Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Salivary gland tumors represent 2% to 6% of all head and neck neoplasms (Ellis and Auclair, 2008). Though uncommon, they present with unique challenges in diagnosis and effective treatment (Silva and Kraemer, 1987). The vast majority (60% to 80%) of these neoplasms arise in the parotid gland, up to 11% in the submandibular gland, less than 1% in the sublingual gland, and approximately 23% in the minor salivary gland (Ellis and Auclair, 2008). The majority (80%) of parotid neoplasms are benign, and most (90%) are located in the superficial lobe (Ranchod, 2010). In contrast, half of all neoplasms in the submandibular gland and minor salivary gland and up to 90% in the sublingual gland are malignant (Ellis and Auclair, 2008).
The prediction of the benign or malignant nature of salivary gland lesions is difficult based solely on clinical history and physical exam because most are painless, firm, and nonfixed (Zheng et al, 1997; Layfield et al, 1987). Preoperative diagnostic studies, including sialography, ultrasound, computed tomography (CT), and magnetic resonance imaging (MRI), may be employed to define the location and circumscription of the tumors (Heller et al, 1993; Olsen et al, 2013). When employed preoperatively, fine-needle aspiration biopsy (FNAB) of salivary gland tumors can significantly aid in establishing a preoperative diagnosis and complements the frozen section interpretation. In general, FNAB has a sensitivity of 77%, specificity of 90%, and accuracy of 83% in distinguishing between benign versus malignant lesions (Arabi Mianroodi et al, 2006). An incisional biopsy, with the exception of the suspicion of lymphoma and sometimes the evaluation of minor salivary gland lesions, is generally contraindicated because of the possibility of facial nerve injury or incomplete tumor resection (Ranchod, 2010; Carvalho et al, 1999). As such, most surgeons rely on frozen section (FS) to determine the required surgical procedure (Heller et al, 1993).
Indications for FS in parotid can be broadly classified into three large groups that essentially serve the purposes of determining adequate tumor resection and avoiding unnecessary intensification of the surgical procedure:
Classification of diagnosis
Inspection of operative margins and specific analysis of an involved nerve or adjacent structures
Evaluation of the intraparotid and regional lymph node status
FS is often the first procedure that renders a pathologic diagnosis. In order to guide appropriate treatment, it is very important for the surgeon to know if the neoplasm is benign or malignant, lymphoma or carcinoma, and low grade or high grade (Olsen et al, 2013). The sensitivity and specificity of distinguishing benign versus malignant neoplasms during intraoperative consultation (IOC) is 43% to 100% and 92% to 100%, respectively (Arabi Mianroodi et al, 2006; Hillel and Fee, 1983; Wong, 2002). In recent years, concomitant with the increased utilization of FNAB, the need for FS has shifted from diagnosis to assessment of margins and deciding whether nerve or neck involvement is present (Seethala et al, 2005). In general, both FNAB and FS provide similar accuracy. Under certain circumstances, FS may be performed to support or dispute the FNAB diagnosis or to establish a diagnosis when the FNAB is nondiagnostic or when the FNAB had been reported as benign and there is still strong suspicion of a malignancy clinically (Seethala et al, 2005). It is important, however, to note that whereas most salivary gland lesions can be accurately diagnosed preoperatively by FNAB, false-negative results (4.7%) may occur and are mainly related to sampling, especially in cystic neoplasms (Layfield et al, 1987). Rare cases of false-positive malignancy interpretations on FNAB have also been reported, especially in highly cellular material with mild to moderate cytologic atypia (cellular pleomorphic adenoma) and lesions with metaplastic squamous epithelium (Warthin tumor, necrotizing sialometaplasia) (Layfield et al, 1987; Taxy, 1992). At least in the parotid, the false-positive rate for malignancy can range from 0% to 12.5% (Wong, 2002). It is important to appreciate that complete correlation of the FNAB and the FS diagnoses affords the most assurance of an accurate final pathology diagnosis. On the other hand, if the FNAB and the FS are significantly discordant, then the surgeon should be alerted to the diagnostic difficulty of the lesion, and management would be guided accordingly (Layfield et al, 1987).
