Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Lymphoplasmacytic lymphoma (LPL) is defined in the WHO Classification of Tumours of Haematopoetic and Lymphoid Tissues, fourth edition, as a small B-cell neoplasm composed of lymphocytes, plasma cells, and plasmacytoid lymphocytes, which does not meet criteria for any of the other small B-cell lymphomas that may also exhibit plasmacytic differentiation. It typically involves bone marrow, but peripheral blood, lymph nodes, and spleen may also be involved. Waldenström macroglobulinemia (WM) is defined as LPL involving bone marrow associated with an IgM monoclonal paraprotein of any concentration, and is found in the majority of patients with LPL. Cases that express IgG or IgA heavy chain may fulfill criteria for LPL but are less common. Most LPL cases lack expression of a specific immunophenotype related to antigens commonly assessed in the workup of B-cell malignancies, including CD5, CD10, CD23, and CD103. Because of this feature and because plasmacytic differentiation may be seen in a number of small B-cell lymphomas, most commonly one of the subtypes of marginal-zone lymphoma, a specific diagnosis of LPL may not always be possible, and some cases are best diagnosed as a small B-cell lymphoma with plasmacytic differentiation with a differential diagnosis provided. However, the recent identification of the MYD88 L265P somatic mutation as a recurrent finding in greater than 90% of LPL/WM may aid in establishing this diagnosis with greater certainty.
LPL/WM is an uncommon lymphoid neoplasm, representing approximately 2% of non-Hodgkin's lymphoma cases diagnosed in the United States between 1988 and 2007 according to Surveillance, Epidemiology and End Results (SEER) registry data. The median age at diagnosis is 73 years with a male predominance. The overall age-adjusted incidence is 3.8 per 1 million persons per year. Incidence increases sharply with age and is higher in Caucasians compared with other races. A role for genetic factors in the pathogenesis of LPL/WM is suggested based on numerous reports of familial occurrence in case-control and larger cohort studies. A study from Asia found lower incidence rates in Japan and Taiwan compared with rates reported in the literature for Asians living in the United States, suggesting that both environmental and genetic factors are involved in LPL/WM development. Among environmental factors, chronic antigenic stimulation secondary to various autoimmune diseases or other inflammatory conditions has been implicated. An etiologic role for hepatitis C virus (HCV) has also been suggested; however, this has not been shown in all studies, and a large study with both serologic and molecular genetic methods for HCV detection found no association with LPL/WM.
The clinical manifestations of LPL/WM can be attributed to two main factors: the effects of the monoclonal IgM paraprotein and tissue infiltration by neoplastic cells. The monoclonal IgM paraprotein causes morbidity via several mechanisms related to its biochemical and immunologic properties, nonspecific interactions with other proteins, antibody activity, and propensity to deposit in tissues. The high concentration of monoclonal IgM molecules and their tendency to form pentamers can lead to serum hyperviscosity through binding of water and erythrocyte aggregation. Symptomatic hyperviscosity is seen in 10% to 30% of WM patients, and serum viscosity increases sharply at IgM concentrations of greater than 3 g/dL, with most patients manifesting symptoms at levels greater than 5 g/dL. Symptoms of hyperviscosity include headaches, visual disturbances, mental status changes, and, in severe cases, intracranial hemorrhage. Cryoprecipitation of the monoclonal IgM (type I cryoglobulinemia) may be seen in up to 20% of patients, with a minority of such patients exhibiting symptoms of Raynaud phenomenon, acrocyanosis, or, less frequently, renal manifestations. In other patients, the monoclonal IgM may behave as a type II cryoglobulin and demonstrate IgG autoantibody activity, leading to symptoms of purpura, arthralgias, renal insufficiency, and peripheral neuropathy. Other autoantibody effects of the monoclonal IgM against red blood cell antigens may result in cold agglutinin hemolytic anemia, whereas binding of peripheral nerve constituents may lead to a sensorimotor neuropathy. The latter manifestation is relatively common in WM, reported in 25% to nearly 50% of patients in some series. Peripheral neuropathy may also be mediated by non-autoimmune effects of the monoclonal IgM protein, secondary to fibrillar or tubular deposits in the endoneurium, amyloid deposition within nerve, or direct infiltration of nerve structures. The monoclonal protein may also deposit in various other tissues as amorphous aggregates leading to dysfunction of affected organs. Deposition of monoclonal light chain in the form of amyloid (primary AL amyloidosis) is much less common in patients with WM, but may lead to similar types of organ dysfunction.
