Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
The clinical approach to infections in patients undergoing hematopoietic stem cell transplantation (HSCT) involves an understanding of basic transplantation techniques, clinical syndromes, host defense defects at different times after transplantation, the natural history of individual infections, and the mechanisms underlying reconstitution of the immune system after transplantation. In general the dominant elements of infectious risks for bacterial, viral, fungal, and parasitic infections after HSCT depend on the pretransplantation exposure history (viral serostatus), whether the transplant is from an autologous or an allogeneic donor source, the intensity and content of preparative regimens, and the number of days after the transplantation under consideration. The distinguishing determinant of infectious risk between autologous and allogeneic grafts is the associated risk incurred by ongoing immunosuppression from prevention of graft-versus-host disease (GVHD), GVHD itself and its therapy; differing paces of humoral and cellular immune reconstitution that are highly associated with donor type also affect the risk. The time period after transplantation defines eras of differing transplantation complications and the evolution of the slowly resolving posttransplantation immunodeficiency: cutaneous and mucosal barrier breakdown, neutropenia, lymphopenia, hypogammaglobulinemia, or a combination of these. Many posttransplantation complications mimic infectious processes, and multiple infections may occur in the same patient at the same time. Therefore the patient undergoing HSCT should be examined in the context of pretransplantation infections, serologic profiles to document infection latency, intensity and content of “conditioning regimen,” available culture data from mucosal surfaces, contemporaneous transplantation complications in the patient's institution, current antimicrobial prophylaxis, presence of active GVHD prophylaxis, and the current degree and duration of neutropenia and lymphopenia.
HSCT involves the intravenous (IV) delivery of hematopoietic stem cells to a recipient whose hematopoietic and immune systems have been ablated or altered by a cytotoxic and immunosuppressive preparative regimen, commonly referred to as the conditioning regimen, given over the 4 to 10 days before HSCT. Hematopoietic stem cells are obtained from the patient (i.e., autologous) or from other individual(s) (i.e., allogeneic). Graft sources (stem cell collections sites) are bone marrow (BM; mostly for pediatric transplantations), filgrastim-stimulated peripheral blood (PB), or umbilical cord blood (UCB).
Autologous HSCT is used with the intent of curative or prolongation of survival in the treatment of multiple myeloma, lymphomas, rarely certain leukemias, some high-risk solid tumors (e.g., neuroblastoma, germ cell tumors), and autoimmune disorders (scleroderma). Its efficacy depends purely on (1) conditioning regimen intensity and (2) disease sensitivity to the conditioning regimen. Hematopoietic cell support only makes sure that the recipient will start and continue to reproduce entire blood cell repertoire after myeloablative conditioning (i.e., hematopoiesis). However, its morbidity and mortality also arise from regimen-related toxic effects and early infections.
HSCT is an option with curative potential in treatment of malignancies (e.g., acute and chronic leukemias), preleukemic disorders (e.g., myelodysplastic syndrome), lymphomas, multiple myeloma, hemoglobinopathies or thalassemias, disorders of BM failure (e.g., aplastic anemia, Fanconi anemia), severe immunodeficiency syndromes, and inborn errors of metabolism (e.g., osteopetrosis, chronic granulomatous disease, Hurler syndrome, inherited leukodystrophies, and other lysosomal disorders). It is currently used investigationally in treatment of diseases such as epidermolysis bullosa. Allogeneic HSCT has been feasible for selected human immunodeficiency virus (HIV)-positive patients with malignant and nonmalignant diseases since highly active antiretroviral therapy became standard treatment for HIV infection.
Allogeneic HSCT has the additional hazards of a higher risk of graft failure (prolonging neutropenia) or GVHD compared with autologous HSCT. Substantial improvements in the supportive care of severely immunosuppressed patients have evolved since the 1980s, and survival has markedly improved over time. As outcomes with transplantation improve, the use of transplantation as an option depends less on the availability of certain donor sources (i.e., whether there is an available matched donor) than in prior decades. Allogeneic donors can be related or unrelated and class I and II human leukocyte antigen (HLA) matched at loci of the major histocompatibility complex or mismatched. Matched unrelated adult volunteer donors (MUD) are available for more than half the general population (unfortunately still problematic for minorities), and unrelated UCB and HLA haploidentical related donors are used increasingly as a source of stem cells for transplantation reported by Center for International Blood and Marrow Transplant Research (CIBMTR) ( https://www.cibmtr.org/ReferenceCenter/SlidesReports/SummarySlides/Pages/index.aspx UCB). UCB has been shown to be an effective alternative for patients who lack a suitable adult donor, but its use is limited by finding a UCB unit with an adequate cell dose for the recipient's size and weight. Strategies such as transplanting two closely matched or ex vivo expanded UCB units have enabled transplantation for adults.
