Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
This chapter includes an accompanying lecture presentation that has been prepared by the authors: ![]() .
.
Risks of gliomas and meningiomas vary significantly across sex, age, histology, ethnicity, and geography. The incidences of glioblastoma (the most common type of glioma) and meningioma increase with age. Gliomas are more common in males across all age groups and histologies and have the highest incidence in non-Hispanic Whites. Meningiomas, however, are more common in females and non-Hispanic Blacks.
In 2016 the World Health Organization (WHO) revised classification of CNS tumors incorporated key molecular alterations with histology resulting in an integrated molecular diagnosis. For gliomas in particular, the identification of five molecular subtypes has significantly improved classification of these patients into groups with more similar clinical outcomes.
New inherited genetic risk factors for glioma and meningioma have been identified through genome-wide association studies (GWASs), including 25 risk loci for glioma and 2 for meningioma. Genetic and epigenetic alterations involving telomere maintenance are a common pathway implicated in glioma and meningioma formation.
Few environmental and developmental risk factors have been confirmed for either glioma or meningioma formation. For both, exposure to ionizing radiation is an established risk factor for both, whereas allergies and atopic conditions have been associated with reduced risk. Increased body mass index (BMI) has been associated with increased risk for meningioma. No consistent associations have been found for glioma with numerous other factors, including cell phone exposure.
Nearly 70,000 new cases of primary tumors of the brain and CNS are diagnosed annually in the United States. Of these, approximately 22% are diffuse gliomas and approximately 37% are meningiomas ( Fig. 142.1 ). Gliomas overall and glioblastomas, the most common type of glioma, account for 71% and 53% of malignant primary brain tumors, respectively. Meningiomas account for 53% of benign and 1.5% of malignant primary brain tumors. The Central Brain Tumor Registry of the United States (CBTRUS)—a compilation