Symptoms related to direct tissue infiltration by neoplastic cells is most commonly due to bone marrow involvement, which leads to peripheral cytopenias. At the time of presentation, the degree of anemia is typically more profound than other cytopenias, because the anemia in WM/LPL is multifactorial in nature and due in large part to increased plasma viscosity leading to inappropriately low erythropoietin production. Other factors contributing to anemia include hemolysis, plasma volume expansion, and gastrointestinal blood loss in patients with involvement of that site. Nodal and splenic involvement may be present, but bulky lymphadenopathy is uncommon and splenomegaly, if present, is typically mild to moderate in degree. Extramedullary and extranodal sites of disease involvement by LPL that have been reported include lung, soft tissue, skin, gastrointestinal and hepatobiliary tracts, kidney, and central nervous system (CNS). Pulmonary involvement, seen in less than 5% of patients, may be in the form of nodules, masses, diffuse infiltrates, or pleural effusions, and results in symptoms of cough (most commonly), dyspnea, and chest pain. Gastrointestinal disease may involve the stomach, duodenum, or small intestine, resulting in malabsorption, bleeding, or obstruction, whereas cutaneous involvement may result in chronic urticaria or in the formation of plaques or nodules. Direct infiltration of the CNS, known as Bing-Neel syndrome, is a rare complication of LPL/WM that is characterized clinically by a variety of neurologic signs and symptoms, including mental status changes, headache, motor dysfunction, vertigo, impaired hearing, and, in some cases, coma. Unlike in plasma cell myeloma, lytic bone lesions are not seen, and hypercalcemia is rare.
Lymphocytosis may be present, but the absolute lymphocyte count is usually lower than in chronic lymphocytic leukemia (CLL). Circulating neoplastic cells may have plasmacytoid features with clumped chromatin, eccentric nuclei, and moderately abundant basophilic cytoplasm, occasionally with a paranuclear hof ( Fig. 15-1 ). Red cell agglutination and rouleaux formation may be present in patients with serum hyperviscosity due to elevated IgM paraprotein.
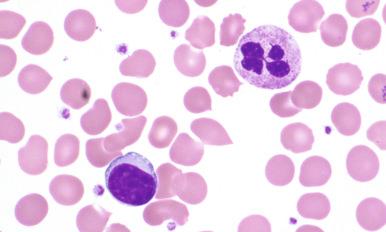
Bone marrow aspirate smears demonstrate lymphocytosis with a morphologic spectrum that includes small, round lymphocytes; plasmacytoid lymphocytes; and plasma cells ( Fig. 15-2 ). Varying patterns of bone marrow involvement may be observed in core biopsy specimens, with interstitial and nodular infiltrates most commonly reported. Less frequently, pure paratrabecular or diffuse patterns of marrow involvement may be seen. The lymphoid aggregates are composed mainly of small lymphocytes with variable numbers of plasmacytoid lymphocytes and plasma cells. Plasmacytic differentiation may be reflected by the presence of Russell or Dutcher bodies. Increased mast cells are almost always present in association with the lymphoid aggregates, and their identification may be facilitated by tissue Giemsa stain or CD117 immunohistochemistry; however, this finding is not considered specific for the diagnosis of LPL (see Fig. 15-2, F ).