The FS correlation with final diagnosis is high, especially for an interpretation of malignancy, because pathologists only render this diagnosis when they are certain, to minimize the consequences of error (Carvalho et al, 1999). The ability to determine a specific type of tumor on FS, however, has a reported range of 36% to 80% partly because of the relative rarity of these neoplasms and their overlapping features (Heller et al, 1993). The FS deferral rate has been reported to be approximately 9% (Hillel and Fee, 1983). Unless the FS diagnosis is certain, the pathologist may wish to defer the diagnosis, provided a manageable plan has been set with the surgeon (Layfield et al, 1987). As an example, the deferral of lesions that appear benign on FS but are clinically and intraoperatively suspicious, atypical, particularly cellular, or lack correlation with the FNAB diagnosis may safely decrease the FS error rate (Dindzans and VanNostrand, 1984). With added experience with FS interpretation for the parotid, the deferral rate is expected to decrease (Olsen et al, 2013).
The pathologist’s primary goal during IOC is to render a manageable, albeit preliminary, histologic diagnosis. It should be sufficiently informative for the surgeon to be able to elect the most appropriate surgical procedure. First and foremost, this requires open lines of communication, with both parties having a clear understanding of the therapeutic implications of a given diagnosis. In most cases the surgeon will choose between two surgical options, namely, management as a benign/low-grade malignant neoplasm or as a high-grade malignant neoplasm. Generally, for benign or low-grade malignant neoplasms, the end point is complete resection with negative margins. Except in very specialized circumstances, such as cervical involvement by recurrent pleomorphic adenoma, a neck dissection is not appropriate for a benign salivary neoplasm. In the case of low-grade carcinomas, a neck dissection is most often not performed unless there is clinical suspicion for cervical lymph node metastases. As one would expect, the high-grade malignancies deserve a more aggressive approach. Occasionally a benign or low-grade neoplasm may involve a portion of the facial nerve, which may necessitate resection of an upper or lower division of the nerve in a subtotal superficial parotidectomy. In general, attempts are made to preserve the facial nerve; however, most decisions concerning the facial nerve rest equally on intraoperative findings and preoperative nerve function (Olsen et al, 2013). In addition, to assure complete resection of high-grade malignancies, a neck dissection may be performed, even in the absence of clinically recognized metastases. Following surgery, many of these patients may also receive radiation therapy, possibly combined with chemotherapy. At times, the morphologic complexities of salivary gland neoplasia may preclude a specific or definitive intraoperative diagnosis. Under these circumstances, the pathologist at a minimum can guide the surgeon in choosing one of these two general therapeutic pathways whenever possible. In the rare event that an intraoperative diagnosis is entirely deferred to the review of permanent sections without any determination of biologic potential, it is prudent to have a direct discussion with the surgeon. Often in this circumstance, a procedure that is in accordance with the surgeon’s personal preoperative and intraoperative assessments may still be performed with the knowledge that in some cases the postoperative final diagnosis may dictate the need for an additional surgical intervention. Whether or not a satisfactory diagnosis can be made on the basis of the frozen sections, the pathologist still serves a very important intraoperative role in the determination of staging and margin status.
The role of the pathologist is to communicate with the surgeon, access the gross findings, and submit the appropriate sections for the IOC. Primary salivary gland tumors can have many overlapping gross and histopathologic similarities, and the distinction of benign and malignant tumors can at times be difficult. It is important to keep in consideration the primary site and the frequency of the tumors at that site ( Table 8-1 ). As stated earlier, parotid tumors are most commonly benign, whereas the majority of minor salivary gland tumors are malignant.
