Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
This chapter includes an accompanying lecture presentation that has been prepared by the authors: . ![]()
Spasticity is best understood as a motor impairment that develops slowly after damage to the CNS and results in uncoordinated movements, mass reflexes, spasms, and postural abnormalities.
Multiple spinal circuit abnormalities occur in spasticity owing to different types of injuries to the CNS, so the pathophysiology of spasticity is complex and varied.
Baclofen is an inhibitory medicine that acts as a selective agonist of γ-aminobutyric acid (GABA) receptors. It closes calcium channels and increases presynaptic inhibition.
Slow intrathecal infusion of baclofen distributes the drug in highest concentration along the spinal cord and thus lowers its central (i.e., brain) side effects.
Intrathecal infusion of baclofen by an implantable drug pump and catheter system has been proven to be effective treatment for severe spasticity caused by multiple sclerosis and spinal cord injury. Over the years its use has been expanded to spasticity due to traumatic brain injury, stroke, and cerebral palsy. It is also effective in some forms of dystonia.
Sudden disruption of intrathecal baclofen can cause withdrawal effects including marked increase of muscle tone, seizures, and hallucinations. A rare withdrawal syndrome of high fever, altered mental state, and muscular rigidity can lead to rhabdomyolysis and death.
Because the brain tissue and spinal cord act as a reservoir for intrathecal baclofen, the kinetics of drug delivery are slow, and it may well take 12 to 24 hours for an overdose of intrathecal baclofen to clear.
Chronic intrathecal infusion of baclofen has been extremely successful in the treatment of spasticity. Because baclofen does not cure spasticity, most treated patients need to take baclofen intrathecally for life. This long-term commitment to treatment is similar to maintaining shunt function in patients with hydrocephalus and may be equally rewarding or frustrating. To make proper use of this powerful tool, neurosurgeons need to understand the pharmacologic properties and distribution of intrathecal baclofen and how pumps are used to infuse the drug. Proper selection of patients requires an understanding of the physiologic features of spasticity and its clinical manifestations. Other neurosurgical methods for reducing spasticity are considered elsewhere; in particular, dorsal rhizotomy for spastic cerebral palsy in children is discussed in Chapter 270 .
Spasticity is a term that refers to a wide variety of motor problems and has numerous associated definitions. , It has been used to connote difficulty with coordinated movements and with spasms, rigidity, abnormal primitive reflexes, and hyperactive reflexes. Researchers studying spasticity tend to stress definitions that emphasize abnormal reflex responses, whereas clinicians tend to stress more global definitions, primarily related to impairment of movement. To further complicate the issue, many clinical syndromes associated with spasticity are caused by injuries at multiple sites in the neuraxis, and the pathologic mechanisms producing motor dysfunction are equally varied.
The narrow physiologic definition of spasticity is “a motor disorder characterized by a velocity-dependent increase in tonic stretch reflexes (i.e., muscle tone) with exaggerated tendon jerks, resulting from hyperexcitability of the stretch reflex, as one component of the upper motoneuron syndrome.” This velocity-dependent increase differentiates spasticity from other, non–velocity-dependent forms of rigidity that can be caused by contractures, dystonias, or Parkinson disease. In addition to hyperactive reflexes, spasticity can be associated with various signs and symptoms designated “positive” or “negative” : positive signs are produced by overactivity (i.e., disinhibition) of certain pathways as a result of injury to a specific part of the motor system; negative signs are caused by lack of function of the injured area. In spasticity, an increase in deep tendon reflexes and resistance to passive stretch of the limb are positive signs. Negative signs are weakness and loss of dexterity. It is the negative signs that usually create the clinically significant problems that are disabling for the patient. Increased muscle tone, clonus, and hyperactive reflexes occasionally interfere with initiation and smooth completion of a movement. Spasms, although often associated with hyperactive reflexes, are not a necessary concomitant of hyperreflexia and should be considered separately.