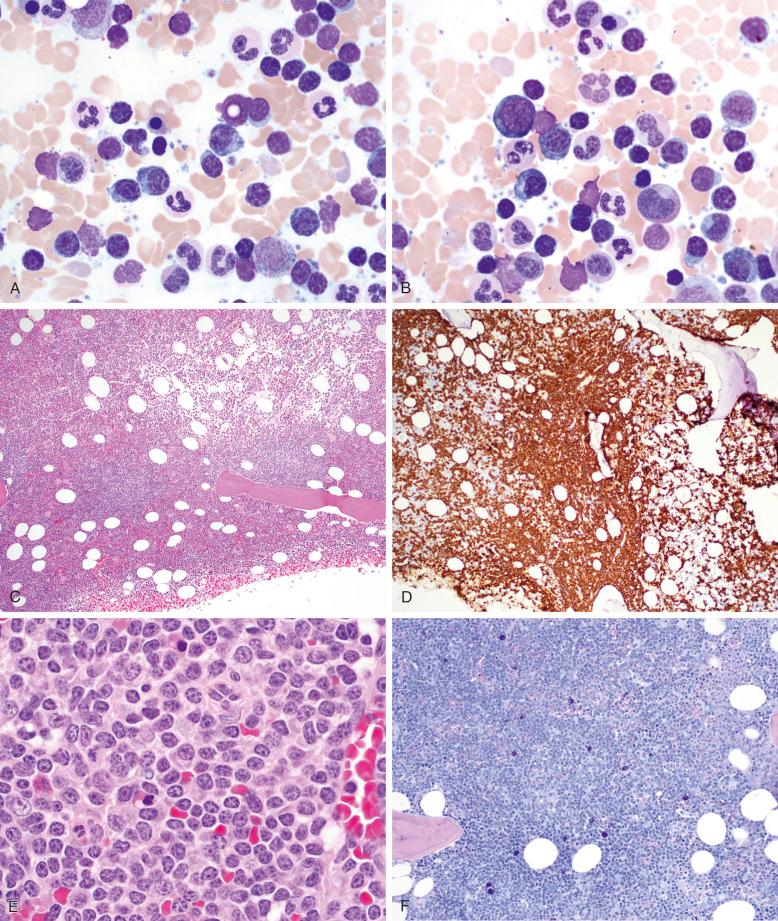
The pattern of bone marrow involvement may raise the differential diagnosis of other small B-cell neoplasms that more commonly involve the bone marrow, including CLL and follicular lymphoma. Distinction from these entities can be made readily on the basis of the immunophenotype of the neoplastic cells as noted later (see the section on immunophenotype later in the chapter). In addition, although nodular aggregates may extend to paratrabecular locations and infrequent LPL cases with predominant paratrabecular patterns of involvement have been described, these paratrabecular aggregates are generally not associated with linear growth along bony trabeculae or fibrosis, as seen in bone marrow involvement by follicular lymphoma (see Fig. 15-2, C ). Bone marrow involvement by marginal-zone lymphoma presents a more challenging distinction, given its shared immunophenotype with LPL. Interstitial involvement is more commonly seen in LPL compared with marginal-zone lymphoma. In contrast to bone marrow involvement by splenic marginal zone lymphoma (SMZL), intrasinusoidal involvement is rare in LPL. Assessment for the presence of the MYD88 L265P mutation may be helpful in difficult cases. Cases with prominent plasmacytic differentiation may raise the differential diagnosis of plasma cell myeloma, particularly the small lymphocytic variant with lymphoplasmacytic morphology. The latter is distinguishable from LPL on the basis of its CD45-negative, CD19-negative, CD56-positive immunophenotype and frequent cyclin D1 positivity by immunohistochemistry corresponding to an underlying CCND1 rearrangement detectable by FISH.