| Parotid | Submandibular | Sublingual | Minor |
|---|---|---|---|
| Pleomorphic adenoma Warthin tumor Basal cell adenoma Mucoepidermoid carcinoma Acinic cell carcinoma Adenoid cystic carcinoma Carcinoma Ex-PA Adenocarcinoma (NOS) Undifferentiated carcinoma |
Pleomorphic adenoma Adenoid cystic carcinoma Carcinoma Ex-PA Adenocarcinoma (NOS) Undifferentiated carcinoma Basal cell adenoma Mucoepidermoid carcinoma Acinic cell carcinoma |
Adenoid cystic carcinoma Carcinoma Ex-PA Adenocarcinoma (NOS) Undifferentiated carcinoma Basal cell adenoma |
Pleomorphic adenoma Adenoid cystic carcinoma Adenocarcinoma (NOS) Basal cell adenoma Mucoepidermoid carcinoma Carcinoma Ex-PA Undifferentiated carcinoma Acinic cell carcinoma |
At the time of the FS, the pathologist should be aware of relevant clinical history, such as age, duration of the mass, laterality, and the prior FNAB results, especially for diagnostically challenging cases. Information such as smoking history may be significant because smoking is commonly associated with Warthin tumor. The presence of a painful mass is more commonly observed in malignant neoplasms. Below the age of 1 year, mesenchymal tumors, specifically infantile hemangioma, are far more common than epithelial tumors. The most common childhood salivary gland tumor is pleomorphic adenoma, followed by mucoepidermoid carcinoma and acinic cell carcinoma (Silva and Kraemer, 1987). Most pleomorphic adenomas have been present for a considerable period of time. Rapid enlargement in a clinically stable pleomorphic adenoma may suggest the development of a carcinoma ex pleomorphic adenoma. Bilateral major salivary gland enlargement usually denotes benign neoplasms or lymphoma. Approximately 1% to 4% of salivary tumors may be bilateral and include lesions such as Warthin tumor, lymphoepithelial lesion, and acinic cell carcinoma (Jia et al, 2012).
The importance of a thorough gross evaluation cannot be overstated. It is the initial phase of diagnosis and serves as the foundation of microscopy. Only a careful and methodical gross assessment can ensure that the most informative tissue samples are submitted for microscopic evaluation. The pathologist should be able to answer these three important intraoperative questions regarding the lesion:
What is it? (diagnosis)
What does it involve? (stage)
Was it entirely removed? (margin status)