A broader and more insightful definition of spasticity emphasizes the movement disorder associated with the velocity-dependent resistance and reflex hyperexcitability, not simply its clinical signs. As has been pointed out by many investigators, the signs are not well correlated with the patient’s inability to perform voluntary movements. Decreasing tone and exaggerated reflexes produced by medications or surgery do not necessarily mean better function. A corollary of this broad view is that the secondary consequences of spasticity that result from changes in muscles and joints have to be treated in addition to the initial CNS causes.
The final common pathway to the muscle is the alpha motoneuron. Many mechanisms influence the output of the alpha motoneuron and may therefore exaggerate this neuron’s response to stretch. The muscles contain receptors called spindles, which are diagrammed in the center portion of Fig. 127.1 . A spindle is made up of intrafusal fibers attached to primary sensory endings. The primary sensory endings, which are extensions of the large, myelinated group Ia afferents, attach at the noncontractile equatorial region of the intrafusal fibers. The spindle organs are attached at both ends of the muscle mass and consist of extrafusal fibers. Because the spindles are attached in this way, they undergo the same changes in length as the overall muscle, and they “monitor” changes in muscle length. The spindles themselves are under control of the fusimotor efferents (i.e., gamma motoneuron system).
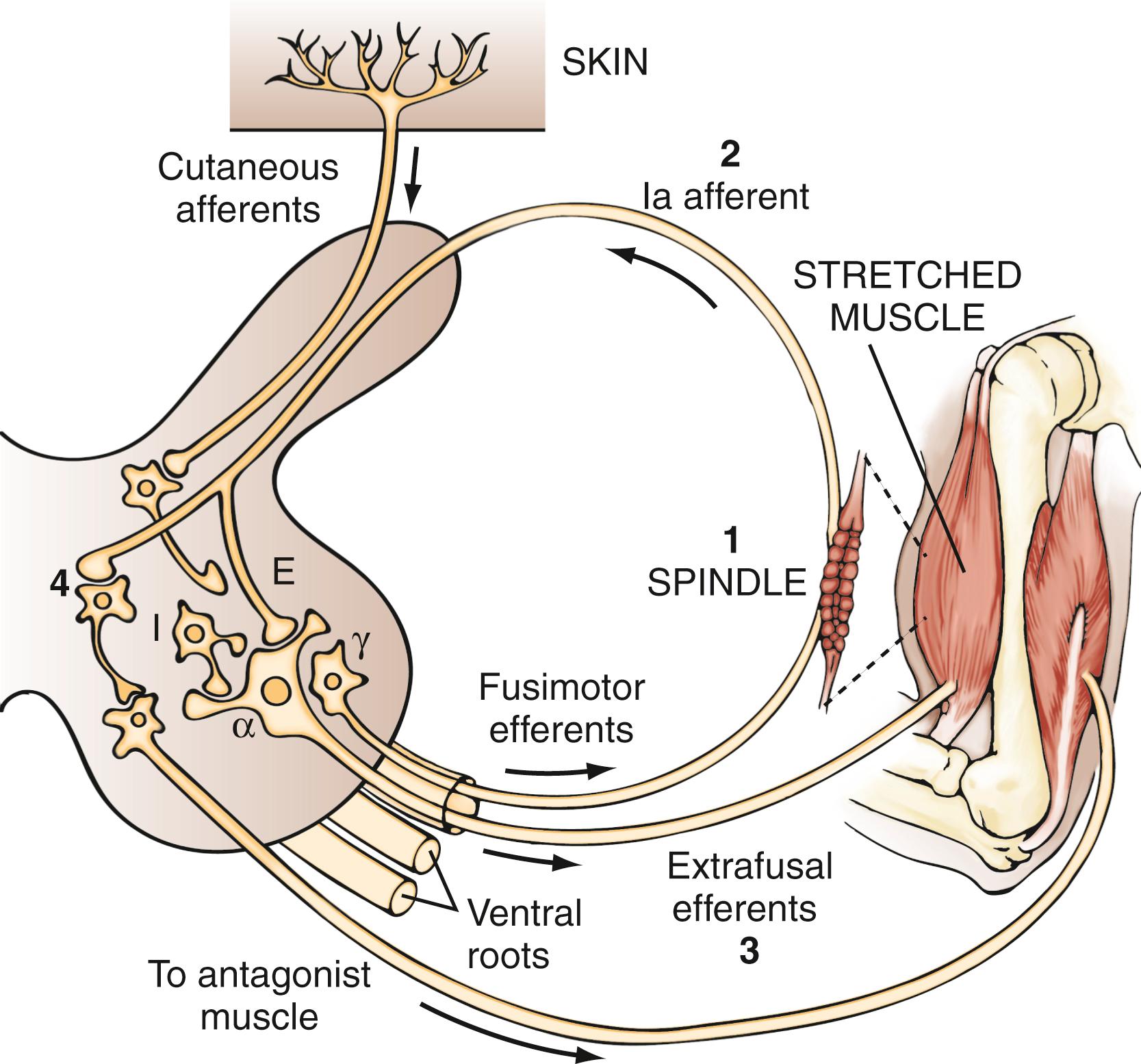
If the stretch reflex operated only in conjunction with the alpha motoneuron, voluntary movements would be difficult to make because a change in alpha motoneuron activity would cause contraction of the extrafusal fibers, which would shorten the muscle; as the muscle shortened, the intrafusal fibers of the muscle would slacken and would not monitor changes in length. Gamma motoneuron activity prevents this problem by shortening the intrafusal fibers, which assists movement by increasing activation of alpha motoneurons through type Ia feedback. Gamma motoneuron activity also increases reciprocal inhibition of the antagonist muscle.
When a muscle is stretched, it contracts in an attempt to regain its original length. This response can be broken down into five events, four of which are diagrammed in Fig. 127.1 :
