Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Shear wave elastography (SWE) assesses stiffness, not fibrosis. In addition to fibrosis, there are some factors or clinical conditions that may lead to an increase of liver stiffness (LS). They are known as confounding factors for fibrosis staging: liver inflammation, mostly gauged using transaminase values, which are indirect biomarkers; acute hepatitis; obstructive cholestasis; liver congestion; and infiltrative liver diseases.
The first report of an increase in LS in a patient with hepatic vascular congestion due to cardiac insufficiency dates back to 2008. The authors describe the case of a patient infected with hepatitis C virus after two heart transplants for severe ischemic cardiopathy followed by primary nonfunction of the graft. The patient had heart failure and showed a very stiff liver measuring 44.3 kPa at vibration controlled transient elastography (VCTE), with signs of cardiac hepatopathy but without liver cirrhosis at histology. One year after a third heart transplantation, a liver biopsy showed that there was a significant improvement of the cardiac hepatopathy, and the VCTE value was 3.8 kPa (i.e., within the normal range). The authors highlighted that vascular hepatic congestion can considerably increase LS up to values that are definitely diagnostic for liver cirrhosis. They also underscored that this increase is entirely reversible upon correction of cardiovascular dysfunction.
Since that report, several studies have investigated the role of the LS in patients with congestive heart disease and without a primary liver disease.
Any disease affecting the right heart may lead to an increase of the pressure in the right atrium, the inferior vena cava (IVC), and the hepatic veins. Hepatic veins do not have valves, thus an increase of pressure in the IVC directly affects the sinusoidal bed, causing centrilobular congestion and sinusoidal dilation. The liver is covered by a minimally distensible but nonelastic capsule; therefore hepatic congestion may lead to an increase of stiffness.
Heart failure is a major health problem with a considerable risk of morbidity and mortality. In patients with congestive heart disease, right-heart catheterization is the gold standard to measure central venous pressure (CVP). However, the procedure is invasive, not readily available, and not useful for following up patients. An indirect noninvasive parameter of the right atrial pressure (RAP), recommended by the guidelines of the American Society of Echocardiography, is the IVC diameter and its changes with breathing ( Fig. 9.1 ). The guidelines recommend IVC diameter be measured just proximal to the entrance of hepatic veins. IVC diameter ≤2.1 cm that collapses >50% with a breath suggests normal RAP (3 mmHg; range, 0–5 mmHg), whereas IVC diameter >2.1 cm that collapses <50% with a breath suggests high RAP (15 mmHg; range, 10–20 mmHg). If IVC diameter and collapse do not fit this paradigm, an intermediate value (8 mmHg; range, 5–10 mmHg) may be inferred or other indices of RAP should be integrated to downgrade to normal or upgrade to high RAP values. It is also underscored that in young athletes the IVC may be dilated in the presence of normal pressure and that the IVC is commonly dilated and may not collapse in patients on ventilators, so it should not be used in such cases for RAP estimate.
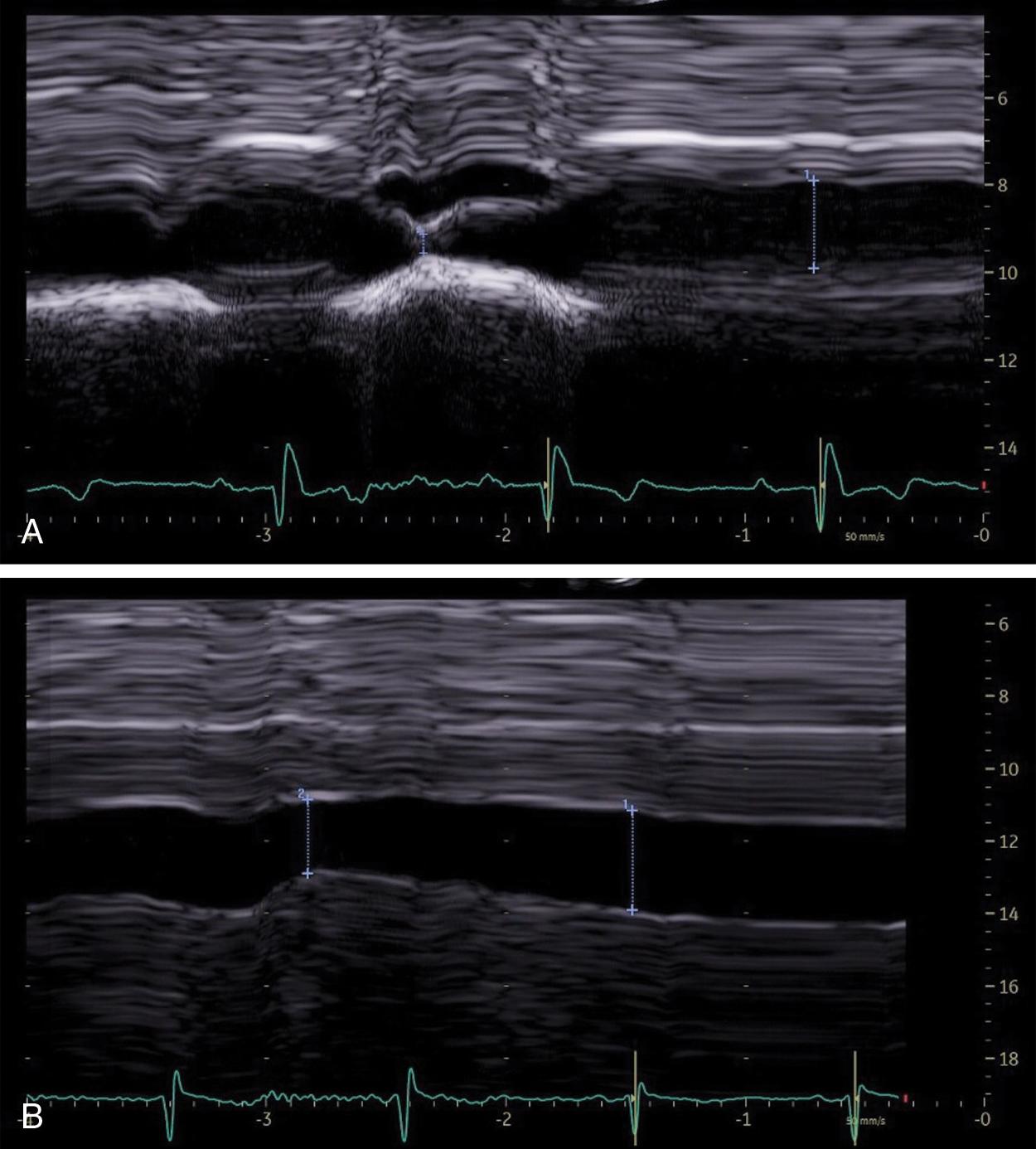
In patients with right-sided heart failure, the LS measurement could be a useful parameter for assessing RAPs and can be repeated over short periods of time.
