Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
The surgical treatment of abdominal aortic aneurysms (AAAs) dates back centuries. Some of the initial approaches involved techniques similar, in some fashion, to modern endovascular techniques. In 1684, Moore reported on the use of large quantities of wire placed intraluminally into the aneurysm sac in order to induce thrombosis of AAA. Later, electrical currents were passed through the wire to further promote thrombosis. A self-expanding endoluminally placed umbrella device was reported by Colt in the early 1900s to treat AAA. In the mid-1900s, the use of endoluminally placed wire with the passage of electricity through it was revived and remained the procedure of choice until conventional operative therapy of AAA was introduced. Operative repair of AAA evolved during the second half of the 20th century. Early techniques ranged from simple aortic ligation to aortic wrapping with cellophane. Neither was successful. In 1951, the first replacement of an aortic aneurysm with an aortic homograft was described by Dubost. However, homografts became aneurysmal over time and the procedure evolved to the use of prosthetic material to reconstruct the aorta. This technique was later modified by Creech, who reported on endoaneurysmorrhaphy with intraluminal graft placement, leaving the aneurysm sac in situ ; this had become the mainstay of treatment until the mid-1990s.
Although excellent results have been obtained with conventional aneurysm repair, it remains a complex, challenging operation that initiates great physiologic stress for patients. The pursuit of a less invasive approach to AAA repair has subsequently evolved. Parodi and colleagues reported the first use of endovascular aneurysm repair (EVAR). This approach allowed for the intraluminal exclusion of an aneurysm with the placement, through the femoral arteries, of an endograft. The hope was that this would decrease the morbidity and mortality of aneurysm repair and allow repairs to be performed in patients with significant comorbidities. The original endograft was constructed of a Dacron tube sutured to a Palmaz stent. Several generations of endografts have since been developed, tested, and put into general clinical use. With the evolution of aortic endografting, our knowledge about the pathophysiology of AAA has changed. Our understanding of the complexities of this mode of treatment is only just being realized and examined. This chapter reviews what is currently understood about endograft repairs of AAAs.
The indications for endovascular repair of an AAA remain the same as conventional repair with regard to the size of the aneurysm and its rate of growth. The classic teaching is that rupture rates for aneurysms depend on the size of the aneurysm. Rupture rates of 5% to 7% per year are estimated for aneurysms between 5 and 7 cm in diameter, and a greater than 20% rupture rate per year is estimated for larger aneurysms. Surgical treatment for patients with these larger aneurysms significantly improves mortality compared with observation. Although it is known that small aneurysms do have the potential to rupture, a recent meta-analysis looking at four randomized controlled trials (ADAM, UK small aneurysm trial, CAESAR, and PIVOTAL) demonstrated no advantage to early repair via open or endovascular surgery in men and women with asymptomatic AAA of diameter 4.0 to 5.5 cm. The authors recommend surveillance in this population.
EVAR is a “less-invasive” technique than open surgery and offers several potential benefits over conventional AAA repair. It at most requires small femoral incisions instead of a large abdominal incision, which may decrease the incidence of postoperative pulmonary complications. The avoidance of extensive retroperitoneal dissection decreases the risk for perioperative bleeding. The period of aortic occlusion is minimal and accounts for the lower incidence of intraoperative hemodynamic and metabolic stress compared with patients undergoing open surgery. Given these differences, endovascular aneurysm repair may be reasonable in patients who are “unfit” for conventional AAA surgery. The durability of endovascular repair as a replacement for conventional surgery in relatively healthy patients has been proven by many clinical trials.
The exact anatomic requirements for placing an aortic endograft vary with device design. There are key aspects of each device and aortic anatomy to be aware of when assessing a patient as a potential candidate for endograft repair. Preprocedural imaging is paramount to properly assess the proximal and distal sites of fixation, as well as to assess the path the endograft will traverse before taking its postdeployment position.
Successful endograft placement is completely dependent on adequate and accurate preoperative planning. One of the major elements distinguishing preoperative planning in open aneurysm repair from endovascular repair is the latter’s increased dependency on imaging to provide information necessary for clinical decisions. Preprocedural imaging allows the surgeon to determine whether a patient is an acceptable candidate for endovascular aortic grafting and which device is best suited for a particular patient; this ultimately allows for determining the proper size of the endograft.