Sensory impulses are generated as a result of stretching of muscle spindles.
The afferent volley ascends to the spinal cord.
It excites discharge of the alpha motoneurons of the same muscle.
It inhibits the motoneuron pools of antagonistic muscles through a disynaptic pathway.
It facilitates, probably monosynaptically, the motoneurons of synergistic pools (not shown in Fig. 127.1 ).
To understand how changes in these mechanisms lead to spasticity, the events that occur when a muscle is stretched must be traced. In a closed-loop system, it is impossible to identify the beginning and end of a sequence of events. For simplicity of presentation, the various mechanisms are discussed in the following order: Ia monosynaptic connection, Ia excitatory polysynaptic pathways, reciprocal Ia inhibition, group II pathways, decreased recurrent inhibition, alpha motoneuron hyperexcitability, gamma motoneuron hyperactivity, and decreased group Ib inhibition. Pierrot-Deseilligny, , Pierrot-Deseilligny and Mazieres, and Sehgal and McGuire discussed these mechanisms in detail. Sehgal and McGuire also provided a detailed explanation of the different electrophysiologic testing procedures that are used to deduce which mechanisms are underlying the spasticity.
When a muscle is stretched, the Ia afferent neuron transmits excitatory messages from its receptor, the muscle spindle, to the alpha motoneuron of the same muscle by means of a monosynaptic connection. It has been suggested that Ia discharge is normally reduced by presynaptic inhibition and that reduced levels of presynaptic inhibition could lead to an increase in the stretch reflex. For example, Calancie and colleagues argued that presynaptic inhibition is enhanced in patients in the acute stage of spinal cord injury but is reduced in the chronic stage. This reduction in presynaptic inhibition could give rise to an increased reflex response. There is also evidence of a decrease in presynaptic inhibition in paraplegic patients but not in hemiplegic patients.
The alpha motoneuron can also be affected by the Ia excitatory polysynaptic pathway ( Fig. 127.2 ). This can be shown by producing vibration-induced activity in group Ia fibers to generate tonic contraction of the vibrated muscle through Ia excitatory polysynaptic pathways; this is the tonic vibration reflex. This reflex can be increased or decreased, or both, in patients with spasticity. Facilitation of this pathway increases stretch reflexes.