Graft source may be a contributing factor when considering the likelihood of infectious complications. Reported infectious events before engraftment have been significantly higher with BM versus peripheral blood stem cells (PBSCs) sources, probably related to longer periods of neutropenia before engraftment. With UCB grafts, engraftment is even further delayed, incurring more infectious complications before engraftment. Furthermore, immune recovery, in particular T cells, is also delayed after UCB HSCT. Therefore infections are significantly higher with UCB grafts compared with MUD. In a CIBMTR study the incidences of bacterial infection at 1 year were 72% and 59% for UCB and MUD, respectively. Incidences of viral infection at 1 year were 68% and 45%, ( P < .0001) for UCB and MUD, respectively. Moreover, bacterial and viral, but not fungal, infections were more common after UCB than mismatched unrelated donor (MMUD) ( P = .0009 and < .0001, respectively). In contrast, a French study showed the cumulative incidence of infection (72% vs. 57%) and mortality (8% vs. 3%) at 18 months was higher with MMUD grafts than UCB grafts due to higher rates of GVHD with PBSC HSCT, which contain a higher T-cell dose.
The conditioning regimen used to prepare the host is a major determinant of outcome because of variable host tissue injury and the potential for induction of prolonged immunodeficiency. Conditioning regimens may include immunosuppressive and cytotoxic chemotherapy alone or in combination with wide-field or total-body irradiation (TBI). Conditioning can damage mucosal surfaces, facilitating transmucosal entry of bloodstream infections (BSI). Indwelling IV catheters can also lead to disseminated BSI. Environmental exposure to airborne dusts and fungal spores can inoculate the sinuses or respiratory tree; therefore patients are required to stay in high-efficiency particulate air (HEPA)-filtered rooms. Prophylactic antibiotic therapies and nutritional changes can deplete commensal and potentially protective elements of the cutaneous and intestinal microbiome. The infectious risks for the patient undergoing HSCT are affected by transplantation complications, including the direct effects of this high-dose conditioning, such as mucositis, hemorrhagic cystitis, diarrhea, and hepatic venoocclusive disease (VOD); GVHD; and relapse of the underlying hematologic or oncologic disease.
Busulfan and melphalan are commonly used alkylating agents that are toxic to myeloid stem cells and mucosal and epithelial cells. Cyclophosphamide-containing regimens predispose patients to hemorrhagic cystitis. Fludarabine is less cytotoxic but intensely immunosuppressive and is often included in reduced-intensity conditioning (RIC) or nonmyeloablative (NMA) conditioning regimens. Antithymocyte globulin (ATG), which alters the function of or depletes T cells and other lymphocytes, can be used as conditioning regimen for aplastic anemia and, at times, for GVHD prevention or treatment. Serum sickness, a syndrome of fever, arthralgia, and rash, can occur with ATG or other xenoprotein therapy; it is treated with corticosteroid therapy. Antilymphoid antibodies, including alemtuzumab or rituximab affecting T and B cells, respectively, can induce prolonged and profound lymphopenia. Some GVHD prevention strategies include ex vivo graft manipulation (CD34 + or CD3 − selection) for T-lymphocyte depletion. A new approach is post-HSCT cyclophosphamide (on days 3 and 4 after grafting) to lyse proliferating alloreactive lymphoid cells, highly active in prevention of GVHD, made HLA haploidentical HSCT widely applicable. Cyclosporine, tacrolimus, mycophenolate mofetil, methotrexate, or sirolimus (and other agents) are often used for weeks to months after allografting to lessen the risks and severity of GVHD. Although all these may reduce the risks for GVHD, these maneuvers also substantially delay immune recovery, particularly in haploidentical (one HLA haplotype matched) or HLA-mismatched HSCT.
TBI may be administered as a single dose or more often “fractionated” in multiple doses given over several days. Diarrhea occurs in virtually all treated patients in the first week after irradiation. It may be treated symptomatically while stool studies are pending to rule out infectious causes. Severe oral mucositis occurs in many irradiated patients and is aggravated by prolonged neutropenia and the use of methotrexate for GVHD prophylaxis. As long as bleeding and oral inflammation do not compromise the patient's airway, mucositis is treated symptomatically. Keratinocyte growth factor (delivered clinically as palifermin) has proven activity in the prevention of oral and intestinal mucositis. Its use to limit mucositis may be of benefit in patients receiving highly toxic conditioning, including high-dose TBI, high-dose melphalan, or post-HSCT methotrexate. Palifermin has not been shown to reduce GVHD, infections, or mortality. Diffuse alveolar hemorrhage (DAH) is another complication of TBI that is associated with decreased pulmonary function after HSCT.