Historically, contrast aortography was used as a routine adjunct to axial imaging, as it was thought to allow for a more accurate determination of vessel length and angulation before CT reformatting was widely available. Preoperative angiography is now rarely employed and reserved for cases where an adjunctive therapeutic intervention (i.e., coil embolization) is necessary. Recently, the advent of flat panel technology has allowed for the development of rotational angiography techniques that facilitate the construction of three-dimensional images found to be comparable to standard multidetector CTA. This technique has been termed C-arm cone-beam CT or FluoroCT. FluoroCT utilizes a modified C-arm with specialized software. This technique uses the spin rate of the C-arm around the gantry to acquire images. Most machines take between 6 and 8 seconds. To get good contrast imaging, it may require a large contrast bolus. This technology appears valuable in complicated aortic cases, such as fenestrated branched endovascular aneurysm repair, due to its ability to image graft to graft and graft to aorta apposition without the use of contrast. However, concerns have developed regarding overall radiation doses delivered to patients and staff, due to pre-, intra-, and postoperative imaging and therefore FluoroCT is not routinely used.
Spiral CT of the abdomen and pelvis is the mainstay of aortoiliac imaging. The imaging protocol is different from the standard protocol for most abdominal CT scans. Acquisition should use a 1:1.5 helical pitch and 3 to 5 mm collimation. Slices of 1.5 mm are ideal for providing adequate information for stent graft planning. The two-dimensional (2-D) images, however, can often be misinterpreted. The axial images may “cut” vessels at an angle, particularly iliac arteries that have some degree of tortuosity—thus creating an ellipse as opposed to visualization of the true lumen diameter. Due to this problem, some physicians recommend three-dimensional (3-D) image processing as a better method to evaluate aortoiliac anatomy for endograft therapy. Although it was common initially to perform an angiogram in addition to CT scan, recent evaluations suggest that high-quality 3-D CT scans alone may provide sufficient data for endovascular graft planning. Currently, proprietary products for CT postprocessing provide the ability to evaluate the 3-D reconstruction of the aortoiliac system rapidly, rotate the images on the screen to obtain better vessel diameter measurements, and provide “virtual endograft” simulation.
Magnetic resonance angiography (MRA) can provide information similar to that of a CT scan. It, too, can provide thin-slice reconstructions and 3-D postprocessing. Its usefulness, however, is often limited by availability and physician expertise. MRA may provide a useful modality in patients in order to avoid the use of iodinated contrast agents. However, use of gadolinium-based contrast agents in MR is not without risk and may result in nephrogenic systemic fibrosis, and gadolinium deposition in bone and parts of the brain.
Intravascular ultrasound (IVUS) is not routinely used in the preoperative evaluation of an endograft candidate. It is an invasive procedure, often performed at the time of angiography. Images produced by IVUS have a similar problem as viewing axial images on a 2-D CT scan. Unless the catheter remains centerline within the aorta, the images produced will be elliptical, which may also provide shorter than required length measurements. Its primary use is at the time of stent graft placement to assess graft position relative to the renal artery ostia; this can help to diminish the amount of contrast agent required.
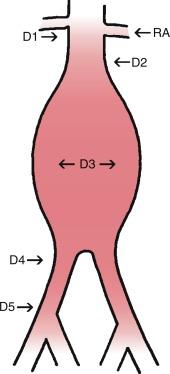
The aortic neck is defined as the area of the aorta cephalad to the aneurysm in which the aortic endograft is placed ( Fig. 38.1 ). This zone of the aorta is important for two reasons during aortic endografting. First, it is the site of proximal fixation that will prevent the device from migrating distally. Second, a circumferential seal must be obtained between the graft and the aorta in this area in order to prevent leakage of blood into the aneurysm sac. The exact length of aortic neck required is somewhat device dependent, but most commercially available devices require a 10- to 15-mm length of aortic neck below the level of the most caudal renal artery. Some investigational devices may allow for shorter necks. One such device is the Medtronic Heli-FX EndoAnchor System, which is indicated for patients with aortic neck lengths < 10 mm and > 4 mm and < 60-degree infrarenal angle. Prospective analysis of 1 year outcomes from 19 US and 3 EU sites demonstrated a rate of type Ia endoleak of 1.9%; however, these results come from a small-sample–size trial ( n = 70). The ANCHOR study demonstrated an overall rate of type Ia endoleak 9.9% in 202 patients who underwent EVAR for various indications. Overall, EndoAnchors are useful in EVAR to prevent proximal site complications, and therefore has been given intent for use by the US Food and Drug Administration.
Additionally, several devices employ the use of a suprarenal, uncovered (or bare) stent to provide additional protection against graft migration. Suprarenal stent fixation may be useful, particularly in patients who have a shorter aortic neck, as it transfers protection against migration to a more normal segment of aorta. The suprarenal stent, however, does not provide any function with regard to creating a circumferential seal.