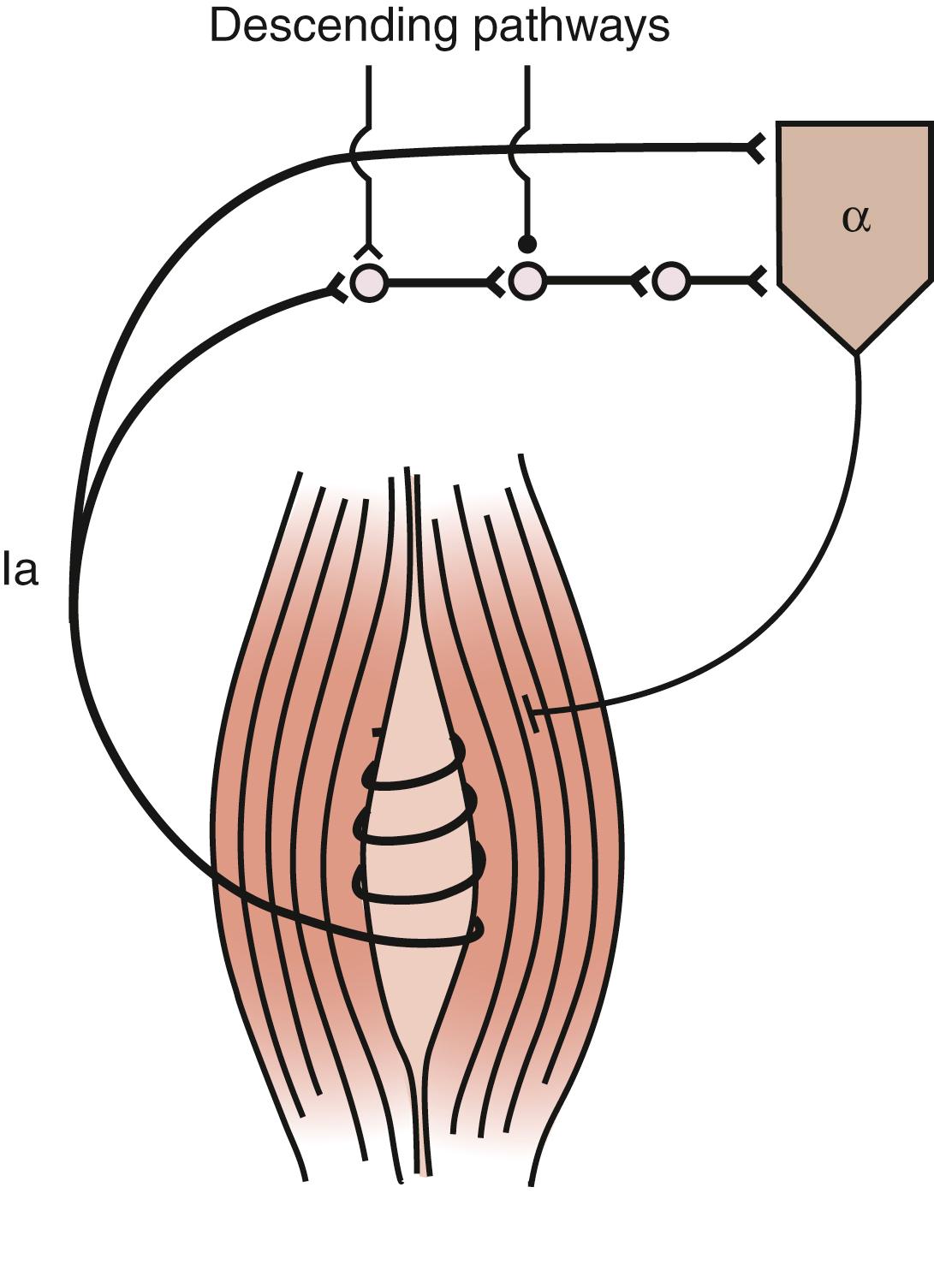
When a muscle is stretched, the reflex evoked in that muscle is normally accompanied by inhibition of the opposing muscle. This finding of reciprocal inhibition has had a pervasive influence on the understanding of movement control. It has been assumed that as a movement is made, stretch-related activation in the antagonist muscle is suppressed by reciprocal inhibition. Lack of reciprocal inhibition can lead to unwanted activation of the antagonist muscle and impede movement. The Ia inhibitory interneuron receives excitatory synaptic input from numerous descending pathways, including the corticospinal tract. Input from the descending tracts is combined with output from the Ia afferents of the contracting agonist muscle. If the Ia interneuron does not receive input from the corticospinal tract, reciprocal inhibition may become ineffective, a phenomenon observed in some patients with spasticity.
