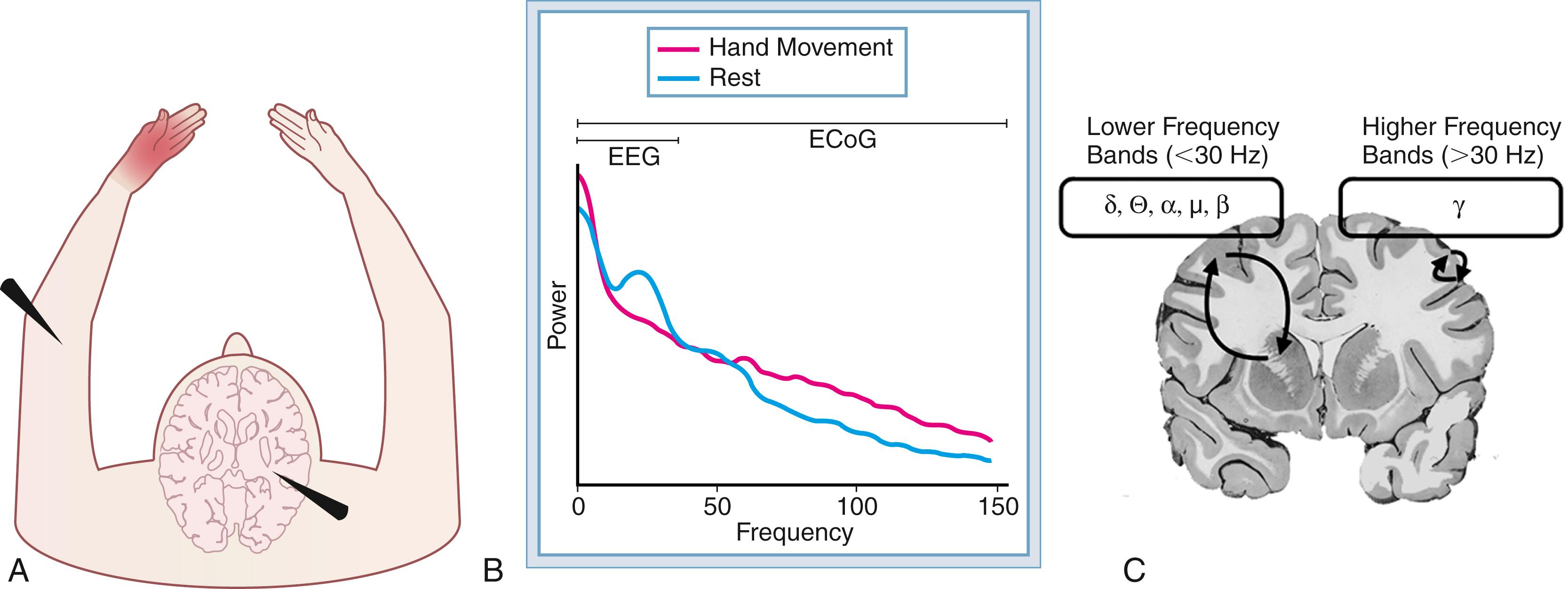
Measured electrical potentials are then digitized with the aid of biosignal amplifiers and analog-to-digital converters for computational analysis. By periodically sampling a continuous signal in regular short intervals, discrete values are obtained and stored in series, thereby converting the signal from an analog to digital domain. The time-varying signal is then analyzed as the sum of multiple sinusoidal signals of varying frequency and amplitudes. This process commonly employs the discrete Fourier transform and is said to convert from the time domain to the frequency domain. In the frequency domain, one can observe the change in power at a given frequency over time, which is known as time-frequency analysis. The distribution of signal power across a range of frequencies is the signal spectrum, and its plot over time is the spectrogram. By time-locking the sampled signal to measured subject data, correlations are made between cortical activity and task performance. One such observation in signals measured over the motor homunculus is the reliably reproducible decrease in spectral power in the 8- to 13-Hz range with overt or imagined movement of the hand contralateral to side of cortical recording.
