Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Chemotherapy and radiation therapy (RT) are two of the major modalities used to treat cancer. Their goal is to kill or inactivate enough cancer cells that the body’s own defenses can control the disease without unacceptable damage to normal tissue. Unfortunately, both RT and chemotherapy are relatively nonspecific and depend on their ability to do more damage to rapidly dividing cancer cells. The therapeutic/toxic ratio is often low; even in highly sensitive tumors such as acute lymphoblastic leukemia, Hodgkin disease, and germ cell tumors, for which the cure rate is high, many patients suffer serious side effects of therapy, either immediately or months to years later.
The nervous system may be expected to be relatively insensitive to the side effects of cancer therapy. It is protected from exposure to many chemotherapeutic agents by the blood–brain, blood–cerebrospinal fluid (CSF), and blood–nerve barriers. Furthermore, most neurons do not reproduce, and glia reproduce only slowly, thus affording protection against agents that are directed against dividing cells. Nevertheless, nervous system toxicity is common, second only to myelosuppression as a reason for limiting the dose of chemotherapy, and often is dose limiting for RT as well. Some newer therapies, especially targeted therapies (i.e., ALK inhibitors) are specifically designed to penetrate the central nervous system (CNS) to treat brain metastases; these agents could carry a greater risk of causing neurotoxicity. The purpose of this chapter is to describe the side effects of these therapeutic modalities on the central and peripheral nervous systems (PNS). The emphasis is on chemotherapeutic agents and radiotherapeutic approaches that are used widely in clinical practice, with particular attention to newer agents and especially the novel immunotherapies that have rapidly transformed the oncology landscape.
Table 28-1 classifies the major chemotherapeutic agents that have been reported to cause central nervous system (CNS) or PNS toxicity. Table 28-2 lists the neurotoxic signs caused by agents commonly used in cancer patients.
| Neurotoxicity * | Neurotoxicity * | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| Agents | Drug | PNS | CNS † | Muscle | Agents | Drug | PNS | CNS † | Muscle |
| Antimetabolites | 5-Azacitidine | + | + | ?+ | Podophyllotoxins | Etoposide (VP-16) | ?+ | ?+ | – |
| 5-Fluorouracil | – | ++ | – | Teniposide (VM-26) | ?+ | ?+ | – | ||
| Capecitabine | ?+ | + | – | Monoclonal antibodies | Bevacizumab | – | + | – | |
| Cladribine | – | + | – | Rituximab | – | + | – | ||
| Cytarabine | + | + | – | Small-molecule inhibitors | Bortezomib | ++ | – | – | |
| Fludarabine | – | ++ | – | Carfilzomib | + | + | – | ||
| Gemcitabine | ?+ | ?+ | + | Gefitinib | – | + | – | ||
| Methotrexate | – | ++ | – | Imatinib | – | + | – | ||
| Nelarabine | ++ | + | – | Selumetinib | – | + | + | ||
| Pemetrexed | + | – | – | Sorafenib | ++ | + | – | ||
| Pentostatin | + | + | – | Sunitinib | – | ++ | – | ||
| Alkylating agents | Carmustine (BCNU) | – | + | – | Tipifarnib | ++ | + | – | |
| Chlorambucil | – | ?+ | – | Vemurafenib | – | + | – | ||
| Cyclophosphamide | – | ?+ | – | Other biologics | Cyclosporine | + | + | + | |
| Ifosfamide | – | ++ | – | Interferons | + | ++ | – | ||
| Lomustine (CCNU) | – | + | – | Interleukins | – | ++ | – | ||
| Temozolomide | – | + | – | Lenalidomide | + | – | – | ||
| Thiotepa | – | + | – | Mycophenolate mofetil | + | + | – | ||
| Platinums | Carboplatin | + | – | – | Tacrolimus | – | + | – | |
| Cisplatin | ++ | ++ | – | Thalidomide | ++ | – | – | ||
| Oxaliplatin | ++ | – | – | Miscellaneous | Dexamethasone | – | ++ | ++ | |
| Taxanes | Cabazitaxel | ++ | – | + | DTIC (Dacarbazine) | – | ?+ | – | |
| Docetaxel | + | – | + | Hexamethylmelamine | + | + | – | ||
| Nab-paclitaxel | ++ | – | + | Ixabepilone | ++ | – | – | ||
| Paclitaxel | ++ | – | + | L-Asparaginase | – | + | – | ||
| Vinca alkaloids | Vinblastine | + | – | – | Procarbazine | + | + | – | |
| Vincristine | ++ | + | ?+ | Retinoids | + | ++ | – | ||
| Vinorelbine | + | – | – | Suramin | ++ | + | – | ||
| Tamoxifen | – | + | – | ||||||
| Immunotherapies (checkpoint inhibitors and CAR T cells) | + | + | + | ||||||
* ?+, questionable; +, rare; ++, common; –, none; CNS, central nervous system; PNS, peripheral nervous system.