In the past, two main patterns of nodal involvement have been described. * In the classic pattern, there is subtotal architectural effacement with retention of small primary or enlarged reactive follicles and patent or dilated sinuses ( Fig. 15-3, A ). The interfollicular areas contain a relatively monomorphous infiltrate of small lymphocytes, plasmacytoid lymphocytes, and plasma cells, without prominent follicular colonization (see Fig. 15-3, B ). Only rare large transformed cells are present. Dutcher bodies, increased numbers of mast cells, or hemosiderin deposition may be present. Other cases demonstrate more complete architectural effacement with a vaguely nodular to diffuse polymorphous infiltrate consisting of small lymphocytes, plasmacytoid lymphocytes, and plasma cells, with greater numbers of large transformed cells resembling immunoblasts. The latter may be numerous in some cases, but should not form large aggregates or sheets, in which case a diagnosis of transformation to diffuse large B-cell lymphoma (DLBCL) should be considered. Clusters of epithelioid histiocytes may be conspicuous in the so-called polymorphous pattern, sometimes termed polymorphous immunocytoma in previous classification systems. In both the classic and polymorphous patterns, extracellular deposits of immunoglobulin, in the form of amyloid or amorphous Congo red–negative amyloid-like material, may be present, or crystal-storing histiocytes may be identified. Pseudofollicles or proliferation centers, as seen in small-lymphocytic lymphoma (SLL), are not a typical feature.
* References .
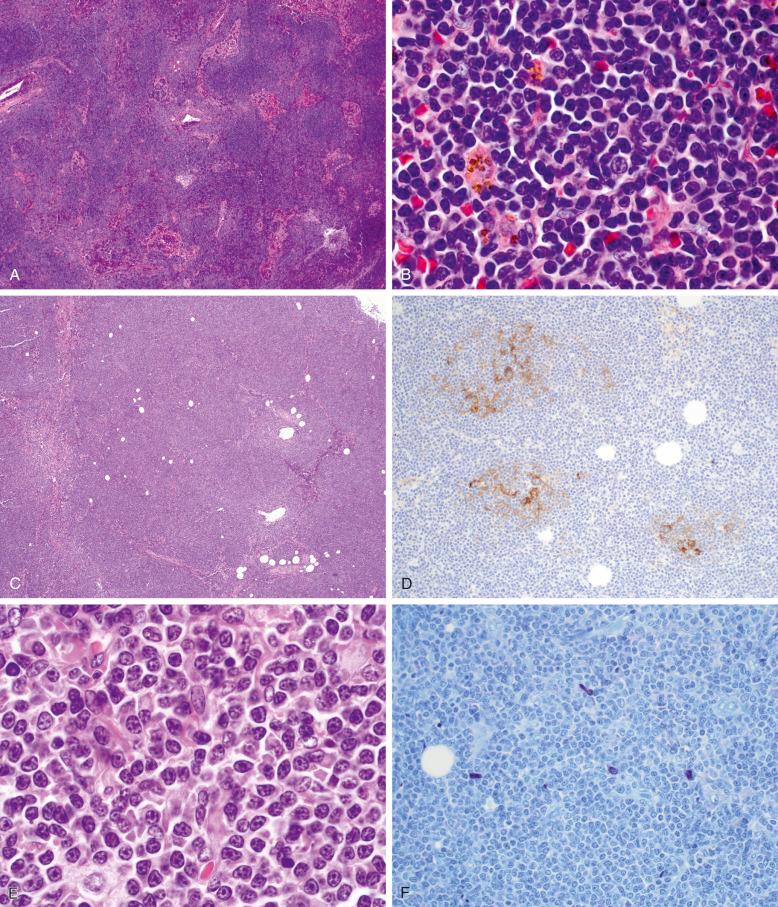
More recently, studies of MYD88 L265P mutation analysis in nodal lymphomas initially diagnosed as LPL, nodal marginal zone lymphoma (NMZL), and small B-cell lymphoma with plasmacytic differentiation have helped to refine the morphologic spectrum of nodal LPL. These studies show that some MYD88 L265P–mutated cases demonstrate complete nodal architectural effacement, a vaguely nodular growth pattern, variably prominent follicular colonization, or focal areas containing pale monocytoid or marginal-zone B cells (see Fig. 15-3, C-F ). Cases harboring the MYD88 L265P mutation had significantly more common bone marrow involvement, elevated serum IgM levels, and presence of a serum M component, suggesting that they represent true LPL cases despite the presence of morphologic features previously thought to be more characteristic of NMZL. Conversely, cases initially diagnosed as LPL with a polymorphous pattern of nodal involvement containing prominent histiocytes and occasional immunoblasts were all negative for the MYD88 L265P mutation, suggesting that they may represent a B-cell neoplasm distinct from LPL. Therefore, at this time, it may be best to classify the latter cases as B-cell lymphoma with plasmacytic differentiation until additional studies are undertaken to characterize them further.
