Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Cardiac fibrillation is an electromechanical dysfunction of the heart that is driven by complex three-dimensional (3D) electrical excitation waves, resulting in incoherent mechanical contraction, loss of pumping function, and risk of sudden cardiac death. The dynamics of vortex-like rotating waves or rotors plays an essential role in the spatiotemporal organization of fibrillation. However, the detailed rotor dynamics, their interaction with each other and with the 3D heterogeneous anatomical substrate, is not yet fully understood. Advanced measurement technologies have been developed during the past decades to elucidate the mechanisms underlying the onset, perpetuation, and control of cardiac arrhythmias. In particular, optical mapping using potentiometric dyes has revolutionized the ability to visualize cardiac dynamics during fibrillation in preclinical research because of its unsurpassed spatial and temporal resolution. However, optical mapping has two main limitations. First, it is sensitive to mechanical motion, resulting in significant motion artifacts during contraction. Second, it can only visualize waves on the heart surface. This chapter discusses recent advances in optical mapping to capture the electrical and mechanical function of the beating heart. We show that optical mapping can provide simultaneous measurements of membrane potential, intracellular calcium, and mechanical contraction in intact isolated hearts. , Using this imaging technique, we observed electromechanical rotors, that is, coexisting electrical and mechanical rotors during fibrillation, and discuss their dynamical and statistical properties. Furthermore, we visualize 3D mechanical rotors inside the ventricular wall using high-resolution 4D ultrasound imaging. We also discuss new diagnostic and therapeutic perspectives that are opening up because of the availability of clinically available 4D ultrasound to resolve electromechanical rotors inside cardiac tissue.
The first report of a cardiac arrhythmia describes the trembling mechanical motion of the heart muscle during fibrillation and dates back to 3500 BCE. , Since then, the mechanical motion of the heart has been a natural and primary observation to assess cardiac function. In 1543 Vesalius described the wormlike deformation of the heart muscle during arrhythmia. The first record of ventricular fibrillation (VF) was obtained by Hoffa and Ludwig in 1850 using a mechanical wave recorder, a so-called kymograph. In 1874 the term fibrillation was coined by Vulpian, linking this phenomenon to the mouvement fibrillaire (i.e., the heart’s quivering mechanical motion). Vulpian observed that contractile patterns evolve in time and allow the characterization of VF progression and type. One year earlier, in 1873, Lippmann invented a sensitive capillary electrometer that enabled Waller’s first measurement of an electrocardiogram (ECG) and led the foundation of the electrical assessment of cardiac function. The first human ECG was recorded in 1887 by Waller, and Hoffman obtained the first ECG recording of VF in 1912. The ECG signal morphology reflects the electrical activation sequence of the heart in health and disease. It represents a milestone in medicine, as it enables the noninvasive, standardized quantification of cardiac function, and has been an indispensable diagnostic tool since its introduction into clinical cardiology in the early 20th century. In 1930 Wiggers used ECG recordings and simultaneous fast cinematography to study the contractile motion of the ventricular epicardium and electrical excitation during VF in canines. Later, the notion that cardiac rhythms observed in the ECG time series are associated with propagating 3D excitation waves in the heart muscle expanded this picture, and the spatiotemporal organization of these waves came into focus. Fig. 37.1A–B shows the transition from a normal heart rhythm to ventricular tachycardia (VT) and VF in the ECG and the corresponding propagation of 3D excitation waves. During normal rhythm, excitation waves propagate periodically through the conduction system and the myocardium, resulting in synchronous excitation, coordinated mechanical contraction, and efficient pumping (see Fig. 37.1B ). VT is driven by a rotating 3D excitation wave, resulting in a fast but still periodic ECG signal and significantly reduced pumping function (see Fig. 37.1C ). The loss of stability of this rotating wave results in a degeneration of VT into VF. During VF, electrical excitation is desynchronized, yielding incoherent mechanical contraction, and loss of pumping function (see Fig. 37.1D ). Multiple mechanisms have been identified that underlie the onset and maintenance of cardiac arrhythmias , ; some of them are still under debate.