| Acute Encephalopathy (Delirium) |
| 5-Azacytidine, 5-fluorouracil, asparaginase, bevacizumab, capecitabine, CAR T cells, carmustine, checkpoint inhibitors, cisplatin, cladribine, corticosteroids, cyclophosphamide, cyclosporin A, cytarabine, dacarbazine, docetaxel, etoposide (HD), fludarabine, gemcitabine, hydroxyurea, ifosfamide, imatinib, interferons, interleukins 1 and 2, methotrexate (HD, IV, IT), nelarabine, nitrosoureas (HD or arterial), paclitaxel, pentostatin, procarbazine, tacrolimus, tamoxifen, thalidomide, thiotepa (HD), tipifarnib, vincristine |
| Seizures |
| 5-Fluorouracil, amifostine, asparaginase, bevacizumab, busulfan (HD), CAR T -cells, carmustine, cisplatin, corticosteroids, cyclophosphamide (HD), cyclosporin A, cytarabine, dacarbazine, docetaxel, erythropoietin, etanercept, etoposide (HD), fludarabine (HD), gemcitabine, hydroxyurea, ifosfamide, interferon, interleukin-2, letrozole, leuprolide, methotrexate, nelarabine, octreotide, paclitaxel, pentostatin (HD), temozolomide, teniposide, thalidomide, topotecan (IT), vincristine |
| Headaches Without Meningitis |
| 5-Fluorouracil, anastrozole, asparaginase, CAR T cells, carmustine, capecitabine, cetuximab, cisplatin, cladribine, corticosteroids, cytarabine, erlotinib, estramustine, etoposide, fludarabine, gefitinib, gemtuzamab, hydroxyurea, ibritumomab, imatinib, interferons, interleukins, isotretinoin, ixabepilone, letrozole, leuprolide, methotrexate, nelarabine, panitumumab, procarbazine, retinoic acid, rituximab (IV and IT), sorafenib, sunitinib, tamoxifen, temozolomide, thalidomide, thiotepa, topotecan, traztusumab, vemurafenib, vincristine |
| Visual Loss |
| 5-Fluorouracil, bevacizumab, carboplatin, carmustine, cisplatin, cytarabine, etanercept, etoposide, fludarabine, interferon, interleukin, ipilimumab, isotretinoin, methotrexate, nitrosoureas (IA), oxaliplatin, paclitaxel, pentostatin, tamoxifen, vincristine |
| Chronic Encephalopathy (Dementia) |
| 5-Fluorouracil, carmofur, carmustine, cisplatin, cytarabine, dacarbazine, fludarabine, ifosfamide, interferon-alpha, methotrexate, rituximab (IT), topotecan (IT) |
| Peripheral Neuropathy |
| 5-Azacitidine, 5-fluorouracil, bortezomib, cabazitaxel, capecitabine, carboplatin, carfilzomib, checkpoint inhibitors, cisplatin, cladribine, cytarabine, docetaxel, etoposide, fludarabine, gemcitabine, ifosfamide, interferon, ipilimumab, ixabepilone, lenalidomide, nab-paclitaxel, nelarabine, oxaliplatin, paclitaxel, pemetrexed, pentostatin, procarbazine, sorafenib, sunitinib, teniposide, thalidomide, tipifarnib, vinca alkaloids |
| Cerebellar Dysfunction (Ataxia) |
| 5-Fluorouracil, cyclosporin A, cytarabine, nelarabine, procarbazine, vinblastine, vincristine |
| Myelopathy (Intrathecal Drugs) |
| Cytarabine, methotrexate, thiotepa |
| Aseptic Meningitis |
| Checkpoint inhibitors, cytarabine (IT), IVIg, methotrexate (IT), monoclonal antibodies, rituximab (IT), thiotepa (IT), topotecan (IT) |
Fludarabine (2-fluoroadenosine arabinoside) is active against a variety of lymphoproliferative neoplasms. It is highly immunosuppressive and has been associated with the development of progressive multifocal leukoencephalopathy (PML) in some patients. In addition, fludarabine can cause delayed neurotoxicity leading to a severe encephalopathy and occasionally cortical blindness. Older patients and those receiving higher doses of the drug are at greater risk.
Methotrexate causes both acute and delayed neurotoxicity. The side effects associated with intrathecal administration are listed in the section on Intrathecal Chemotherapy.