The electroencephalogram in humans is commonly segmented into meaningful components in the frequency domain and represented by Greek alphabet character designations. These are classically defined as the delta band below 4 Hz, theta band from 4 to 8 Hz, mu band from 8 to 13 Hz (also known as alpha ), beta band from 13 to 30 Hz, and gamma band beyond 30 Hz. The mu rhythm is described in relation to motor electrophysiology over the Rolandic cortex ( Fig. 132.1A ). Decreases in mu band spectral power with activity (see Fig. 132.1B ) are thought to represent event-related desynchronization. This release of synchronized cortical rhythms is thought to be orchestrated by thalamocortical circuits modulating motor cortical systems (see Fig. 132.1C ). , Real-time measurements of power fluctuations in this spectral band provide a source signal for motor actions. Wolpaw and colleagues demonstrated the transformation of this source signal to effector output by training subjects to control a virtual cursor displayed on a computer screen. In effect, the velocity of cursor movement was derived in real time from changes in the mu spectral power volitionally modulated by the user. Importantly, similar modulations of the electroencephalogram are also observed in imagined motor actions, thereby allowing similar control without peripheral activation of motor units.
EEG correlates of imagery-based activity are critical for clinical application to disease-affected patient populations. Translation to those affected by neuromuscular disorders would be impractical if overt motor function is required for the production of an adequate source control signal. By definition, then, those who would benefit from such a prosthesis would be the very population incapable of initiating a control signal, obviating its clinical applicability. It is therefore critical that individuals affected by neurological disorders such as amyotrophic lateral sclerosis (ALS), locked-in syndrome, or spinal cord injury (SCI) are still capable of modulating cortical rhythms in the absence of end organ effect. Thankfully, preservation of this function has been demonstrated in subjects with long-term deficits in whom volitional modulation of stable electrophysiologic structure was maintained.