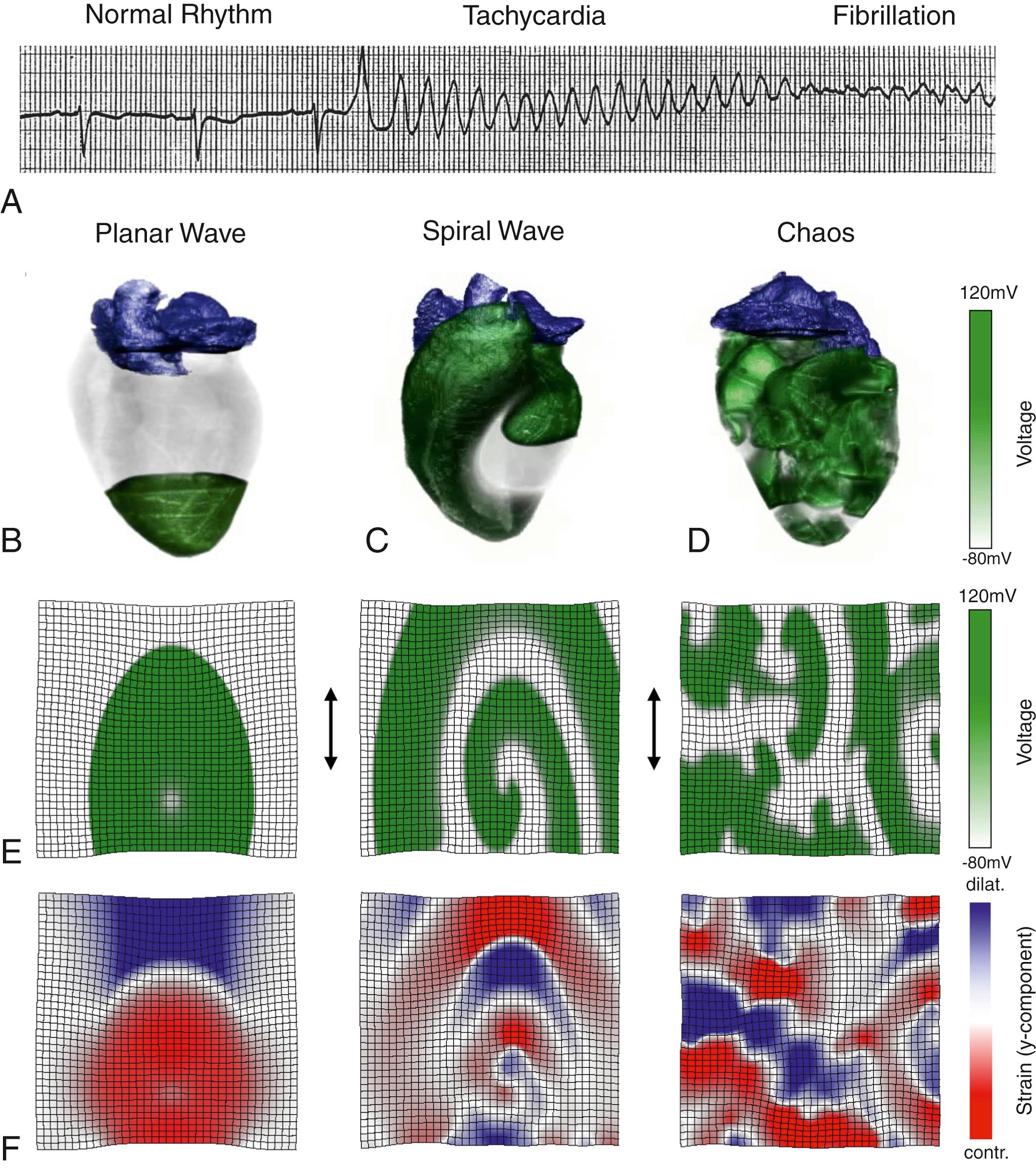
In the heart, electrical excitation and mechanical contraction are intimately related. At the cellular level, electrical excitation leads to mechanical contraction through the excitation-contraction coupling. Electrically excitable cells couple to their nearest neighbors via gap junctions, enabling the diffusive propagation of electrical excitation waves through the cardiac muscle. The dynamics of electrical and mechanical waves in the heart depend on the cells’ local ionic dynamics, heterogeneity, the anisotropy of the tissue’s electrical and mechanical properties, and the organ’s geometry and anatomy. The spreading electrical excitation wave (see Fig. 37.1E , green) triggers local mechanical contraction that propagates as an elastic deformation wave (see Fig. 37.1F ). This mechanical wave consists of propagating adjacent zones of contracting (red) and dilating strain (blue). In the diseased heart, excitation-contraction coupling and mechanoelectrical feedback may be altered, resulting in contractile dysfunction and the onset of arrhythmias. Cardiac arrhythmias are complex spatiotemporal excitation patterns. Correspondingly, the elastic deformation waves become more complex. During VT, the rotating electrical excitation leads to rotating zones of mechanical contraction and dilation or rotating zones of contractile and dilating strains (see Fig. 37.1F ).
Despite the intrinsic coupling of electrical and mechanical processes in cardiac tissue, their underlying physical principles are very different, as are the properties of the emergent electrical and mechanical waves. Electrical waves in the heart are solitary excitation waves (and, for example, annihilate under collision) with a typical conduction velocity of 1 m/s. In contrast, electromechanical waves are elastic deformation waves initiated by the excitation waves via the excitation-contraction coupling. However, their wavelike propagation may be altered because of subsequent interactions between the heart’s mechanically interconnected physiologic structures. Passive elastic waves traveling through the heart can also effectively introduce nonlocal mechanical interactions because of the large speed of sound (approximately 1500 m/s) in cardiac tissue. The mechanical deformation waves represent the heart’s spatiotemporal organization, analogous to, but not identical to, the electrical excitation waves. Electrical excitation and mechanical contraction appear to be two sides of the same coin, suggesting that the heart’s function in general and cardiac arrhythmia, in particular, should be considered as electromechanical phenomena. The dynamics of electric rotors are a hallmark of cardiac tachyarrhythmias. This chapter discusses the extension of this concept to the dynamics of electromechanical rotors.