RIC or NMA regimens have been developed with the goal of permitting donor-derived hematopoietic and immunologic reconstitution. These lesser-intensity regimens may provide a somewhat weaker anticancer effect and rely on the graft-versus-tumor effects to eradicate underlying malignancies. Doses of TBI are usually not more than 2 Gy, compared with 12 to 14 Gy in fully ablative transplants. Fludarabine, ATG, or lower doses of cytotoxic drugs may be used with induction of extended immune suppression. This approach is often used for older patients or those with significant medical comorbid conditions. They are intensely immunosuppressive but less cytotoxic, resulting in less mucosal, enteric, and hepatic injury in the early weeks after transplantation, and perhaps because of that associated with fewer risks for GVHD.
In general, engraftment is most rapid, and thus neutropenia is briefest, when the patient and allogeneic donor are completely matched at all genetic HLA loci (most often considering only HLA-A, B, C, and DRB1). Similarly, identical twin (syngeneic) transplantations or those in which hematopoietic stem cells collected from the recipient (autologous) are associated with prompt neutrophil recovery. Allogeneic HSCT has the highest chance of prompt engraftment when fully HLA-matched sibling donor transplants are used, but less than 30% of intended recipients have a matched sibling donor available. Greater HLA mismatch augments risks for graft failure, acute and chronic (c)GVHD, and consequent prolonged immunodeficiency.
Killer immunoglobulin-like receptors (KIR) interact with HLA and other molecules (KIR ligands) that bind to and modulate the function of natural killer (NK) lymphoid cells. After HSCT, altered NK-cell function may modify risks for infection, GVHD, or malignant relapse. KIR ligands can be grouped based on their amino-acid sequence determining the KIR-binding epitopes, primarily in HLA-C and HLA-B molecules, but, owing to linkage disequilibrium, are not inherited coordinately with HLA. T-cell recovery and NK-cell proliferation and functional maturation are not altered by KIR ligand match or mismatch status. However, recipients of stem cells from unrelated donors with an activating NK-cell immunoglobulin-like receptor (KIR) (B/x) genotype have decreased infectious complications believed to be due to enhanced NK-cell function. The role of KIR genotype in donor selection before HSCT, especially for less relapse, is of research interest at this time.
Preventive strategies include protective isolation for reduced exposure to pathogens, enhancement of host immune reconstitution with hematopoietic growth factors, prophylaxis during high-risk periods with targeted antimicrobial chemotherapy, and suppression of subclinical infection with preemptive therapy, which is best facilitated by scheduled periodic surveillance. Prophylaxis or preemptive strategies are more effective than treatment after infection is established, and the mortality rate among patients with established infections continues to be high despite appropriate therapy. Prophylaxis can be tailored for patients who have a higher risk of reactivation, therefore screening for lifelong infections, including syphilis, tuberculosis, toxoplasmosis, HIV, hepatitis B virus (HBV), hepatitis C virus (HCV), and herpes simplex virus (HSV) should be tested at screening period. Of note, false-positive serum antibody results can occur after IVIG infusions, and false-negative serum antibody results can occur in the early months after transplantation. For this reason, viral serostatus, as well as serostatus of a few nonviral infections, are checked before transplantation so that the risk of reactivation is clear.
After mucositis has cleared and oral alimentation has resumed, oral therapy is the preferred route for antiinfective prophylaxis.
HC is a common complication that can lead to gross hematuria, clots, urinary retention, and impairment of renal function. Severe forms (grade III–IV) of HC are associated with increased nonrelapse mortality. Cystitis that occurs within the first weeks after marrow infusion usually is noninfectious in origin, caused instead by the administration of cyclophosphamide, especially at myeloablative doses in the conditioning regimen. Prophylactic measures include mesna (for binding and elimination of the alkylator metabolites of cyclophosphamide), forced diuresis, and continuous bladder irrigation. Supportive care for established cystitis may also necessitate large-bore catheter drainage or bladder irrigation and transfusions. Later in the posttransplantation period, severe GVHD, HLA-mismatched status (MMUD, UCB) and infection are contributing causes of cystitis. The majority of infectious agents inducing cystitis are viral, usually either the polyomavirus BK virus or adenovirus. Infection with HSV, CMV, the polyomavirus JC virus, human herpesvirus 6 (HHV-6), various bacteria, and Strongyloides occurs in lower frequencies. Although data are predominantly derived from studies in renal transplants, there is evidence that quinolone use (levofloxacin) does not have a preventive or therapeutic role for BK virus reactivation and disease. Neither BK viruria nor viremia confirm that BK virus is causal for hemorrhagic cystitis. Polyomaviruses are shed in the urine in many HSCT patients without clinical symptoms (see Chapter 144 ). Higher urine or detectable blood viral loads of BK virus may indicate a higher risk for hemorrhagic cystitis. No standard antiviral treatment is currently available for viruria caused by BK virus or adenovirus, although intravesicular cidofovir or low-dose IV cidofovir has been administered when ongoing hemorrhage or large mucosal clots persist despite continuous bladder irrigation.
