Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Spontaneous isolated arterial dissection within the splanchnic circulation without associated aortic dissection or associated connective tissue disorder is a rare entity, but is being reported with increasing frequency. This most likely reflects improved imaging modalities, particularly abdominal computed tomography (CT), and increasing awareness of this condition rather than an increasing prevalence. Spontaneous isolated superior mesenteric artery dissection (SISMAD) was first reported by Bauersfeld in 1947. Due to the low prevalence of spontaneous isolated visceral artery dissection and the variety of clinical presentations, there is a lack of consensus regarding treatment guidelines. The goal of this chapter is to review the current literature on visceral dissection and help establish the best contemporary treatment guidelines.
The superior mesenteric artery (SMA) is the most common visceral artery affected, followed by the celiac artery. , In 1959, Ford reported an incidence of 0.06% SISMAD from a cohort of 6666 autopsies. Prior to 2001, only 46 cases of SISMAD were reported. As of 2016, that number has increased to more than 622 cases. , There is a lower incidence of spontaneous isolated celiac artery dissection (SICAD). There are even fewer reports of concomitant SISMAD and SICAD. Garrett et al. reviewed the literature through 2008 and reported 13 concomitant spontaneous isolated visceral artery dissections (SIVADs). Spontaneous isolated dissection of the splenic, hepatic, middle colic, and inferior mesenteric arteries have also been reported. Sixty-seven percent to 88% of isolated splanchnic artery dissection have been observed in males. , , , Typically, patients present in the fifth to sixth decade of life. , , , Sixty percent to 70% of patients with an aortic dissection present with hypertension; in contrast, only 30% to 40% of patients with isolated splanchnic dissections present with hypertension , , , , (see Ch. 83 , Aortic Dissection: Epidemiology, Pathophysiology, Clinical Presentation, and Medical and Surgical Management). Although isolated splanchnic dissections have been reported on all continents, the majority of cases have been reported in Korea, China, and Japan, suggesting a genetic predisposition.
There are multiple theories on the etiology of isolated splanchnic artery dissections and the cause may vary between different visceral vessels. Proposed etiologies of SIVAD include connective tissue diseases (Marfan syndrome, Ehlers–Danlos syndrome, Loeys–Diez syndrome), cystic medial necrosis, segmental arteriole mediolysis, Behçet disease, and fibromuscular dysplasia , , (see Ch. 141 , Aneurysms Caused by Connective Tissue Abnormalities). However, the majority of patients with SIVAD do not have any of these arteriopathies. Tobacco use, atherosclerosis, alcohol abuse, obesity, heavy weight lifting, and pregnancy have also been suggested as risk factors for SIVAD. , ,
In the majority of SISMADs, the dissection originates 1 to 3 cm from the SMA ostium. , , , At this position, the SMA transitions from a fixed retropancreatic position with an acute turn into a mobile mesenteric root. Hyperdynamic forces occurring at this transition point most likely result in this zone’s becoming the origin for most SISMADs. Park et al. conducted flow dynamic studies using computational fluid dynamic models that showed greater shear stress on the anterior convex portion of the SMA just distal to the ostium. These authors suggested that the etiology of SISMAD is more likely due to elevated shear stress at this location in the SMA rather than an underlying connective tissue disease or uncontrolled hypertension. Solies et al. also suggested high shear stress as the etiology of SISMAD due to the 1- to 3-cm origin of SISMAD. , This theory is analogous to Type II aortic dissections originating at the ligamentum arteriosum due to luminal stress at the transition from a fixed to an unfixed point. Another proposed hypothesis is development of primary hemorrhage in the media due to a rupture of the vasa vasorum, which then leads to dissection. Jia et al. have linked chromosome locus 5q13-14, which is associated with familial ascending aortic aneurysms and dissections, with SISMAD.
There is no clear etiology for SICAD. , In theory, the median arcuate ligament may represent a fixed transition point for the origin of celiac dissection. However, DiMusto et al. did not find any evidence of SICAD originating from the median arcuate ligament in their reported series of 19 SICAD.
There are several case reports describing the histologic findings of SIVAD. Common features include fragmentation of elastic fibers, loss of smooth muscle cells, myxoid degeneration, and atheromatous changes. No definitive correlation to arteritis, cystic medial necrosis, or other arteriopathies was identified. , , It appears that only a minority of SIVAD cases are associated with other arteriopathies.
The majority of current published literature on SIVADs comes from east Asian countries. Corrected for population, Korea appears to have the highest number of reports per capita, followed by Japan and then China. , This suggests, but does not prove, a higher prevalence of SIVAD in Asian populations.