Splenic involvement by LPL has not been well described, but older published series and illustrated reviews support the presence of nodular and diffuse infiltrates of lymphoplasmacytic cells involving the red pulp, in a similar distribution to other small B-cell neoplasms with a leukemic pattern of dissemination. The morphologic spectrum of the lymphoma cells, including small lymphocytes, plasma cells, and intermediate forms, is analogous to that seen in bone marrow and lymph node specimens ( Fig. 15-4 ). This cytologic appearance may give rise to the differential diagnosis of SMZL, which may demonstrate plasmacytic differentiation in some cases. Pathologic features favoring a diagnosis of LPL over SMZL include relative sparing of the white pulp with absence of a marginal-zone growth pattern, absence of monocytoid or marginal-zone–type cytology, and conspicuous plasmacytic differentiation that is usually readily apparent by both morphology and on immunohistochemical or in situ hybridization studies of tissue sections (see Fig. 15-4, B-E ). In addition, LPL patients typically have higher IgM paraproteinemia and more extensive disease involving the bone marrow or lymph nodes, with secondary splenic involvement resulting in a milder degree of splenic enlargement compared with SMZL. Other small B-cell lymphoma entities with diffuse red pulp involvement that may enter into this differential diagnosis include hairy cell leukemia (HCL) and splenic diffuse red pulp small B-cell lymphoma (SDRPSBL). Distinction between LPL and HCL is usually straightforward on clinical, morphologic, and immunophenotypic grounds. Although SDRPSBL cases may show subtle plasmacytoid features, they usually lack strong features of plasmacytic differentiation such as cytoplasmic immunoglobulin deposition by immunohistochemistry or in situ hybridization.