VOD is a syndrome of liver toxicity that occurs at any time after the onset of the high-dose conditioning regimen, usually before day 30. It is characterized by painful hepatomegaly, 5% or greater weight gain, and hyperbilirubinemia (bilirubin levels >2 mg/dL). Severe VOD (sometimes called “sinusoidal obstruction syndrome” [SOS]) with marked jaundice or ascites leads to multiorgan failure involving the kidneys, heart, and lungs and high mortality. The only US Food and Drug Administration (FDA)-approved drug for severe VOD/SOS is defibrotide, which stabilizes endothelial cells by reducing endothelial cell activation and by protecting endothelial cells from further damage, resulting in the restoration of the thrombofibrinolytic balance. Anticoagulant or antithrombolytic therapies are mostly ineffective and associated with risk of serious bleeding.
Cyclophosphamide followed by TBI increases the risk for VOD because recurrent endothelial damage, along with reduced hepatocyte glutathione, increases vulnerability to radiation toxicity. Other risk factors for VOD include elevated aminotransferases before HSCT, mismatched or unrelated donor HSCT, and history of abdominal radiation therapy. Genetic predispositions in urea cycle enzymes (e.g., glutathione- S -transferase) may, in some patients, increase risks for VOD. Conditions that may mimic VOD include cholestasis in patients with septicemia; hepatic infiltration secondary to infection or tumor; pericardial tamponade; cytomegaloviral or other viral hepatitis; and intraabdominal inflammatory diseases, such as pancreatitis, peritonitis, or cholecystitis. In addition, early GVHD and cyclosporine-induced cholestasis are noninfectious causes of liver toxicity that may coexist with or mimic VOD. Diagnosis of VOD may be difficult in early stages when treatment is more effective, and ultrasonographic assessment of hepatic portal venous flow and newer techniques, such as ultrasound point shear wave elastography, showed shear-wave velocity as significantly elevated in patients with VOD after an HSCT prospective study in 134 patients who received allogeneic HSCT between 2011 and 2016. Transjugular liver biopsy and measuring corrected hepatic sinusoidal pressure gradient during the procedure with a relatively safe profile (three major complications in 141 HSCT patients) can be important to make correct diagnosis. Hepatotoxic and nephrotoxic drugs should be avoided in patients with VOD.
Acute GVHD (aGVHD) is a major, life-threatening complication, developing in 40% to 80% of patients after allogeneic transplantation. The risk for developing GVHD is higher in older patients and with partially matched or unrelated donor HSCT and is associated with the higher doses of donor T cells infused with filgrastim-mobilized peripheral blood grafts. In aGVHD donor, T lymphocytes mount an immune attack against the recipient's tissues that is amplified by proinflammatory cytokines. The favorable antineoplastic impact accompanying GVHD is called the graft-versus-leukemia (GVL) effect. T cells and possibly NK and other lymphoid cells mediate GVL effects after allogeneic HSCT by the production of inflammatory cytokines and by direct target lysis.
The primary target organs of aGVHD are skin, liver, and gastrointestinal tract (GIT), therefore its common clinical manifestations include rash, cholestatic hepatitis, nausea, vomiting, and diarrhea. aGVHD can be lethal, and one of the major clinical problems remains to predict who will have severe GVHD in real time. Recently, models to predict patients at risk of poor outcomes have been developed using both clinical and laboratory data.
Effective immunosuppressive agents for the prevention of GVHD include calcineurin inhibitors (cyclosporine or tacrolimus) with or without methotrexate, mycophenolate mofetil, and sirolimus; they are usually started before transplantation in T-cell replete HSCT. T cells can be removed ex vivo (negative selection or CD34 + cell selection) or in vivo (e.g., ATG, alemtuzumab use in conditioning) from the graft; however, these methods may result in an increase of some complications (e.g., graft failure, infections such as CMV and Aspergillus , and relapse). In addition, posttransplant cyclophosphamide, used especially after haploidentical HSCT, has become an effective method for prevention of both acute and cGVHD. GVHD itself can compound and prolong post-HSCT immunodeficiency. The corticosteroids or other immunosuppressive drugs used as treatment of GVHD may impair phagocytic function and directly worsen lymphopenia and cellular immune deficiency. Patients with acute and cGVHD have splenic dysfunction and thus an added risk for infection with encapsulated bacteria, such as Streptococcus pneumoniae, Neisseria meningitidis, and Haemophilus influenzae. T-cell depletion of the donor graft is less associated with GVHD but more with infection and sometimes with much-delayed immune recovery.