Knowledge of the type of surgical excision (e.g., capsular dissection, subtotal superficial parotidectomy, or total resection), the reason for the IOC, and the management implications of the FS diagnosis should be a priority before handling the specimen. Equally important is the knowledge of whether the surgeon had provided orientation because this information can potentially direct frozen and permanent section sampling and assure that the specimen is inked appropriately. After identification of the gross lesion and determining if residual uninvolved salivary gland parenchyma is present, it is common practice to serially section the specimen in a manner that does not affect the evaluation of a close margin.
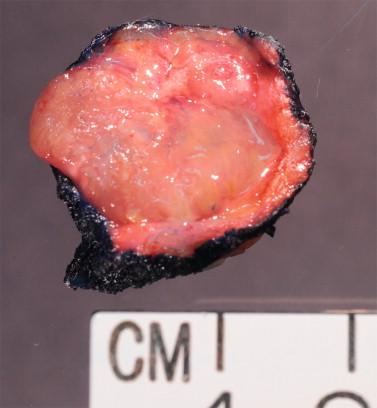
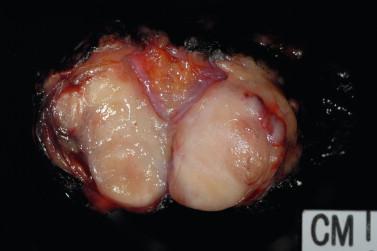
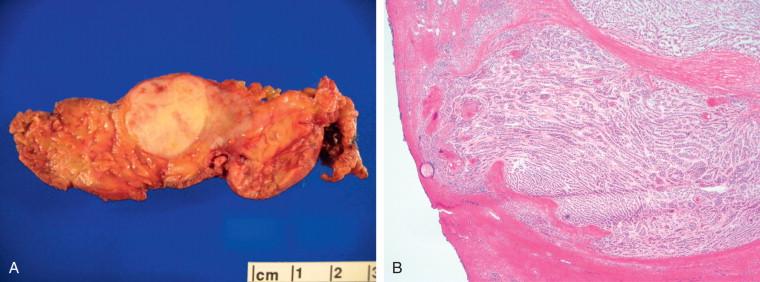
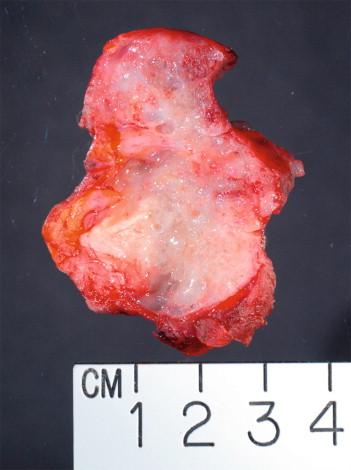
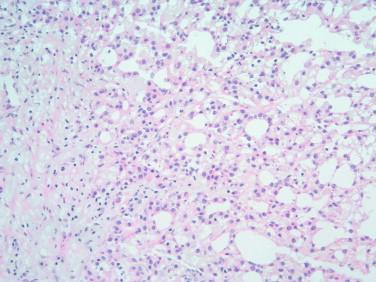
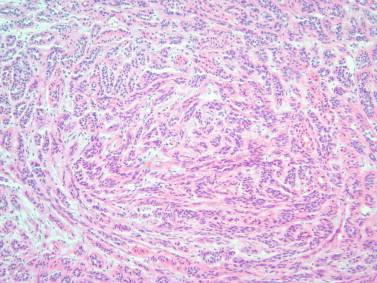
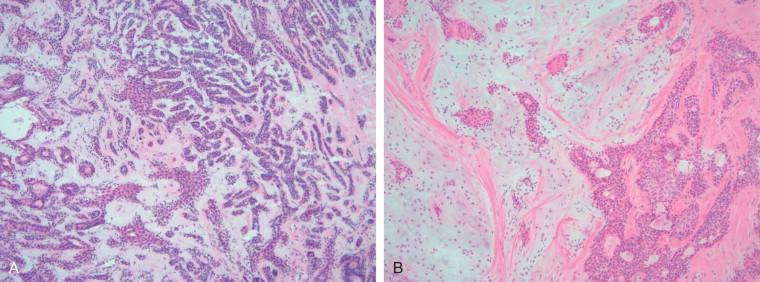
Assessing the interface, circumscription of the tumor, and identifying solid and cystic components of the lesion, along with areas of hemorrhage and necrosis, are important before a FS to assure that the sample is representative of the lesion. The gross appearance, however, does not generally determine the nature of the lesion. Salivary gland neoplasms can be solid, cystic, or have both components, and these features are not reliable indicators of biologic behavior. Benign processes (e.g., lymphoepithelial cyst, Warthin tumor, cystic hyperplasia) and a malignant tumor (e.g., mucoepidermoid carcinoma, cystadenocarcinoma) can contain a cystic component. Warthin tumor, in classic cases, is encapsulated, gray-tan, and rubbery and can demonstrate slitlike spaces containing oilylike fluid ( Figure 8-1 ). Similarly, pleomorphic adenoma ( Figure 8-2 ) tends to be well circumscribed and lobulated with a gray cartilaginous-like matrix. A well-circumscribed lesion, however, does not necessarily indicate whether the tumor is benign or malignant. Benign neoplasms (e.g., pleomorphic adenoma, oncocytoma, basal cell adenoma, myoepithelioma), low-grade neoplasms (e.g., epithelial-myoepithelial carcinoma), and metastasis (e.g., melanoma, squamous cell carcinoma) can appear to have well-circumscribed edges. Low-grade neoplasms can have minimal invasion into the surrounding parenchyma that cannot be noticed grossly ( Figure 8-3 ). Polymorphous low-grade adenocarcinoma, adenoid cystic carcinoma, and carcinoma ex pleomorphic adenoma can also have minimally invasive edges. Multifocal nodules are also not reliable indicators of benign versus malignant because pleomorphic adenomas and low-grade neoplasms, such as epithelial-myoepithelial carcinoma and acinic cell adenocarcinoma, can be multinodular. Mucoepidermoid carcinoma ( Figure 8-4 ) should be considered in cystic, mixed cystic, solid lesions, and solid lesions that exude mucoid material. Solid infiltrative tumors with hemorrhage and necrosis are likely to be malignant.