With these basic tenets in place, EEG-based brain computer interfaces have led the field in human neuroprosthetic research. However, one major shortfall of electroencephalography is the physical separation of the cortical source signal and the scalp-based recording electrodes. This distance is occupied by the meninges, bone, and scalp, which results in limited spectral and spatial resolution. For a scalp based electrode to record a measurable signal from the cortex, electrical potentials must be summated across an area of cortex approximately 6 cm 2 in diameter. The spatial resolution of separable source signals is thereby inherently limited. Similarly, destructive summation of temporally overlapping signals results in cancelation and therefore unresolved higher frequencies in EEG recordings. These limitations may be mitigated in part by intracranial placement of recording electrodes at the cost of increased invasiveness ( Fig. 132.2 ).
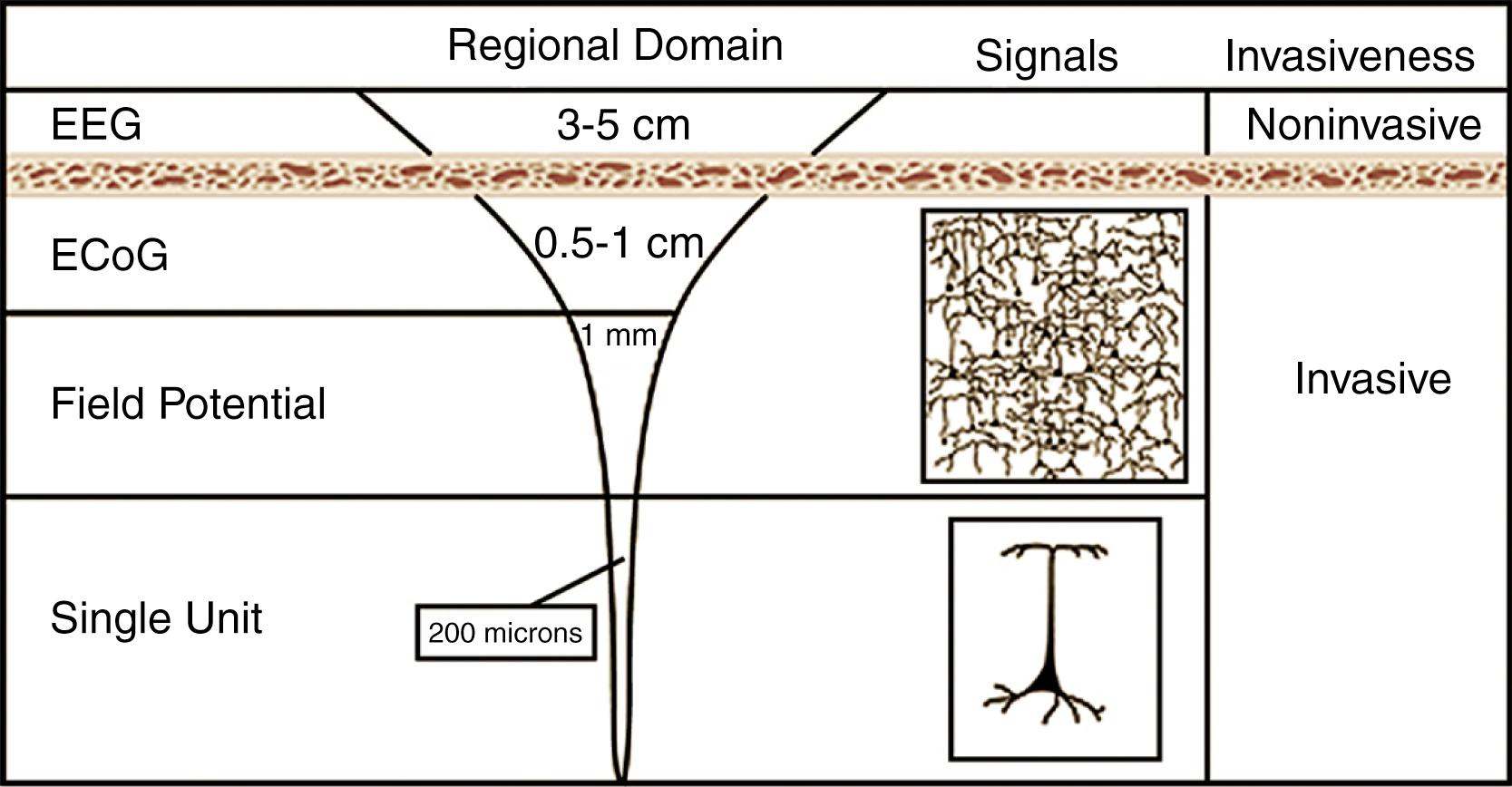
Recording electrodes placed directly into the substance of the brain, known as intraparenchymal electrodes, allow for high-resolution measurements of electrical potentials. Although maximally invasive, this method of signal acquisition provides the greatest fidelity in both spatial and spectral domains in very small anatomic locations. Additionally, spike-sorting algorithms allow for detection of action potentials to the resolution of individual neurons. These source signals are commonly referred to as single units. Because of the risk associated with placement of intraparenchymal electrodes, the bulk of experimental data has been conducted with animal models.
Research in animal models has shown great success and illustrates vast potential for future neuroprosthetic applications. A large contribution to our current understanding of motor system electrophysiology and movement encoding has been garnered from macaque monkey experiments. With sufficient understanding of motor electrophysiology, the neural signals associated with intention of movement can be decoded. Beyond the ability to infer intentions from neuronal activity about endogenous limb movement, these signals can be transposed to control a robotic limb in substitution. This has been advanced by research teams performing real-time control of robotic arms with macaque monkeys via electrode arrays implanted in the motor cortex. , Some experiments have demonstrated quite complex levels of control, such that the arm was dexterous enough to enable a monkey to feed itself. Success with neuroprosthetic control in primates subsequently led to translation in limited human clinical trials ( Fig. 132.3A ). , , Human trials have enrolled patients with a variety of disabling neurological conditions including traumatic spinal cord injury, , brainstem stroke, and neurodegenerative diseases such as ALS and spinocerebellar degeneration.