The presentation of SIVAD varies greatly, and may include bowel ischemia, aneurysmal formation with or without rupture, or as an incidental finding on imaging. With modern CT and computed tomography angiography (CTA), a larger number of incidental SIVADs have been identified. In some series, as many as one-third of SIVADs are found incidentally. , Symptomatic SIVAD presents with abdominal pain in 90% of cases. Pain is described as severe and tearing in the mid-epigastric area with occasional radiation to the back. Other potential SIVAD symptoms may include nausea, emesis, melena, and diarrhea.
Abdominal pain out of proportion to physical findings suggests the diagnosis of mesenteric ischemia, which may be a clue to the diagnosis of SIVAD. Without the presence of bowel ischemia, abdominal pain is believed to originate from the visceral nerve plexus due to inflammation from the arterial dissection. It has been suggested that increased length of a SISMAD causes more periarterial inflammation and more severe pain. , , Persistent abdominal pain that does not resolve is more likely due to mesenteric ischemia rather than inflammation of neural tissue (see Ch. 133 , Acute Mesenteric Ischemia: Epidemiology, Pathophysiology, Clinical Evaluation, and Management). Patients with SIVAD have a wide variety of clinical presentations and, thus, multiple treatment algorithms have been proposed, which creates diagnostic and therapeutic management challenges for the clinician.
There are no specific laboratory tests for SIVAD; however, elevated lactate levels correlate with end organ malperfusion. In patients with SIVAD without mesenteric ischemia, leukocytosis as well as elevated C-reactive protein and/or erythrocyte sedimentation rate may be present.
Prior to 1975, all SIVAD cases were diagnosed by autopsy. The first case of SISMAD was reported in 1947. This patient had a SISMAD originating 2 cm from the SMA ostium that led to bowel infarction and death. The first documented survivor after treatment of SISMAD was reported in 1992, wherein angiography demonstrated the lesion after laparotomy for ischemic bowel. Digital subtraction angiography (DSA) supplanted conventional arteriography by minimizing contrast exposure and providing improved definition of distal mesenteric branch vessels compared to other imaging modalities. However, DSA is a more invasive test with limited ability to identify end-organ malperfusion when compared with contrast-enhanced CT.
CTA and contrasted-enhanced CT are now used to diagnose more than 95% of contemporary cases of SIVAD. Less commonly used are duplex ultrasonography (DU), DSA, and magnetic resonance angiography (MRA), respectively. Although DU is a reliable, inexpensive, and noninvasive modality for diagnosis and surveillance of mesenteric vascular disease, it may have low diagnostic sensitivity. Subhas et al. reported that DU only identified 56% of SISMADs. An intimal flap may be difficult to visualize sonographically and acute thrombosis of the false lumen may hinder discrimination of the false channel. Bowel gas and obesity also render DU studies difficult to perform successfully even with experienced technologists. MRA may demonstrate similar evidence of disease, but is both less efficient and less commonly available in the acute setting compared to CT.
Contrasted-enhanced CT is capable of identifying dissection flaps, false lumens, reentry tears, thrombus, intramural hematomas, aneurysm formation, and rupture of splanchnic arteries. When visualization of the dissection itself is limited, increased attenuation of the mesenteric fat may suggest the diagnosis. , , CT not only identifies the SIVAD, but also demonstrates the characteristics of the dissection such as its origin, length, and concomitant lesions. CT is also able to identify other markers of end-organ malperfusion. CTA should now be considered the gold standard for diagnosis of SIVAD.