When possible, a section that includes the interface between tumor and normal salivary gland tissue should be submitted for frozen section evaluation. Furthermore, when applicable, a section closest to the surgical margin or an area suspicious for tumor infiltration into the surrounding tissue would allow for diagnosis and assessment of tumor around interface and margins.
The technical quality of the histologic sections can impact the effectiveness of the IOC. Extensive freeze artifact, incomplete tissue sections, and/or poor staining may result in erroneous, interpretations. More specifically, the distinction between low-grade salivary carcinomas and benign salivary neoplasms often rests upon the presence or absence of invasion, whereas the distinction between high-grade and low-grade salivary carcinomas is usually determined by cytologic features. Missing tissue limits the visualization of the tumor periphery or the freeze artifact, and poor staining significantly obscures the cytomorphologic details, so the true biologic potential of the lesion may not be recognized. Effort should be made to obtain the best possible frozen section histology, and there should be a low threshold for obtaining deeper tissue sections if the original section is not technically satisfactory or not representative of the tissue that has been frozen. In situations when the quality of the sections cannot be optimized, the gross specimen may need to be revisited to procure an additional sample. If, despite all attempts, the microscopic evaluation remains technically limited, then it may be necessary to issue an intraoperative diagnosis that reflects this limitation.
The accurate histologic diagnosis of salivary gland lesions is based on an assessment of the following parameters: tumor pattern, architectural pattern, cytoarchitecture, cytomorphology, and stromal features. In addition, some staging criteria and margin status are evaluated by microscopy. These tasks are accomplished by viewing a combination of low-, medium- and high-magnification fields. At low magnification, the pathologist can make a broad determination of tumor pattern, including the interface between the lesion, nonlesional tissues, and margins. One may also determine the relative proportions of lesional cells to stroma and general staining qualities of these components. More information regarding architecture, cytoarchitecture, and tumor periphery can be gained at medium magnification, building upon the impressions formed at low magnification. A detailed analysis of the cytoarchitecture, cytomorphology, and cellular-stromal interface should be performed at high magnification. A specific diagnosis should be made only when definitive histologic criteria for the lesion in consideration are met. If a definitive diagnosis is not possible, the pathologist can render a “manageable diagnosis” that will still allow the surgeon to perform the appropriate surgery. Consideration for additional therapy can be undertaken once the permanent sections have been reviewed and a final diagnosis has been rendered.
Much information regarding the tumor pattern is gathered during the gross evaluation. Microscopic examination serves to confirm the gross impression and to confirm the diagnosis. Questions that should generally be addressed include:
Is the tumor a single nodule or multinodular?
Is a tumor capsule present, and, if so, does the neoplasm invade the capsule?
For tumors that lack a capsule, is the interface well defined, indistinct, or frankly infiltrative?
Determination of the presence and relative amounts of solid, cystic, and mixed solid/cystic areas also begins macroscopically, but with histologic evaluation, various architectural patterns may be identified within the solid and/or cystic areas.
Some of the patterns that can be encountered in salivary neoplasia include tubular, nested, trabecular, reticular, microcystic, cribriform, solid, papillary, and micropapillary. Again, important considerations include:
Is there a single or dominant pattern throughout?
If multiple patterns are present, is there a constellation of patterns or single characteristic pattern that is often associated with a certain lesion or groups of lesions?
Is there just a haphazard mixture of patterns?
In some cases, the response to these questions can lead to a specific diagnosis. For example, recognition of a dominant microcystic pattern should raise suspicion for an acinic cell carcinoma ( Figure 8-5 ). Adenoid cystic carcinoma is usually limited to two or three patterns (i.e., tubular, cribriform, and/or solid), and these combinations assist in the diagnosis. Polymorphous low-grade adenocarcinoma demonstrates several different architectural patterns, but the finding of a targetoid or “eye-of-the-storm” pattern is very characteristic ( Figure 8-6 ). A haphazard collection of patterns in a well-defined and lobulated neoplasm would be the expected finding in many pleomorphic adenomas ( Figure 8-7 ).
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here