Group II endings (i.e., secondary endings), like group Ia endings, are located on intrafusal muscle fibers and are most sensitive to dynamic stretch. Their role in spasticity is not well understood.
Motor axons give off recurrent collaterals that activate Renshaw cells, which in turn inhibit alpha motoneurons, thereby creating a recurrent inhibitory circuit. Renshaw cells are excited or inhibited by supraspinal control. At rest, recurrent inhibition has complicated manifestations. In about 40% of patients with spasticity who are tested, there is no evidence of abnormal recurrent inhibition at rest. In patients with hemiplegia (most often from stroke) and in patients with spinal cord injury (most often from trauma), recurrent inhibition increases at rest. In contrast, patients with progressive paraparesis caused by hereditary spastic paraparesis or amyotrophic lateral sclerosis exhibit reduced recurrent inhibition.
During active movements in patients with spasticity, an increased reflex response may be caused by lack of inhibition of Renshaw cells. In healthy individuals, Renshaw cells are inhibited, which in turn inhibits the Ia inhibitory interneuron directed to the antagonist motoneuron. This allows reciprocal inhibition to function to suppress a stretch reflex in the antagonist muscles. Lack of descending control of Renshaw cells leads to impairment of voluntary movements and prevents modulation of control of the antagonist muscle, as has been shown by Katz and Pierrot-Deseilligny.
The alpha motoneuron receives excitatory input from the segmental and descending pathways. Lesions at numerous levels of the CNS may upset the delicate balance between excitatory and inhibitory input that influences the alpha motoneuron. There may also be a change in the intrinsic properties of motoneurons in spasticity. However, the influence of alpha motoneuron hyperexcitability is impossible to assess in humans because it requires knowledge of the firing level of motoneurons deprived of any sensory input. There is no evidence for or against the involvement of alpha motoneuron hyperexcitability in spasticity, although Noth argued that it does play a role.