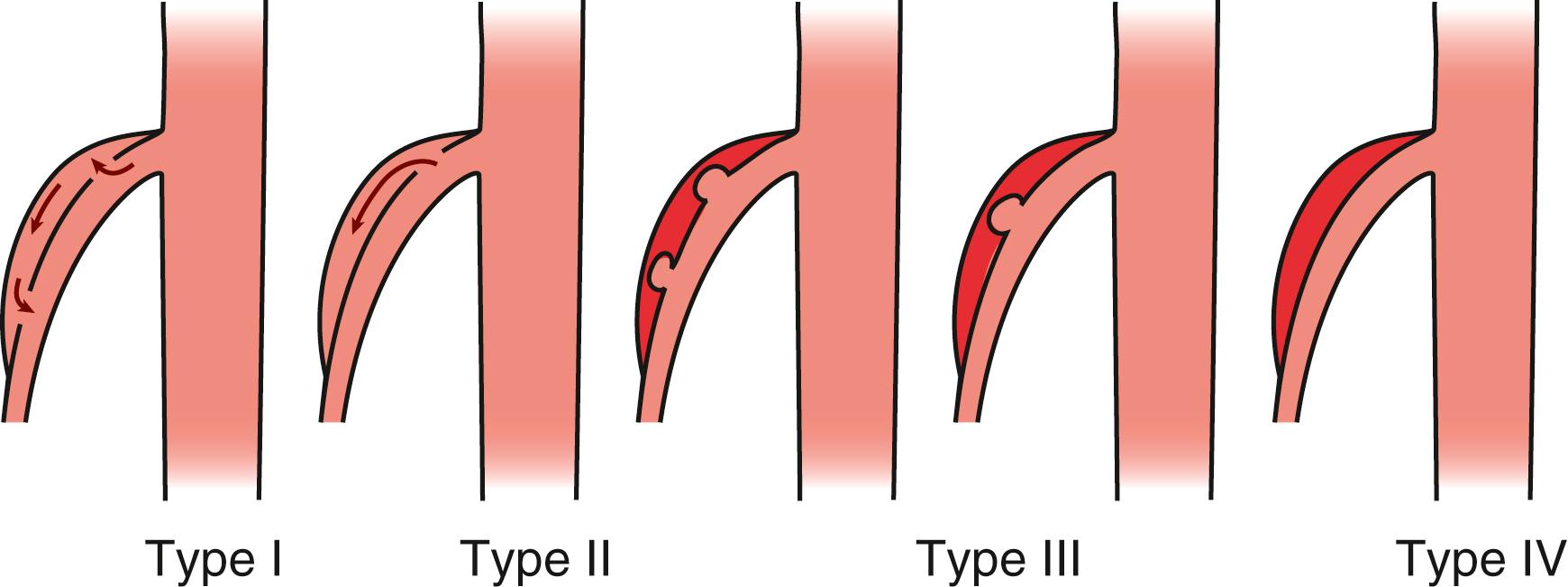
A number of classification systems have been developed for SISMAD. The goals of a classification system are to assist with risk assessment and determination of the best treatment algorithm for different types, extent, and characteristics of SISMADs. In 2007, Sakamoto et al. reported the first classification system for SISMAD. This group characterized SMA dissections based on CT appearance: Type I, patent false lumen with both entry and reentry; Type II, a “cul de sac”-shaped false lumen without reentry; Type III, thrombosed false lumen with ulcer defect; and Type IV, a completely thrombosed false lumen without ulcer ( Fig. 135.1 ). In Sakamato’s series, all Type I SISMAD were treated successfully with conservative measures such as anticoagulation and medical management. In patients with Type II SISMAD, the true lumen may be compromised by false lumen compression. They recommended that these patients be followed very closely for signs of mesenteric ischemia. Type III SISMADs had the highest rate of intervention in contrast to Type IV SISMADs that did not require any intervention. Although this series only included 12 patients, it was the first classification system that attempted to determine best treatment using image classification. A criticism of this system is the exclusion of patients with thrombotic occlusion.
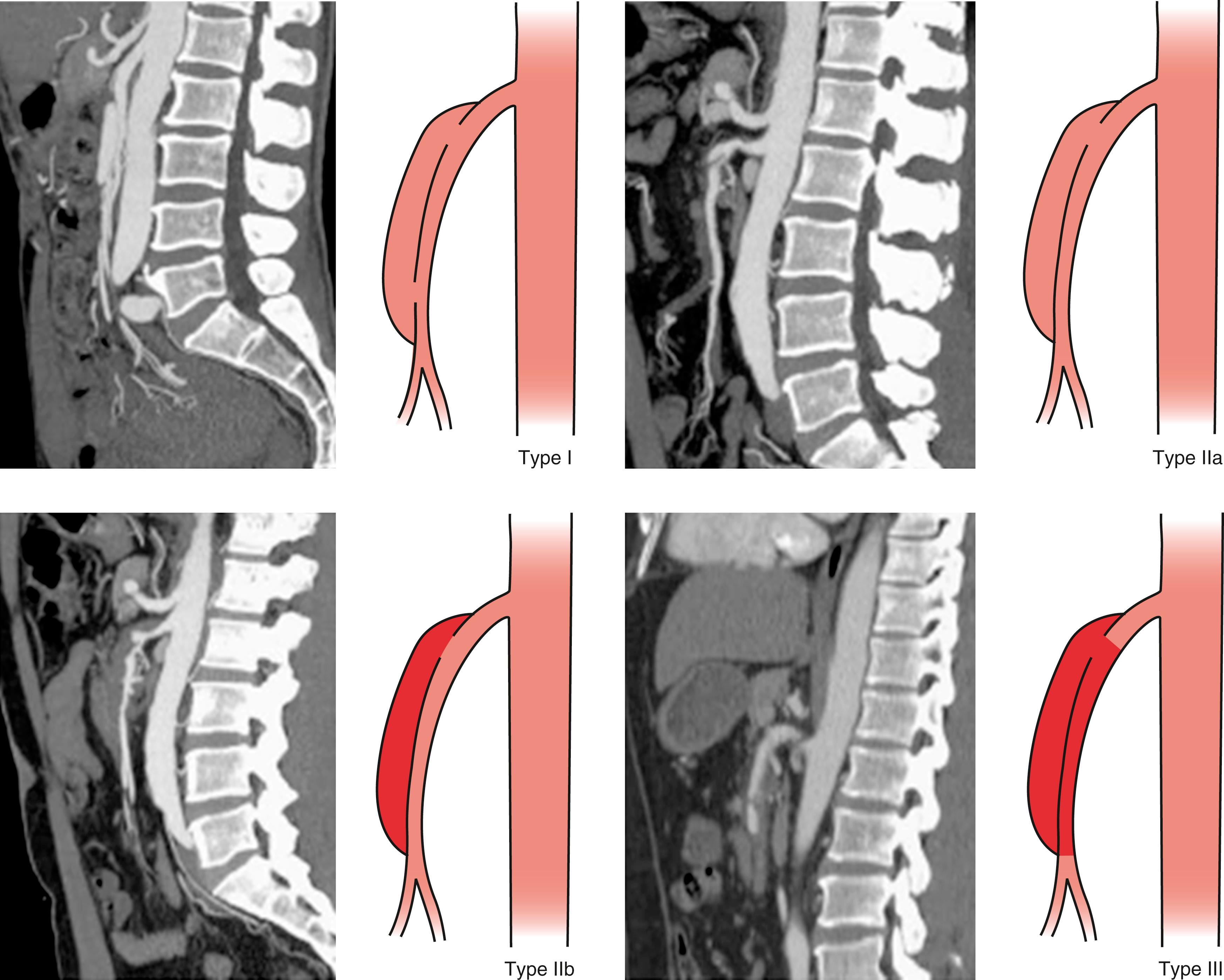
Yun et al. reported a classification system with three types of SISMADs. Type I, patent true and false lumen revealing entry and reentry sites; Type IIa, patent true lumen but no reentry of the patent false lumen; Type IIb, patent true lumen but no reentry of thrombosed false lumen; and Type III, dissection with occlusion of SMA ( Fig. 135.2 ). This classification system was based on a clinical series of 32 patients. The authors were able to correlate pain severity with length of dissection, but could not predict outcome or recommend treatment guidelines based on the classification system due to small sample size. Four of the 32 patients underwent intervention (one endovascular and three open surgeries) for persistent pain due to bowel ischemia, but the type of SISMADs in these patients was not described. Notably, due to the simplicity of Yun’s classification system, it has become the most commonly used.
Zerbib et al. described a more complex classification system by adding more categories to Sakamato’s scheme. Type V is aneurysmal dissection with stenosis of the distal segment of the SMA. Type Va is total thrombosis of SMA and Type VIb shows partial SMA thrombosis. These authors compared outcomes of reported cases in the literature using their classification system. Their results showed that Types II and III lesions required more interventions in contrast to Types I and IV. Unfortunately, they were unable to predict clinical outcome based on this classification system.