of population-based data from the Centers for Disease Control and Prevention (CDC) National Program of Cancer Registries (NPCR) and the National Cancer Institute (NCI) Surveillance, Epidemiology, and End Results (SEER)—published a report in 2018 based on data from 2011 to 2015. Overall the incidence of these tumors did not significantly changed from 2000 to 2015, aside from a slight increase in nonmalignant CNS tumors (from 2004 to 2009). This increase has been attributed to improved ascertainment and reporting of nonmalignant cases over this interval, , rather than to a true increase in disease incidence. This review will provide an update on the epidemiology, molecular landscape, and risk factors for the most common primary brain tumors in adults—diffuse gliomas and meningiomas. Please see additional chapters for thorough reviews of the epidemiology of pediatric, pituitary, and nerve sheath tumors.
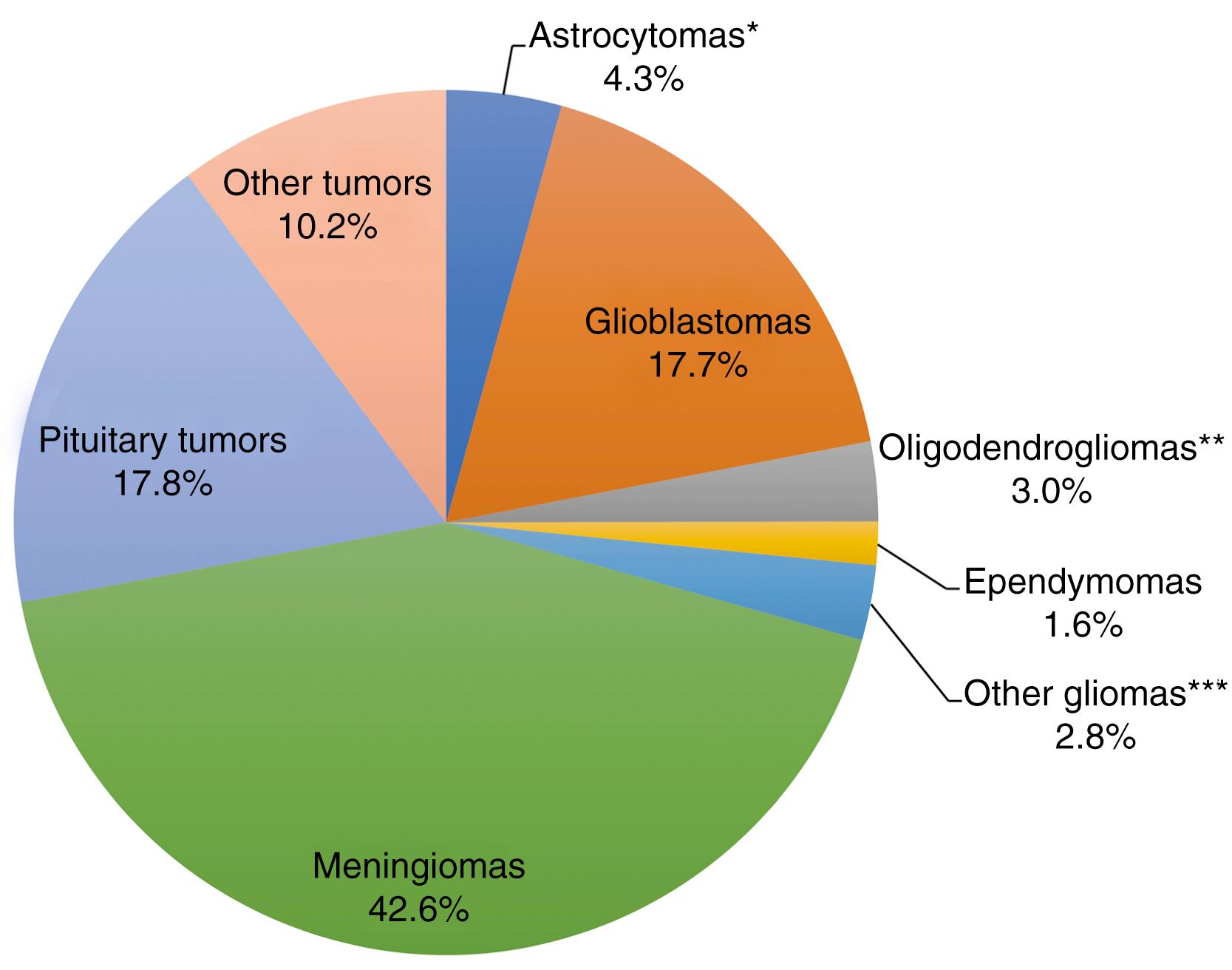
The epidemiology of gliomas and meningiomas varies significantly across sex, age, histology, ethnicity, and geography. For all gliomas, the CBTRUS reports an annual age-adjusted incidence rate (AAAIR) of 6.8 per 100,000, and incidence increases with age. Gliomas are more common in males across all age groups and histologic subtypes ( Fig. 142.2 ). The incidence is highest among non-Hispanic Whites , and in countries with populations of primarily northern European ancestry. It is important to note that the histologic data captured in CBTRUS are based on the World Health Organization (WHO) classification system from 2000. , In 2016 the WHO updated its classification of primary CNS tumors to integrate several molecular markers with histology to better identify subtypes of tumors with the hope of grouping similar clinical outcomes. , They identified five different subtypes: glioblastoma, IDH wild-type; glioblastoma, IDH mutant; astrocytoma, IDH wild-type; astrocytoma, IDH mutant; and oligodendroglioma, IDH mutant, 1p/19q co-deleted. More details regarding this shift in classification are discussed later in this chapter, but it is important to note that population level data on these subtypes through CBTRUS are not yet available and therefore are not reported here.