A stroke-like syndrome affecting adults or children occasionally follows systemic high-dose methotrexate infusion. The disorder usually follows the second or third treatment by 5 or 6 days and is characterized by alternating hemiparesis associated with aphasia and sometimes encephalopathy or coma. Unequivocal seizure activity is rare, and the electroencephalogram (EEG) typically is slow. Magnetic resonance imaging (MRI) displays foci of hyperintensity on fluid-attenuated inversion recovery (FLAIR) sequences and diffusion-weighted imaging displays well-delineated hyperintense areas affecting the deep white matter but not conforming to a vascular territory. Apparent diffusion coefficient (ADC) maps demonstrate decreased signal intensity in a corresponding distribution, suggesting restricted diffusion, similar to an acute vascular event. Patients generally recover spontaneously in 48 to 72 hours with complete or partial resolution of the imaging abnormalities. Recurrences are rare with subsequent treatments. The pathogenesis is unknown.
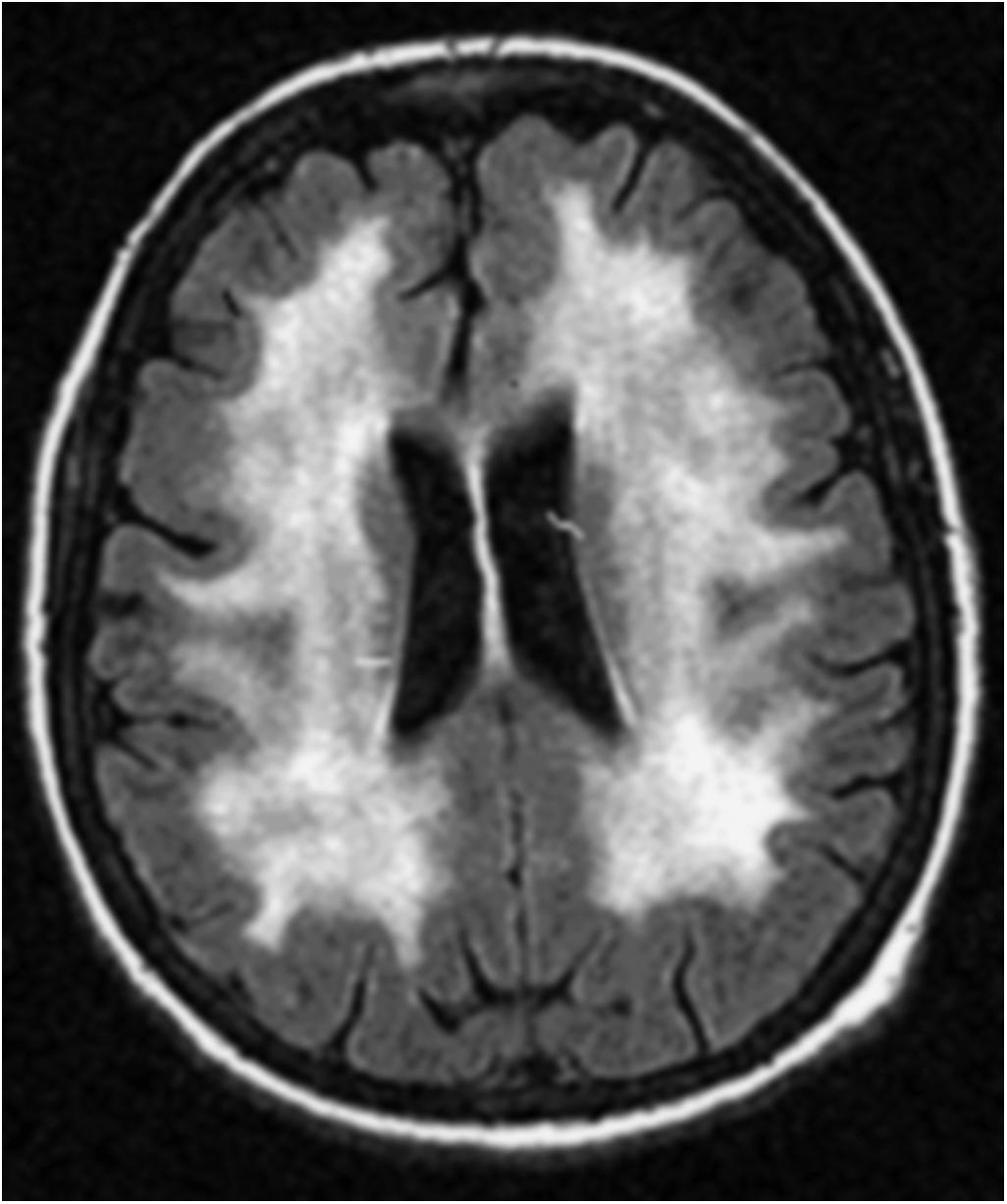
Leukoencephalopathy may appear months to years following therapy, beginning insidiously or abruptly with personality changes and learning disability. The disorder generally follows repeated doses of intravenous high-dose methotrexate or intrathecal methotrexate, but it may occur after standard doses as well. Although the syndrome can be caused by methotrexate alone, it is enhanced by brain RT and the combination of systemic and intrathecal drug. The sequence of modalities is probably also important. When methotrexate is administered concurrently with or follows cranial RT, the synergy is particularly toxic. The clinical course varies. Patients may recover slowly over weeks or months, their symptoms may stabilize with a mild to moderate dementia, or relentless progression may occur, with spastic hemiparesis or quadriparesis, severe dementia, and coma, ending in death. Seizures can occur, usually late in the course. The MRI reveals cerebral atrophy, bilateral and diffuse periventricular white matter hyperintensity on T2-weighted or FLAIR sequences ( Fig. 28-1 ), ventricular dilatation, and, sometimes, cortical calcifications. Similar findings may occasionally be seen in asymptomatic patients who have received methotrexate. Focal enhancement may be present in the early stages, but does not persist. Neurologic signs are usually preceded by radiographic white matter changes on MRI, and identical radiographic findings may be seen in patients years after prophylactic treatment with intrathecal or intravenous methotrexate, even in the absence of RT. No effective treatment exists.