Millonig and coworkers published the pivotal study in 2010 reporting that the LS is directly influenced by CVP. They showed that the clamping of the IVC in landrace pigs created a visible swelling of the liver and an increase in LS from 3.9 to 27.8 kPa. The reopening of the vein led to a rapid decrease in LS down to 5.1 kPa within 5 minutes ( P < 0.05). The reversible elevation of LS by increased venous pressure was highly reproducible in all five animals, suggesting that LS is directly controlled by the intravenous pressure in the absence of fibrosis or other causes of LS. For a direct correlation between hydrostatic pressure and LS, the authors also used the model of an isolated pig liver by clamping the portal vein, hepatic artery, and IVC distal and proximal to the liver. The IVC was then cannulated and the intravenous hydrostatic pressure was increased by infusion of isotonic saline solution. LS linearly increased with increasing intravenous pressures, and at a 36-cm water column, the maximum measurable LS with VCTE. (i.e., 75 kPa) was reached. The increase in LS was completely reversible and almost reached initial levels (5.5 kPa) within 5 minutes after resetting hydrostatic pressure back to a 0-cm water column.
Moreover, in a group of 10 patients with decompensated congestive heart disease that clinically recovered over a mean hospitalization interval of 7.2 days, they found a significant decrease of the LS from a median initial LS of 40.7 to 15.3 kPa. Thus treatment of cardiac insufficiency by diuretic therapy, concomitant weight loss, and clinical resolution of edema resulted in a decrease of initially elevated LS.
The decrease of the LS in patients with congestive heart disease who had a clinical improvement after treatment was confirmed in another small series of patients.
A study reported that LS could be an indirect marker of the RAP in patients with right-sided heart failure that was more accurate than IVC findings. In the study, both LS and IVC parameters were obtained within 3 hours before right-sided cardiac catheterization. There was a high correlation (r) between LS and RAP assessed with right-heart catheterization (r = 0.95), and the regression equation to predict RAP was −5.8 + (6.7 × natural logarithm of LS value in kilopascals). The authors found that an LS cutoff value of 10.6 kPa (with VCTE) identified RAP >10 mmHg with sensitivity and accuracy higher than the IVC parameters (sensitivity 0.85 vs. 0.56, accuracy 0.90 vs. 0.74, P < 0.05 for both).
The authors outline that IVC diameter and respiratory variation offer only semiquantitative assessment of RAP and may lead to erroneous inference, especially in patients with intermediate values, whereas LS gives a quantitative assessment of RAP and remains reliable even in patients on mechanical ventilation and with severe tricuspid regurgitation in which the use of echocardiography is usually limited.
However, it should be noted that 16 of the 105 (15.2%) patients who were screened were excluded. This is not a negligible percentage and may raise concern on the applicability of the technique in this setting. On the other hand, it should be highlighted that congestive heart disease may lead to organic liver disease, and this latter may be a confounder when the LS is used to noninvasively assess the CVP. For that reason it is of utmost importance to exclude cases with suspected organic liver disease in research studies.