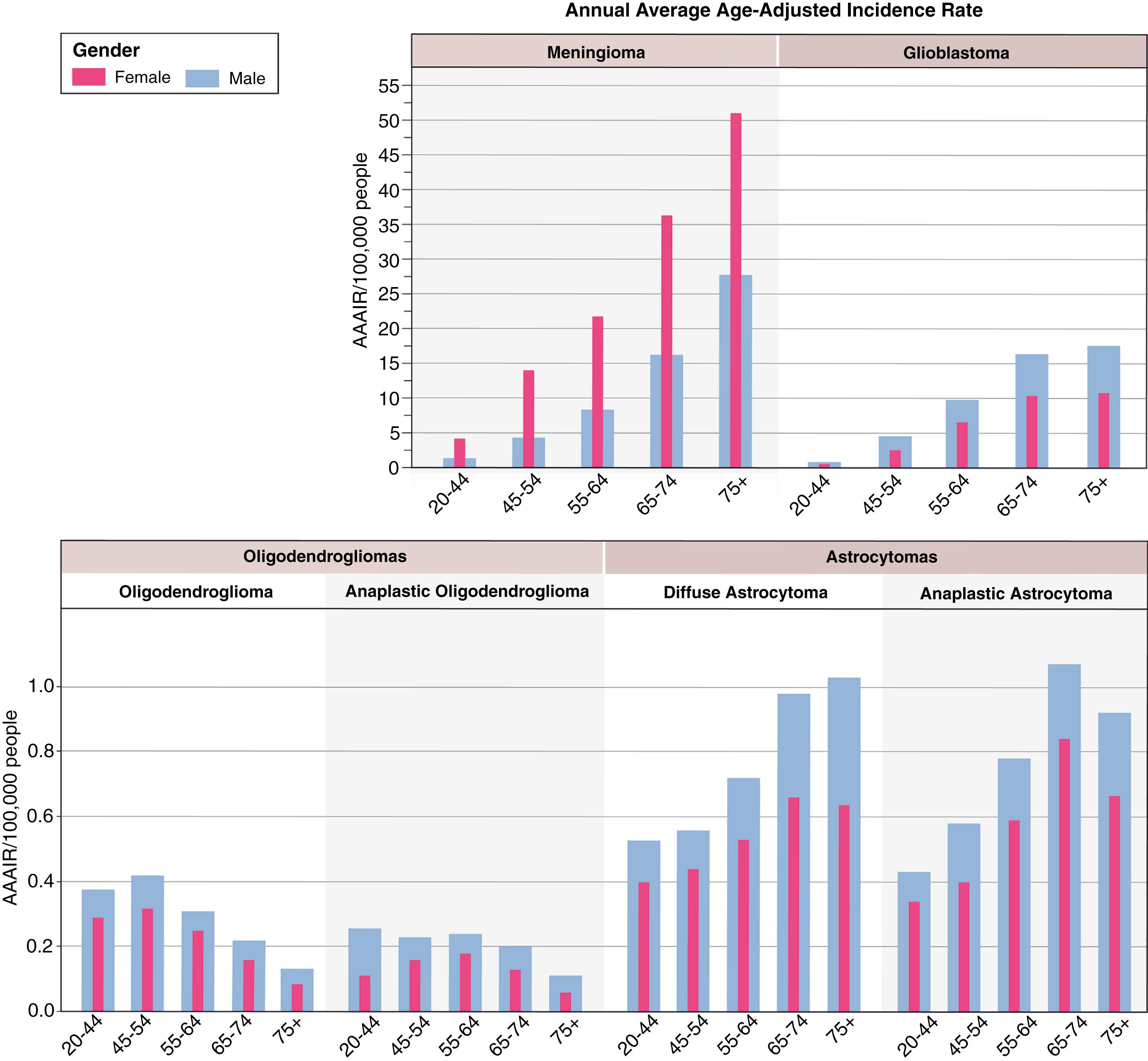
Meningiomas are more common than gliomas, with distinct epidemiology compared with gliomas (see Fig. 142.2 ). Similar to gliomas, incidence of meningioma increases with age, although there is a sharper increase in incidence after age 65 in meningioma. In contrast with gliomas, however, meningiomas are most common in females, and non-Hispanic Blacks have highest incidence. There was little revision to meningiomas in the 2016 WHO classification system, and classification is still solely based on histologic characteristics, with 3 grades—benign (grade I), atypical (grade II), and anaplastic (grade III)—and 15 histopathologic subtypes. Therefore the historical CBTRUS data remain reflective of current classification for these tumors. There is some suggestion that the annual incidence rates of meningioma may be increasing slowly, although this may at least in part be due to improved ascertainment. In a study from the Swedish national cancer registry between 1980 and 2012, 13,441 cases of meningioma were evaluated and a discrete positive trend in incidence in all age groups except those older than 75 years was found.