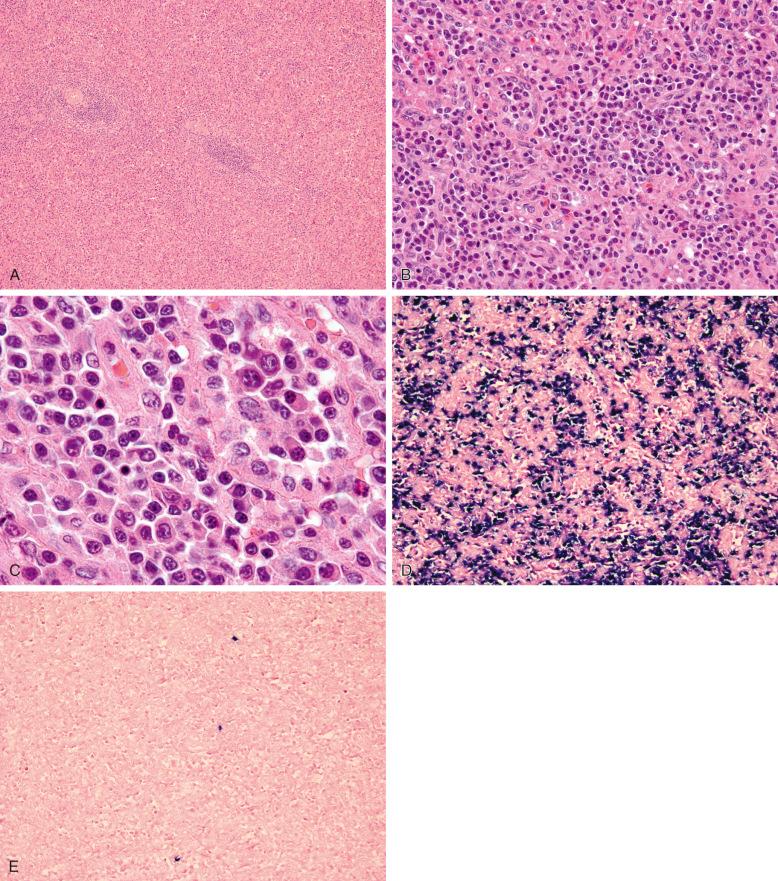
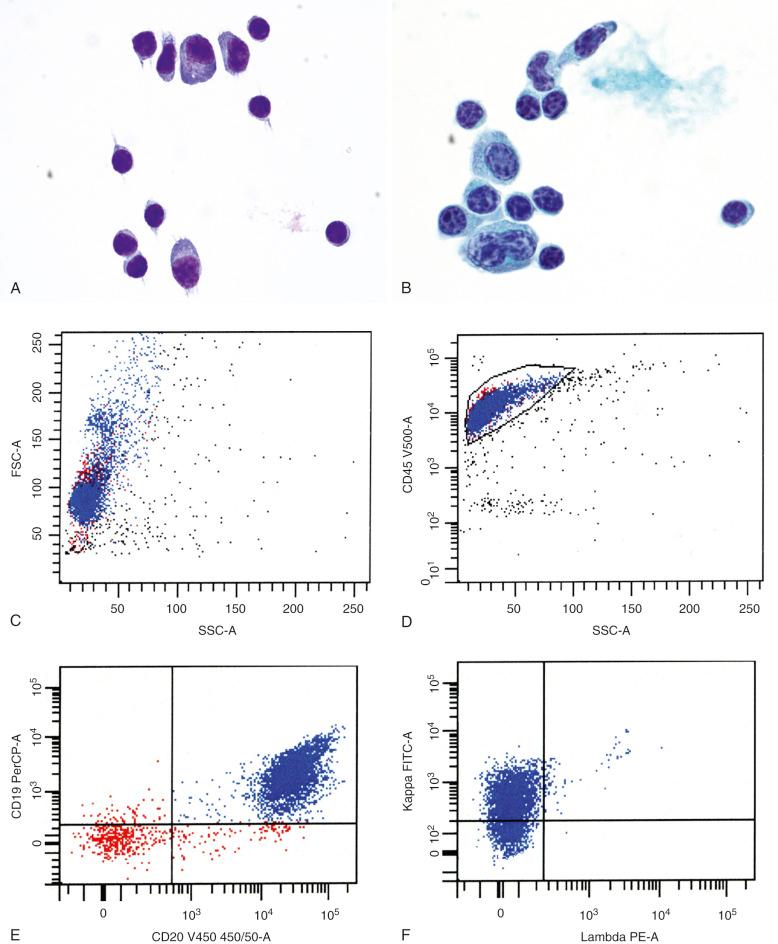
Common sites of extramedullary and extranodal disease were described earlier (see the section on clinical features earlier in the chapter). Among the few histologic descriptions in the literature, LPL involving extramedullary sites may show some features that overlap with extranodal marginal-zone lymphoma (EMZL), including the presence of nodular and diffuse infiltrates of lymphoplasmacytoid cells, focal clusters of monocytoid B cells, and presence of Dutcher bodies. However, in gastrointestinal sites such as the stomach and colon, lymphoepithelial lesions and colonization of pre-existing follicles have not been described. Skin biopsies have been reported to show interstitial, nodular, or diffuse dermal infiltration by lymphoplasmacytic cells with perinodal and periadnexal accentuation; focal epidermal ulceration may be present in rare cases. Hepatic involvement, reported in cases with splenic disease, has been reported to show expansion of portal tracts and sinusoids by small plasmacytoid lymphocytes. In cases of CNS involvement (Bing-Neel syndrome), cytologic evaluation of cerebrospinal fluid (CSF) specimens may show a lymphocytic pleocytosis consisting of plasmacytoid lymphocytes and plasma cells, similar to the spectrum of cell populations seen in other tissues ( Fig. 15-5, A-B ). Laboratory evaluation of such specimens via flow cytometry, electrophoresis, immunofixation, and, more recently, MYD88 L265P mutation analysis may help confirm the diagnosis (see Fig. 15-5, C-F ). In some cases, however, the paucity or absence of neoplastic cells in CSF specimens, as in other subtypes of lymphoma involving the CNS, may preclude a definitive diagnosis, requiring brain biopsy.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here