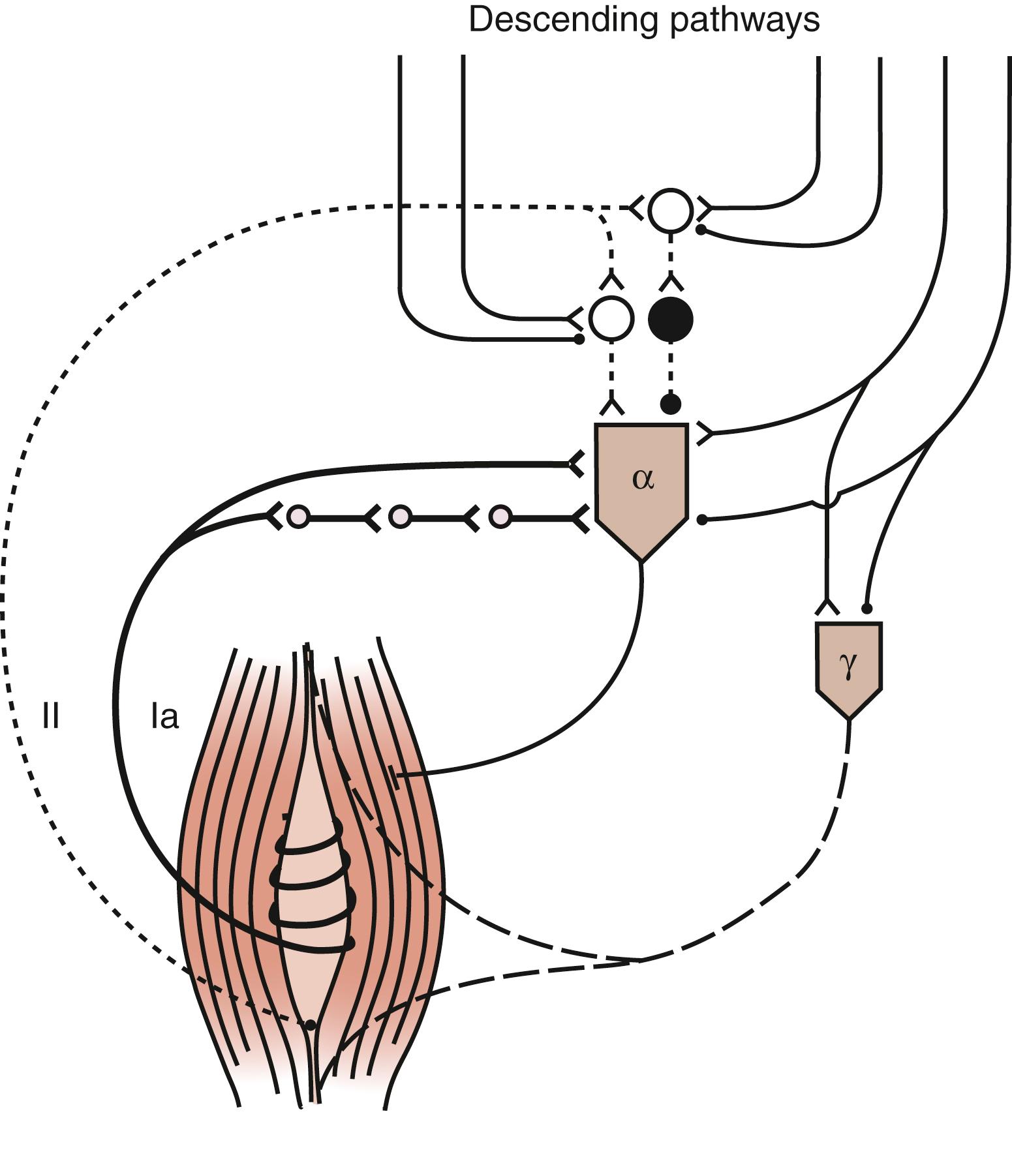
The gamma motoneuron system modulates the length of the intrafusal fibers and is therefore responsible for establishing the firing thresholds for group Ia and group II neurons ( Fig. 127.3 ). Incorrect threshold settings for the afferent neurons could lead to a hyperactive response. Evidence to support gamma motoneuron hyperactivity is derived from data from research with decerebrate cats, from selective blockade of the fusimotor system with local anesthetics, from the Jendrassik maneuver, and from comparison of the tendon jerk reflex and the H-reflex. Selective blockade of the fusimotor system has been used clinically in patients with spasticity. Injection of dilute procaine into the motor points of spastic and rigid muscles decreases muscle tone. This effect seems to support the view that spasticity is related to an imbalanced fusimotor system in that stretch reflexes are abolished as a result of block of the gamma motoneuron system, but voluntary muscle power is maintained by the output of unaffected larger alpha motoneurons. However, at a different site, intrathecal and epidural injections of local anesthetic did not reveal a link between increased fusimotor activity and spasticity. The strongest evidence against the gamma motoneuron hyperactivity hypothesis is the finding that the discharge rate of primary spindle endings is the same in individuals with and without spasticity. Increased gamma motoneuron activity should lead to increased sensitivity of the discharge rate of primary spindle endings in patients with spasticity. Although the gamma motoneuron hyperactivity hypothesis has been influential, the evidence to support it is indirect, inconclusive, and circumstantial.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here