Because many brain tumors, especially glioblastoma, have such a poor prognosis, investigators have sought to identify factors associated with longer survival in order to discover mechanisms associated with tumor progression. The following variables are significant favorable prognostic markers across all glioma subtypes: younger age at diagnosis; IDH1/2 (isocitrate dehydrogenase) mutation; higher Karnofsky performance scale functional score; marital status (i.e., married); and greater extent of resection. In glioblastoma, promoter methylation of MGMT (O6-methylguanine-DNA methyltransferase), a DNA repair enzyme, is also a favorable prognostic factor and predictive of response to temozolomide (TMZ). , The presence of 1p/19q co-deletion, which in combination with presence of IDH mutation now defines oligodendrogliomas and distinguishes them from astrocytomas, also confers a favorable prognosis and predicts response to treatment with radiation and combination chemotherapy with PCV (procarbazine/lomustine/vincristine). In IDH- mutated astrocytomas, deletion of CDKN2A/B , leading to cell cycle dysregulation, is emerging as a marker that predicts worse outcomes. ,
Glioblastoma was the first cancer systematically analyzed by The Cancer Genome Atlas (TCGA), which identified recurrent mutations and copy number variations in three core pathways—receptor tyrosine kinase signaling, activated oncogenes in p53 signaling, and cell cycle dysregulation in RB signaling. Further data from other groups as well as TCGA data collected for low-grade gliomas , expanded the understanding of gliomagenesis including identifying subtypes with similar survival outcomes. Gene expression profile studies classified glioblastoma into four subtypes based on similar pathway activation, and more recently CpG island methylation signatures were used to identify six distinct glioma subtypes. , The telomere lengthening mechanism of gliomas either through alterations in the TERT (telomerase reverse transcriptase) gene or via mutations that affect alternative lengthening of telomeres (such as ATRX ) also differentiates glioma subtypes with respect to prognosis. ,
Identification of these genetic and copy number alterations, as well as epigenetic changes in gliomas and several other CNS tumors, motivated a restructuring of the 2016 World Health Organization Classification of Tumors of the Central Nervous System to incorporate molecular markers along with histology for an integrated diagnosis. Given the ease of testing for IDH1 R132H (which comprises >80% of all IDH1/2 mutations) with immunohistochemistry and 1p19q co-deletion with fluorescence in situ hybridization, as well as their strength as prognostic factors, these two tumor markers were included in the WHO 2016 classification system. , The Consortium to Inform Molecular and Practical Approaches to CNS Tumor Taxonomy (cIMPACT-NOW) was formed to provide updated timely updates as research evolves and new clinically relevant molecular markers are identified. , Updates thus far have included further clarification of astrocytomas that meet molecular features of glioblastoma but failed to meet histologic criteria, and those with mutations in H3K27M diffuse midline gliomas.
These improvements in molecular characterization have led to greater accuracy in classification of diffuse gliomas, but survival overall remains poor, particularly for glioblastoma, IDH wild-type. The addition of the alkylating chemotherapy agent TMZ to radiation has led to improvement in 1-year and 5-year survival rates since its approval in 2004, but median overall survival for glioblastoma, IDH wild-type remains limited to 15 to 18 months. , The 5-year survival rate for glioblastoma is 5.8%, for other astrocytomas is 44.4%, and for oligodendrogliomas is 70.0%. , Non-Hispanic Whites have lower survival outcomes compared with Hispanic Whites, Blacks, and Asian or Pacific Islanders across all tumor types. ,
Population-based data from Norway and Finland suggest that survival of patients with meningioma improved between the 1950s and 1990s , ; this improvement may have been due to earlier diagnosis accompanying advances in imaging technology. Favorable prognostic factors include female sex, younger age at diagnosis, and the performance of resection. Survival from meningioma is generally poorer for patients with malignant versus nonmalignant tumor histology, with a 5-year survival rate of 64.3% for malignant meningiomas versus 86.8% for nonmalignant meningiomas, which account for 96% of all meningiomas.