Intravenous high-dose cytarabine (ara-C; 3 g/m 2 per 12 hours for 8 to 12 doses) may cause central or peripheral neurologic disorders. Cerebellar dysfunction occurs most frequently in older patients and in those with pre-existing renal dysfunction, usually at a cumulative dose ≥36 g/m 2 ; however, it has been reported after a single dose of 3 g/m 2 . Patients develop dysarthria, nystagmus, and appendicular and gait ataxia. Confusion, lethargy, and somnolence may also occur. With cessation of the drug, complete resolution of symptoms and signs generally occurs within 2 weeks, but some have persistent deficits.
Peripheral neuropathy, axonal or demyelinating or both, is a rare complication of ara-C. Other reported toxicities include seizures, intracranial hypertension, reversible ocular toxicity (blurred vision, photophobia, burning eye pain, and blindness), bulbar and pseudobulbar palsy, Horner syndrome, the “painful legs, moving toes” syndrome, brachial plexopathy, reversible bilateral lateral rectus palsies, and acute aseptic meningitis (after intravenous injection). There is no treatment for any of the neurotoxic effects of ara-C but many patients recover spontaneously.
Nelarabine is a purine analogue that is used in the treatment of T-cell acute lymphoblastic leukemia. Motor and/or sensory peripheral neuropathy occurs in approximately 20 percent of patients. Central neurotoxicity most frequently includes somnolence and fatigue around 1 week after drug administration. Other symptoms include headache, seizures, ataxia, tremor, amnesia, and paraplegia has also been reported. The deficits may not be reversible and so drug discontinuation is recommended.
Gemcitabine is a deoxycytidine analogue. Sensory neuropathy occurs in approximately 10 percent of patients, and autonomic neuropathy has also been reported. Gemcitabine can also cause an acute myositis, which may be multifocal. Patients may present with painful symmetric weakness of the proximal muscles, with elevation of muscle enzymes in the blood. Symptoms resolve rapidly with discontinuation of the drug and with corticosteroids, and patients often do well with rechallenge. Gemcitabine can also cause a focal myositis hours to days after administration, in a field previously irradiated months or even years earlier, known as radiation recall. Many other agents have also been associated with radiation recall, including capecitabine and docetaxel.
Ixabepilone is an epothilone that stabilizes microtubules and induces apoptosis. Neuropathy is the most common neurotoxicity, occurring in more than 60 percent. It is predominantly sensory, but a mild motor neuropathy may occur infrequently. The sensory neuropathy is generally mild to moderate and improves with drug discontinuation, typically within 1 to 2 months. Alternatively, dose reduction can be employed if the severity is no worse than grade 2 by National Cancer Institute common toxicity criteria (NCI CTC), and stopped if it reaches grade 3. Less common neurologic side effects include headache and dizziness.