Clinical hepatitis in HSCT recipients can range from fever accompanied by abdominal pain to fulminant illness. Infectious hepatitis must be distinguished from several common noninfectious causes, including liver dysfunction related to the “conditioning regimen” (VOD), aGVHD, cholestatic liver injury related to sepsis, and chemical hepatitis related to either drugs or hyperalimentation.
Clinically important viral hepatitis syndromes that occur after transplantation include acquisition or reactivation of infection with HBV, HCV, varicella-zoster virus (VZV), adenovirus, HSV, CMV, and HHV-6. Reactivation of HBV is more likely than HCV to result in fulminant hepatitis, although this occurs in only a minority of infected patients. Disseminated VZV and adenovirus infections may manifest as elevations in serum aminotransferase levels; these elevations can precede the appearance of other disease manifestations by several days. In rare cases liver biopsy with viral culture and polymerase chain reaction (PCR) assay are needed to establish a diagnosis of severe hepatitis in the early postengraftment period. Viruses such as hepatitis G virus and transfusion-transmitted viruses are not known to influence the outcome of HSCT. Hepatitis E virus, transmitted by the fecal-oral route, is now increasingly identified as a pathogen in HSCT, as a cause of both acute and chronic hepatitis. Screening strategies for donors are not established, and diagnostics should include PCR. Therapy with ribavirin has been effective.
HBV (surface antigen [HBsAg], surface antibody [HBsAb], and core antibody [HBcAb]) and HCV serologic profiles are tested in donor and recipient before HSCT. Patients who are HBsAg positive or HBcAb positive are at high risk for reactivation, whereas the risk is extremely rare in those with HBsAb positive. Pretransplantation imaging studies or liver biopsy may be needed to evaluate HCV-seropositive patients with abnormal liver enzyme levels or tender hepatomegaly. Donors and recipients with positivity for HBsAg and HBcAb should be tested for viral load with PCR studies for HBV DNA before transplantation because the risk for HBV hepatitis can be reduced by treatment to lower a detectable viral load. Lamivudine, entecavir, and tenofovir are commonly used to suppress HBV replication (see Chapter 145 ). Current stratifications of risk are discussed in Chapter 306 . The more potent activity of entecavir has led to support for its use in preference to lamivudine, particularly in patients with HBsAg present in the serum at the time of immunosuppression.
A transplant from an HBV-infected donor can be used if no alternative donor is available or if the intended recipient is already seropositive. HBV can be transmitted from an HBsAg-positive (or, less likely, an HBcAb-positive) donor to a recipient who is either HBV naïve or HBsAb positive but HBcAb negative. The risk for transmission is low when an HBV-positive donor has an undetectable viral load and/or has nonreplicative HBV. If the recipient is HBV naïve before transplantation, the subsequent infection is more likely to have clinical consequences. If the transplant can be delayed, then HBV vaccination of the recipient or use of HBV immune globulin or both may reduce the likelihood of hepatitis after transplantation. HBV immunity can be transferred from an HBsAb-positive donor to an HBV-naïve recipient. Through adoptive immune transfer, HBV infection can be cleared from an HBsAg-positive recipient by transplant from an HBsAb-positive (i.e., immune) donor, which generally follows a period of hepatitis (elevation of liver functions).
After transplantation the following recipients should be monitored periodically with PCR testing of HBV DNA viral load: (1) those with liver enzyme elevation suggestive of activation of HBV from latency, (2) those with transplants from HBV-infected donors, and (3) those with known infection before transplantation. High HBV viral load (>10 5 copies/mL) is the most important risk factor for clinically apparent reactivation in recipients positive for HBsAg. Among recipients in whom PCR studies of HBV DNA yield positive results persistently after transplantation despite treatment, the risk for fatal liver disease may be up to 12%.
HCV-positive donors and recipients should undergo RNA viral load testing. Although best avoided, but if there are no other options, an HCV-infected individual can serve as a donor. In contrast to HBV, however, the rate of transmission of HCV from an HCV RNA-positive donor approaches 100%. Interferon (IFN) can be used to suppress HCV replication in donors, but its limited efficacy, systemic and hematologic toxicity, and delayed response may not contain active HCV to render a donor suitable for donation. Direct-acting antiviral (DAA) therapies are highly active in HCV and have rendered IFN-containing regimens obsolete for almost all HCV genotypes, although the evidence comes from non-HSCT patients (see Chapter 47, Chapter 154 ). In a study, no patient receiving DAA therapy after transplantation discontinued HCV therapy, and efficacy was 85%. However, dose adjustments per renal function and drug interactions may be needed.