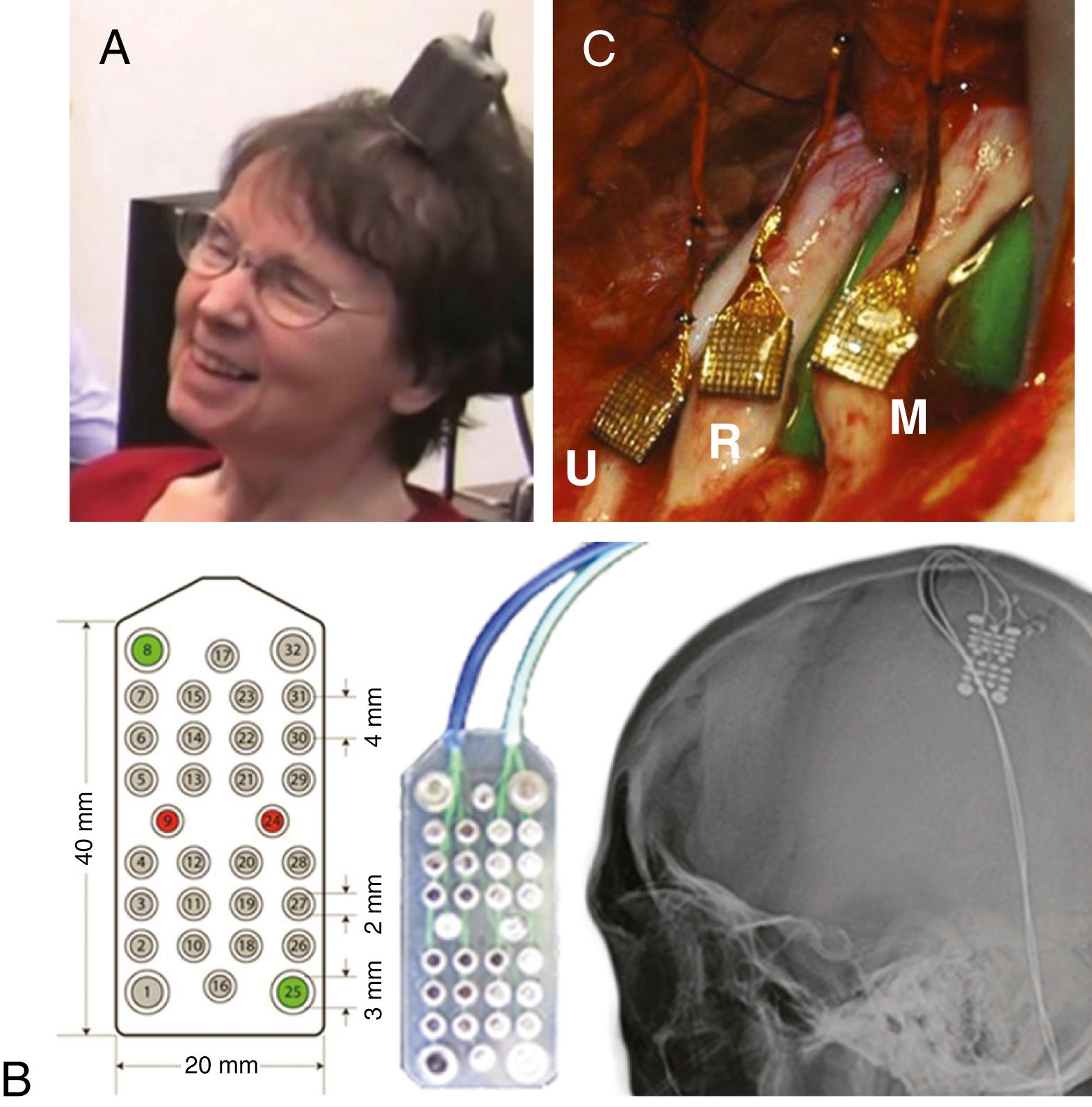
Although the clinical trials in humans demonstrate promising proof of concept in functionality, long-term clinical viability of these constructs remains a barrier. Long-term recording with intraparenchymal electrodes is impeded over time by the body’s immunologic response to the introduction of a foreign object in the brain. When depth electrodes are implanted for a prolonged time, a process of gliosis occurs resulting in scar formation at the electrode interface. Over time, gliosis functionally insulates the electrode, limiting its recording ability, and ultimately prohibits its ability to adequately interface with the brain. , Materials engineering efforts are actively exploring new methodologies and substrates from which to build electrodes to reduce or eliminate the gliosis process from inhibiting electrode function.
Advances in fabrications techniques now allow for micrometer-size electrode development to better match the physical properties of the brain’s parenchyma (see for review). A sufficiently thin profile allows electrodes to be flexible and induce less sheer force on the surrounding tissue, potentially leading to less inflammation. Less induced inflammation is expected to limit the gliotic response and lead to greater longevity of signal fidelity. However, it raises a new technical challenge of inserting an exquisitely flexible electrode in the relatively firm parenchyma and overlying meninges. Novel solutions include use of a stiffening device or shuttle that can assist with insertion but does not remain implanted , or a robotic device that can assist with placement. These techniques have shown promise in animal models but have not reached human trials at the time of this writing. Although innovations in electrode design continue to rapidly advance the field, many notable hurdles remain and must be solved before long-term neuroprosthetics are achieved with intraparenchymal electrodes.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here