Despite the improvements in the management of heart failure, there is still a high rate of hospital readmission due to heart failure. The same group of the previous study has assessed the prognostic value of the LS by VCTE in a series of hospitalized patients with heart failure. Of the 226 patients who were screened, 55 (24.3%) were excluded (37 for organic liver disease and 18 for invalid LS measurement). The LS was assessed before discharge in the remaining 171 patients and was stratified into three groups on the basis of the LS value: Group1: ≤4.7 kPa, corresponding to a RAP of 4.6 mmHg on the basis of the regression equation found in their previous study ; Group2: 4.7 to ≤6.9 kPa, estimated RAP 6.9 mmHg; Group3: >6.9 kPa, estimated RAP ≥7.0 mmHg. The authors found that the patients in Group3 were in the advanced New York Heart Association functional class and that they had a significantly higher risk of death or readmission to the hospital for heart failure than those in the other two groups. The LS value was able to predict cardiac events with a hazard ratio of 1.13 per 1-kPa increase in LS. LS showed a good predictive value for worse outcomes in patients regardless of severity of diastolic dysfunction. IVC diameter was also associated with the incidence of cardiac events; however, this parameter did not show significant predictive ability in the model that included both LS and IVC.
An LS value of 10.1 kPa had 0.73 sensitivity and 0.90 specificity for predicting worse short-term cardiac events. Notably, LS showed an incremental prognostic value when combined with previously established variables for predicting worse outcomes, including B-type natriuretic peptide. The results of this study indicate that the value of LS at discharge in patients with heart failure can be used as a reliable indicator of subclinical residual liver congestion, which reflects the severity of heart failure and adverse cardiac events, even in patients with optimized treatment and without visible edema or elevated liver function tests.
The prognostic value of the LS in patients with decompensated heart failure has been investigated in a few studies. Of note, the applicability of the technique in these studies is affected by the presence of other factors that could likely increase the LS, and invalid LS measurement or patients lost to follow-up should also be taken into account. In a study that excluded some 30% of patients for the previously mentioned reasons, 105 patients with acute decompensated heart failure were divided into two groups using an arbitrary LS value of 8.8 kPa by VCTE. In a median follow-up period of about 5 months, cardiac events (i.e., death or readmission to hospital) occurred in 54% of patients with LS ≥8.8 kPa and 25% of patients with LS <8.8 kPa ( P = 0.001). After adjusting for age, sex, and indices related to organ congestion, an LS ≥8.8 kPa was still significantly associated with cardiac events.
Another study assessed LS with VCTE both on admission to the hospital and the day of discharge in a series of 149 patients with acute decompensated heart failure. Overall there was a significant decrease of the LS during the hospitalization. An LS value higher than 13 kPa on admission and an LS value higher than 5 kPa at discharge was associated with an increased risk of 1-year all-cause death or readmission to the hospital. The authors highlight that LS increase is a nonspecific sign, therefore it would be difficult to use as the method for the differential diagnosis of liver dysfunction. However, in the clinical setting of an already established diagnosis of heart failure, after excluding other confounding factors, this limitation is not so relevant, and LS increase and its associations with negative prognosis in acute decompensated heart failure may be interpreted through two interrelated mechanisms of congestive hepatopathy: parenchymal congestion and congestion-induced fibrosis, both related to unfavorable outcomes.
Using a point shear wave elastography (pSWE) technique in patients with heart failure, it was reported that the changes in LS in patients with heart failure significantly correlated with changes in CVP in multivariate analysis and that an LS cutoff value of 7 kPa could predict a CVP >10 mmHg with 89.6% sensitivity and 87.5% specificity. , Another group reported that a high LS value on admission was an independent determinant of worse clinical outcomes in patients with acute decompensated heart failure.
All the published studies confirm that LS can be a marker of congestive heart failure; however, it is not yet clear what cutoff value should be used to define the risk of adverse cardiac events. In fact, this value ranges from >5 to 8.8 kPa. Of note, the “rule of 5” proposed by the Baveno VI consensus on portal hypertension has proposed that LS value with VCTE up to 5 kPa may exclude liver fibrosis.
It should also be emphasized that there is an interaction between the liver and the heart: heart failure and liver disease often coexist, because of systemic disorders and factors/diseases that affect both organs (alcohol abuse, drugs, inflammation, autoimmunity, infections). Moreover, there is a complex interaction between the heart and the liver: heart failure may lead to irreversible liver disease; conversely, liver disease may cause cardiac dysfunction and failure in the absence of other cardiovascular abnormalities. Therefore, in some cases, the increase in LS may be due to both liver congestion due to heart disease and liver disease, even when other causes of primary liver disease are excluded. Extensive fibrosis can be seen in chronic or severe cases of congestive hepatopathy. On this regard, it is worth mentioning that liver biopsy data were not available in the majority of the studies reported previously.
A study in a small series of patients with end-stage chronic heart failure who underwent left ventricular assist device implantation reported that the LS values were affected both by the central venous congestion and by the histologic changes of the liver. On the other hand, in patients with severe heart failure who required a left ventricular assist device, it has been observed that the incidence of major adverse events was lower when the LS was ≤12.5 kPa.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here