In contrast to HBV, which induces immune-mediated hepatitis, HCV can induce direct hepatotoxicity. Hepatitis most commonly occurs 2 to 3 months after transplantation; severe hepatitis is rare. There is no strong evidence to support HCV as a cause of increasing incidence of VOD, especially when conditioning regimens are not risk factors for VOD, or GVHD. However, even without direct hepatic failure, HCV seems to be associated with a higher nonrelapse mortality in the first year. Beyond 10 years, the long-term complication of HCV infection is cirrhosis. No data have demonstrated a correlation between hepatitis C genotype and type or severity of liver disease after transplantation. Because of the myelosuppressive effects of IFN and other antiviral agents, their use in the treatment of hepatitis C after HSCT is limited. However, in a study with 20-year follow-up, antiviral treatment (IFN with or without ribavirin) showed a strong trend to reduce the risk for severe liver complications (odds ratio, 0.33; 95% confidence interval, 0.11 to 1.03; P = .058). In the study of 195 patients, 33 died, among which 6 died from liver complications. The cumulative incidence of severe liver complications (death from liver failure, cirrhosis, and liver transplantation) was 11.7% at 20 years after HSCT. A new American Society for Blood and Marrow Transplantation task force recommends long-term liver function test (alanine aminotransferase) monitoring in all HCV-positive recipients/donors. HCV RNA monitoring should be measured in all patients at entry into care, and monitoring of viral load should be performed in patients receiving HCV treatment or liver dysfunction.
Infectious pneumonias must be distinguished from noninfectious pulmonary complications after HSCT, which can include pulmonary edema, pleural effusion, alveolar hemorrhage, radiation injury (pneumonitis or fibrosis), drug reactions, adult respiratory distress syndrome, idiopathic pneumonia syndrome, cytolytic thrombi (causing multiple peripheral lung nodules), obliterative bronchiolitis, bronchiolitis obliterans with organizing pneumonia, and cGVHD. Management of noninfectious pneumonias requires that lower respiratory tract infection (LRTI) be ruled out or recognition of its coexistence. Their pathophysiologic processes may be distinct: Some syndromes may be more likely to respond therapeutically to high-dose corticosteroid therapy.
Diffuse alveolar hemorrhage (DAH) begins with dyspnea and alveolar infiltrates, with progressively bloody return during bronchoscopic examination and alveolar lavage. Although DAH definition requires no infection, infection-associated alveolar hemorrhage and DAH are related clinical syndromes with similar clinical presentation, risks, and associated high mortality.
The syndrome usually occurs in the second to fourth weeks after HSCT, generally before platelet engraftment and perineutrophil engraftment. Because DAH occurs around neutrophil engraftment, tissue damage due to leukocyte influx into lungs is suggested. Delayed engraftment, thrombocytopenia, infection, toxic effects of drugs, TBI, intensely cytotoxic regimens, and solid malignancy have been implicated as risk factors. Very-high-dose corticosteroids are recommended, but DAH remains highly mortal.
Idiopathic pneumonia syndrome is a process of widespread alveolar epithelial injury that is characterized clinically by diffuse interstitial infiltrates and varying degrees of respiratory failure in the absence of active LRTI. It is believed to be related to the chemotherapy or TBI, or both, used as part of the conditioning regimen, which induces proinflammatory cytokine release and increasing alveolar capillary permeability. Idiopathic pneumonia syndrome occurs in 8% to 17% of patients but may be more frequent after allogeneic than autologous transplantation and thus has been implicated, at least in animal models, as a component of the GVHD reaction. Mortality rates are 60% to 80%. Idiopathic pneumonia syndrome occurs classically during two peaks: one in the first few weeks and the other in the second and third month after transplantation. Corticosteroids and etanercept may yield a clinical response. More frequent use of sensitive diagnostic techniques has revealed a high incidence of occult infections, including due to human rhinovirus, CMV, HHV-6, and Aspergillus . The detection of these viruses may be associated with increased mortality. This diagnostic yield changes the “precision” of the diagnosis of idiopathic pneumonia, which should lack detectable pathogens. The uncertain role of this detection on the applicability of immunosuppressive regimens is not yet clear.
Diarrhea, a common symptom after transplantation, is primarily a result of noninfectious causes, such as regimen-related gut mucosal toxicity and GVHD. Diarrhea is associated with infection in less than 20% of cases. The list of infectious agents responsible for diarrhea includes Clostridioides difficile (formerly Clostridium difficile ), adenovirus, rotavirus, norovirus, enterovirus, coxsackievirus, HHV-6, Escherichia coli, Salmonella, Giardia, Strongyloides, Cryptosporidium, and Campylobacter spp. Infection with C. difficile occurs with increasing frequency. Outbreaks of diarrhea have been reported for Cryptosporidium and enterovirus. From other countries, reports of diarrhea have been associated with Trichostrongylus spp.