Approximately 60 percent of patients receiving paclitaxel (Taxol) at ≤250 mg/m 2 dose develop paresthesias of the hands and feet that do not usually progress and may resolve despite continued therapy. The neuropathy is predominantly sensory and affects all modalities. Paclitaxel causes axonal damage, with secondary demyelination, probably reflecting damage to the cell body. A few patients also develop proximal muscle weakness that resolves; it is usually associated with peripheral neuropathy. Acute arthralgia and myalgia of the legs that curtail activity (and are sometimes mistaken for neuropathy) may occur 2 to 3 days after a course of paclitaxel. Encephalopathy and seizures may occur, but are rare. Docetaxel causes the same type of sensory neuropathy as paclitaxel but is less neurotoxic. Paclitaxel or docetaxel neuropathy is enhanced by prior or subsequent neurotoxic chemotherapy, particularly cisplatin or vinorelbine. To date there are insufficient human data to support any neuroprotective agents against taxane-induced (or any chemotherapy-induced) peripheral neuropathy.
Nab-paclitaxel, an albumin-bound formulation of paclitaxel, has a higher incidence of grade 3 sensory neuropathy compared to standard paclitaxel. Patients may improve with discontinuation, and the drug may be rechallenged at a reduced dose.
Cabazitaxel is a semisynthetic taxane that can be effective in docetaxel- and paclitaxel-resistant tumors. Peripheral neuropathy can occur but is rarely severe.
Vincristine affects primarily the peripheral nerves but can also be toxic to the CNS, cranial nerves, and autonomic nervous system ( Table 28-3 ). Vinorelbine and vinblastine are much less neurotoxic. A dose-limiting sensorimotor neuropathy appears in virtually all patients. The earliest complaint is tingling and paresthesias of the fingertips and later of the toes. Fine movements of the fingers and toes are often impaired. Muscle cramps, usually diurnal, affect arms and legs and may be the first symptom of neurotoxicity. Weakness, especially of the extensors of the feet and hands, is frequent. Foot drop is either unilateral or bilateral; unilateral foot drop is more common in patients who have lost weight and habitually sit with crossed legs, causing peroneal nerve compression. The weakness is usually tolerable, but rarely patients may become bedbound or quadriparetic, particularly if there is a pre-existing neuropathy. The sensory symptoms, weakness, and lost reflexes are reversible, but may require months to improve after the medication is stopped. Neurophysiologic studies show an axonal neuropathy.
| Toxic Effect | Subacute (1 day–2 weeks) | Intermediate (1–4 weeks) | Chronic (>3 weeks) |
|---|---|---|---|
| Peripheral neuropathy | Depressed Achilles reflex (universal) | Other tendon reflexes depressed, paresthesias | Sensory loss, weakness, “foot drop” gait |
| Myopathy? | Muscle pain, tenderness, (especially quadriceps); jaw pain | – | – |
| Autonomic neuropathy | Ileus with cramping abdominal pain | Constipation, urinary hesitancy, impotence, orthostatic hypotension | – |
| Cranial neuropathy (uncommon) | – | – | Optic atrophy; ptosis; sixth, seventh, and eighth cranial nerve dysfunction; hoarseness; dysphagia |
| “Central” toxicity | – | Seizure, SIADH | – |
Vincristine occasionally causes focal neuropathies of peripheral or cranial nerves. The most common is oculomotor nerve involvement with ptosis. Less frequent is ophthalmoplegia with diplopia. The recurrent laryngeal nerve, facial nerve, acoustic nerve, and optic nerve are also affected occasionally. These various neuropathies may be bilateral or unilateral. Night blindness due to retinal damage has also been reported.
Autonomic neuropathy, characterized by colicky abdominal pain and constipation, occurs in almost all patients. Rarely, paralytic ileus develops and may be fatal. Prevention of constipation is essential, and all patients should receive a prophylactic bowel regimen. Other manifestations of autonomic dysfunction include bladder atony, impotence, and postural hypotension.
CNS toxicity may result from hyponatremia due to inappropriate secretion of antidiuretic hormone (SIADH). Encephalopathy and focal or generalized seizures not associated with SIADH have also been reported. Cortical blindness and other CNS signs, including athetosis, ataxia, and parkinsonian-like symptoms, usually reverse after treatment is discontinued.
High-dose ifosfamide has been associated with a reversible encephalopathy of varying severity, which is a dose-limiting adverse effect. The most common manifestations include confusion, stupor, and mutism, rarely evolving into coma; less common features include seizures, hallucinations, personality changes, blurred vision, extrapyramidal symptoms, cerebellar symptoms, and urinary incontinence. EEG abnormalities are found in 65 percent of patients, and nonconvulsive status epilepticus has been reported. Although death or permanent disability can occur, the encephalopathy is usually reversible and can be treated with methylene blue.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here