Prognostic factors for meningioma have not been thoroughly studied, perhaps because these patients typically have a favorable prognosis, unlike those with glioma. In general, as in glioma, older age at diagnosis and higher histologic grade contribute to poorer prognosis. Results from a large study of 9000 patients revealed the following factors associated with prognosis for nonmalignant meningioma: age, tumor size, and surgical and radiation treatments. In contrast, only age, surgery, and radiation treatment were associated with prognosis in patients with malignant meningiomas. A study focused on prognosis of atypical meningiomas found intratumoral calcifications and brain invasion at imaging to be associated with longer progression-free intervals, whereas older age and poorer performance status were associated with higher mortality. Further imaging characterization of meningioma will likely be beneficial to better define prognosis in the future.
Over the past several years there has been an increased effort to understand the underlying biology of meningioma and its impact on behavior and prognosis. Multiple studies dating back to the 1960s have underscored the importance of copy number alterations in meningioma. The most common alteration seen is loss of chromosome 22q, which is seen in 60% to 70% of sporadic meningiomas. , However, there are a number of other mutations that are mutually exclusive with NF2 mutation and that are found primarily in grade I meningiomas. TRAF7 (tumor necrosis factor [TNF]) is mutated in approximately 20% of meningiomas ; this mutation is commonly seen in conjunction with mutations in in v-akt murine thymoma viral oncogene homolog 1 (AKT1) or Krüppel-like factor 4 (KLF4). , AKT mutations are seen in 10% of sporadic meningiomas and are mutually exclusive with KLF4 mutations. All three of these mutations localize to the anterior and middle fossa skull base. Also mutually exclusive with NF2 mutation is mutation in Smoothened ( SMO ; part of the Frizzled family receptors), which occurs in 3% to 6% of meningiomas; these tumors are exclusively observed in grade I tumors, and typically localize to the anterior midline skull base. Another 6% of grade I meningiomas harbor a mutation in RNA polymerase II (POLR2A) and localize to the anterior skull base/tuberculum sellae.
The incidence of loss of chromosome 22q increases with grade and is present in 75% to 85% of WHO grade II and III tumors. , Deletion of chromosome 1p is also commonly seen in meningioma and is also associated with higher grade. , In addition, loss of 9p and 14q is also associated with higher grade tumors as well as with increased recurrence rates. Mutations in the Switch/sucrose nonfermentable (SWI/SNF) chromatin-remodeling complex, including SMARCB1 and SMARCE1, have a higher frequency in anaplastic meningiomas and are associated with worse prognosis. Another marker of clinically aggressive meningiomas that has been recently identified is mutation in the telomerase reverse transcriptase (TERT) promoter, specifically in the hotspot regions C228T or C250T, which has been identified in 6.5% to 11% of meningiomas. ,
Gene expression profiling has also identified genetic signatures associated with prognosis, one of which found a 37-gene signature, and a second an 18-gene signature. , Most of these genes were associated with normal development, cell proliferation, and angiogenesis. , Epigenomic changes may also contribute to increased risk of meningioma. DNA methylation has become a focus for studies on mechanisms of tumorigenesis. For meningioma there is evidence of hypermethylation in several notable genes, including tissue inhibitor of metalloproteinase 3 (TIMP3), cyclin-dependent kinase inhibitor 2A (CDKN2A), and tumor protein 73 (TP73). Hypermethylation in TIMP3 is seen in 40% to 60% of grade III meningiomas and is associated with shorter progression-free intervals. , In addition, promoter methylation of TP73 has been reported in 70% to 80% of high-grade meningiomas Global methylation signatures are also likely important for classification and prognosis of meningioma, and unsupervised clustering of methylation patterns has found distinct subgroups associated with outcomes, even after adjustment for other known prognostic factors. Other studies using unsupervised clustering of DNA methylation data identified two major groups and six subgroups with markedly different clinical outcomes, and supported the notion that non– NF2- mutant meningiomas follow a more benign course. Another important epigenetic factor that has been recently identified is the trimethylation of H3K27, which is associated with a subset of meningioma with clinically aggressive behavior. Accordingly, prediction models of clinical behavior of meningioma based on methylation are being developed and may be more useful to predict clinical outcomes and guide therapy than traditional grade.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here