Typhlitis, or neutropenic enterocolitis, is an anaerobic infectious syndrome that is relatively common and may be associated with diarrhea during neutropenia. Typhlitis is preceded by fever, abdominal pain, and right lower quadrant tenderness that may be accompanied by rebound tenderness. Appendicitis can mimic typhlitis. Computed tomography of the abdomen reveals right-sided colonic enlargement and inflammation with thickening of the mucosa. Therapy against anaerobic bacteria should be added to the medical regimen.
Skin eruptions are often noninfectious, occurring as a direct result of radiation effect from conditioning therapy or secondary to aGVHD or drug reaction. Rashes from conditioning regimens can result in the sudden onset of marked erythema over large areas of the body and blistering on the hands and feet. A skin biopsy can assist in distinguishing infectious from noninfectious causes of rash. For all lesions suspected to be infectious, samples should be submitted for culture or biopsy. The most common infectious causes are VZV, catheter-related exit site or tunnel infections, and cutaneous manifestations of disseminated bacterial or fungal infections. Focal areas of bacterial cellulitis may occur on the lower extremities in the setting of edema from heart failure, VOD, lymphedema, and impaired venous return.
Osteomyelitis is uncommon after HSCT. The spectrum of organisms can include atypical mycobacteria, yeasts, and molds, in addition to bacteria. In rare instances osteomyelitis follows marrow aspiration from the sternum or marrow harvest from the iliac crest. When prolonged pain and fever occur after BM harvest, osteomyelitis caused by Staphylococcus aureus should be considered.
Historically, three risk periods of immunologic deficiency occur predictably in graft recipients after HSCT ( Fig. 307.1 ). They are the preengraftment period, the early postengraftment period (until day 100), and the late postengraftment period (after day 100). An understanding of the immune deficiencies in each risk period and the period of peak risk for individual infections that are observed with standard infection prophylaxis helps the clinician recognize uncommon manifestations of these infectious pathogens ( Table 307.1 ).
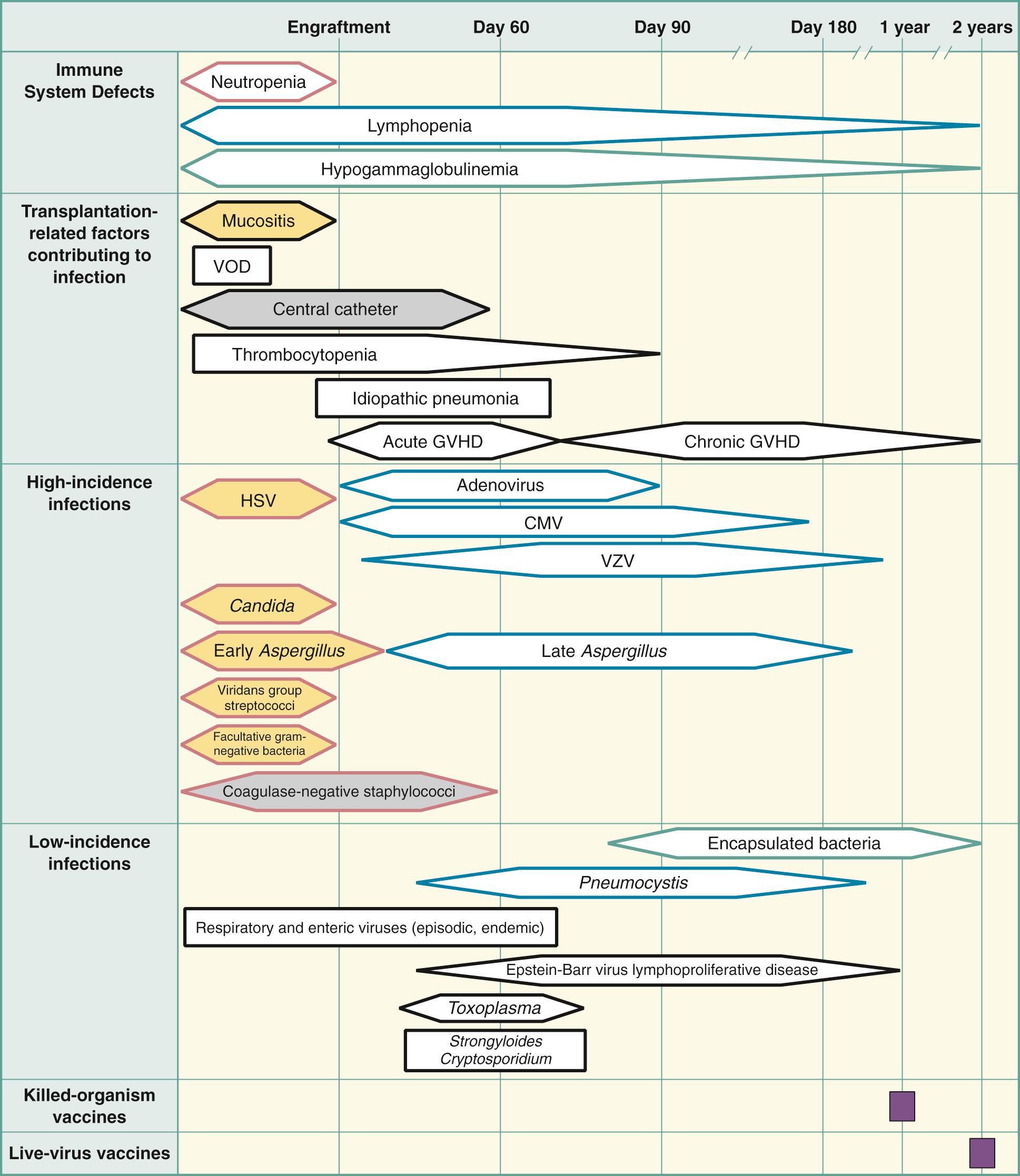
| ORGANISM | PEAK TIME PERIOD OF RISK (WEEKS AFTER HSCT) | USUAL PROPHYLAXIS | INCIDENCE |
|---|---|---|---|
| Preengraftment Risk Period (1–4 Weeks) | |||
| Herpes simplex virus (seropositive) | 1 – 2 | Acyclovir or valacyclovir | 5% – 9% |
| Gram-positive bacteremia (most commonly coagulase-negative staphylococci, viridans group streptococci, and enterococci) | 1 – 4 | Prophylactic broad-spectrum antibiotics | 20% – 30% |