In addition to the length of the neck, other anatomic characteristics are important when determining whether patients are suitable candidates for endovascular aneurysm repair. These include aortic neck angulation, the shape of the neck, and the quality of the neck. Neck angulation refers to an alteration in the direction the aorta takes with regard to the centerline pathway. Acute angulation of the aortic neck can greatly affect the endograft’s ability to obtain a proximal seal. Aortic neck angulation of greater than 60 degrees compared with the centerline is often considered prohibitive for endovascular aneurysm repair. There are devices undergoing preliminary trials that would allow for greater neck angulation, up to 90 degrees. The shape of the aortic neck also affects the ability of the graft to obtain a seal as well as fixation. A conical-shaped neck ( Fig. 38.2 ) is generally thought to be unstable and predisposes to distal migration. An increase in diameter from the top of the neck to the bottom of greater than 10% is often believed to be a contraindication to routine aortic endografting. The presence of circumferential thrombus or aortic calcification can also negatively affect an endograft’s ability to obtain a proximal seal.

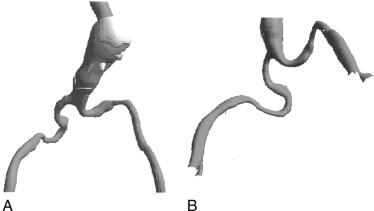
The iliofemoral arterial system is important in endograft placement for two reasons. First, most endografts are placed through the common femoral artery and must traverse the iliofemoral system to reach the aorta. Iliac artery diameter and tortuosity can adversely affect the ease with which the endograft traverses this course. This topic is covered in more detail below. Certainly, the presence of significant atherosclerotic disease can cause arterial narrowing that inhibits the placement of the device. In addition, tortuosity of the iliac arteries can hinder placement of the grafts ( Fig. 38.3 ). Second, the iliofemoral system is important because it is the site of the distal seal between the endograft and the iliac artery, preventing retrograde flow of blood into the aneurysm sac. Many of the features necessary for an adequate aortic neck are also necessary for the distal landing zone. The presence of thrombus, calcification, and tortuosity can significantly hinder the iliac limb seal. Ectatic or aneurysmal iliac arteries obviously affect the ability of the graft to seal against the iliac limb. Most available endograft systems require a femoral artery of 8 mm and common iliac artery diameter of 8 to 25 mm. Some endograft systems, such as Medtronic Endurant, require at least a 15-mm segment of iliac artery to be of adequate caliber and free of significant disease in order to obtain a distal seal, whereas others, such as Zenith from Cook Medical and Excluder from Gore, only require 10-mm distal landing zone. If this is not present, adjunct interventions can be performed to assist in placing the device (i.e., iliac artery conduit placement or coil embolization of the internal iliac artery). Management of these complicated situations is discussed in more detail later.
Endograft design can greatly affect the ability of the device to be placed in patients, particularly in patients with complex anatomy. Alterations in the characteristics of the grafts are what distinguish one manufacturer’s device from another. Some of the key elements in endograft design are outlined as follows.
Standard endograft insertion involves placement of the device through an arteriotomy in the common femoral artery, from where the graft traverses the external iliac and common iliac arteries. The ability to deliver the endograft safely and effectively in this fashion is a prerequisite for effective repair. Three factors are important determinants of device delivery.
With the placement of most endografts through the iliofemoral arterial system, any site along this pathway can represent a size limitation, the most common of which is the external iliac artery. Inadequate diameter or the presence of extensive calcifications can exclude standard endograft placement. It is intuitive that the size of the delivery system cannot be larger than the size of the iliac arteries that it traverses. Most sheaths are sized based on inner diameter, so knowledge of the outer diameter of the sheaths is therefore required for safe graft placement. Different manufacturer’s devices have different size measurements for the delivery systems, and thus one device may be suitable for placement, whereas another is not. Most delivery systems easily traverse an iliofemoral segment of 7 to 8 mm in diameter (or a sheath that does not exceed 21 French [Fr] outer diameter), although several designs that provide a lower profile system are now available. Lower profile delivery systems have allowed for the advent of totally percutaneous femoral artery access. A recent meta-analysis demonstrated no difference between percutaneous approach versus surgical cut-down for common femoral artery access including short-term mortality, aneurysm exclusion, wound infection, bleeding complications, and hematoma.
Tortuosity, another anatomic variant, affects the ability to deliver adequately the endograft system. Tortuous iliac vessels can be “straightened” with the use of stiff guidewires, but this is not always possible or desirable. The ideal delivery system easily traverses these arteries on the basis of an intrinsic degree of flexibility. Again, different delivery systems have different abilities to track through tortuous iliac arteries, and thus some may be more successfully placed than others in this anatomic variant. Delivery systems composed of long, flexible, tapered tips pass more easily than those with short, stiff, blunt tips. In addition, other aspects of device construction, such as metallic struts that provide columnar strength, increase device rigidity and limit use in tortuous vessels.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here