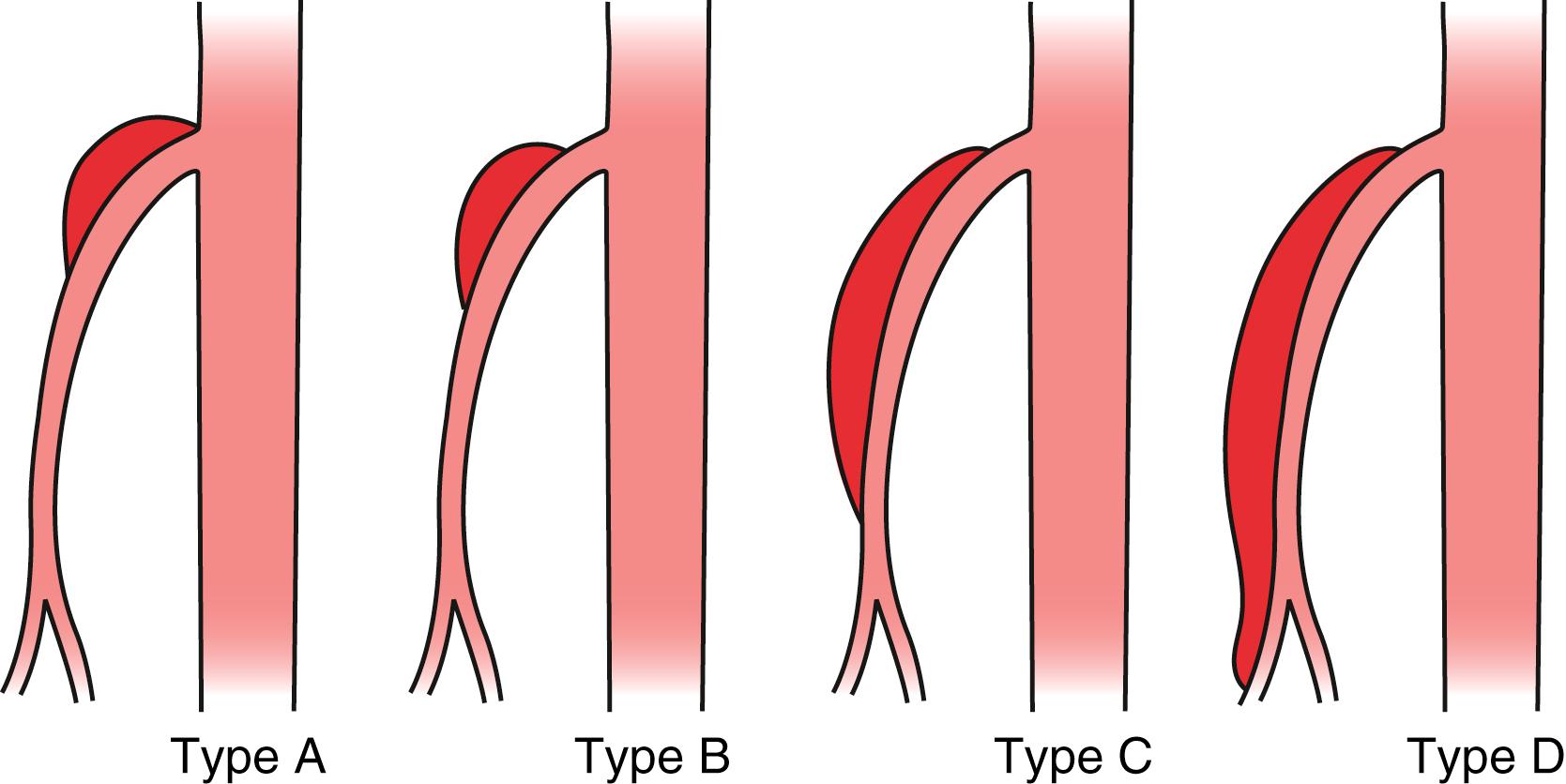
Luan and Li described a classification system based on the location and extent of the SMA dissection rather than luminal patency. These authors suggest four types of dissection. Type A is described as a dissection at the curve of the SMA and extending proximally to the SMA ostium. Type B is described as a dissection limited to the curve of the SMA. Type C is described as extending from the curve of the SMA distally, but not involving the ileocolic or distal ileal artery. Type D is described as extending toward the ileocolic and/or ileal artery ( Fig. 135.3 ). In this series, patients with Type B dissection were symptomatic less often in contrast to patients with Type D dissection, who presented with more severe pain. Unfortunately, management strategies are difficult to assess due to a sample size of only 20 patients. Type B patients were more likely to have successful treatment with conservative treatment compared with Type C and D patients.
Other classification systems have been proposed, but unfortunately have not added to improvement of clinical management strategies. Dissections of other visceral vessels have not been included in these systems. There is lack of wide acceptance of any one algorithm at this point in time.
Management of SISMAD or other visceral vessel dissection depends on the clinical circumstances including morphology of the dissection, resultant symptoms, and the patient’s clinical course. Little can be stated with authority at this time due to the low prevalence of reported SISMAD cases in the English medical literature worldwide and fewer cases including vessels other than the SMA. The majority of these cases have been reported within the last 2 decades, a time period with major advances in diagnostic imaging and therapeutic interventions.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here