| Gram-negative bacteremia | 1 – 4 | Prophylactic broad-spectrum antibiotics | 5% – 10% |
| Clostridioides difficile (formerly Clostridium difficile ) diarrhea | 1 – 5 | — | 5% – 10% |
| Candida | 1 – 4 | Fluconazole, micafungin, voriconazole | Systemic infection: <5% colonization: 30% |
| Aspergillus and other molds | 1 – 4 | HEPA air filtration, Itraconazole, voriconazole, posaconazole, micafungin, or low-dose amphotericin | <5% |
| Respiratory viruses | 2 – 5 | Isolation, hand washing | 15% |
| Idiopathic pneumonia syndrome | 2 – 4 | — | 8% – 17% |
| Early Postengraftment (4–26 Weeks) and Late Postengraftment (26–52 Weeks) Risk Periods | |||
| Cytomegalovirus (seropositive) | 7 – 26 | Ganciclovir or foscarnet | <5% (end-organ disease), up to 40% (antigenemia/viremia) |
| Varicella-zoster virus (seropositive) | 4 – 52 | — a | ≤50% |
| Aspergillus and other molds | 4 – 26 | Itraconazole, posaconazole, or voriconazole | 10% – 15% |
| BK virus | — | — | ≤50% (shedding) |
| Adenovirus | — | — | 1%–5% |
| Pneumocystis jirovecii | 4 – 104 | Trimethoprim-sulfamethoxazole | <1% |
| Toxoplasma gondii (seropositive) | 2 – 8 | — b,c | 2%–7% |
| Infrequent Infections (May Span Multiple Risk Periods) | |||
| Herpes simplex virus (seronegative) | — | — | <2% |
| Cytomegalovirus (seronegative) | — | Blood product screening or filtration, letermovir | 1%–4% |
| Varicella-zoster virus (seronegative) | — | — | <3% |
| Streptococcus pneumoniae d , | — | Vaccination, penicillin e,f | <1% |
| Haemophilus influenzae d , | — | Vaccination, penicillin e,f | — |
| Neisseria meningitidis d , | — | Penicillin f | — |
| Human herpesvirus 6 | — | Ganciclovir or foscarnet | 5% – 60%, few treated |
| Epstein-Barr virus | — | — | <1% (disease) |
| Nocardia spp. | — | — c | <1% |
| Legionella spp. | — | — | <1% |
| Mycobacterium spp. | — | Screening, then prophylaxis | <1% |
| Listeria monocytogenes | — | — | <1% |
a Antiviral medications used to prevent other viral infections may be acting as prophylaxis against reactivation of varicella-zoster virus.
b Prophylaxis with pyrimethamine-sulfadoxine may be used for seropositive patients in countries with a high rate of seroprevalence.
c The sulfa component of Pneumocystis prophylaxis may be acting as prophylaxis against Toxoplasma or Nocardia infection.
d Risk is increased in patients with chronic graft-versus-host disease.
e Efficacy in transplant recipients is undetermined.
f Increasing penicillin resistance may indicate a need for macrolides or extended-spectrum quinolones.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here