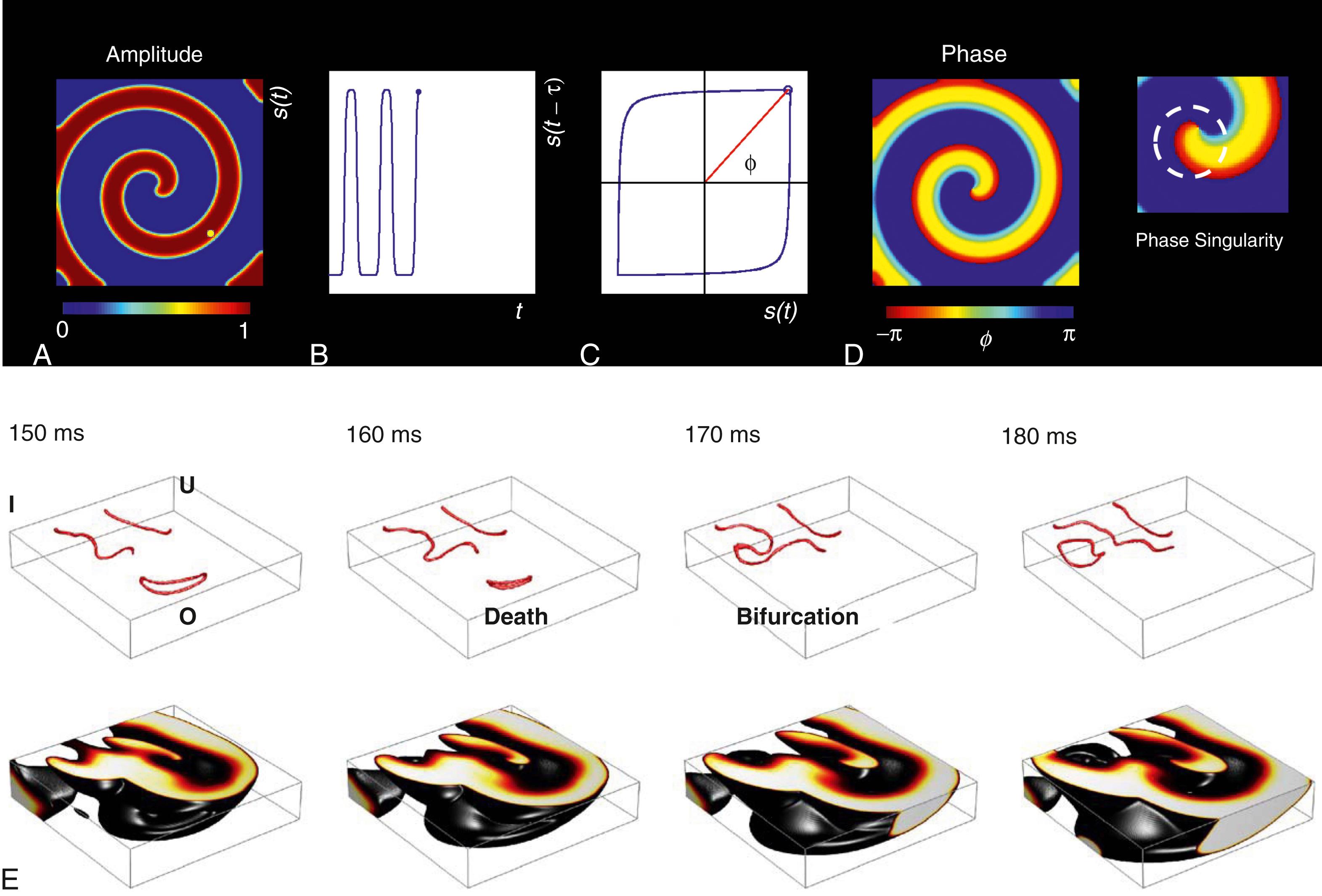
Cardiomyocytes are excitable cells that respond to a super threshold stimulus by an action potential, followed by a refractory phase during which it is not possible to evoke another action potential. In tissue, the cells are electrically coupled, forming an excitable medium where excited cells can induce action potentials in neighboring cells. This interaction may not only result in plane or circular electrical excitation waves but can also support rotating waves, as illustrated in Fig. 37.2A . Such rotors (also called spiral waves in 2D or scroll waves in 3D) are an immediate consequence of the existence of the refractory phase. They are characteristic for any kind of excitable medium, in cardiac dynamics and beyond. A single rotor located at a fixed position represents a periodic motion corresponding to a closed orbit in the state space of the system. The periodic motion on this limit cycle can be described by a single variable called the phase (angle). This picture also holds if we consider the dynamics at a specific location x where the rotating spiral wave leads to a periodic signal s(t) = s(t + T) with period T (see Fig. 37.2B ). The signal s(t) can be any measured quantity, for example, the extracellular potential, the local calcium concentration, or mechanical strain. To describe and analyze the periodic motion using a phase variable ϕ(t), we first have to extract this quantity from the observed signal s(t). A straightforward approach is to introduce a variable that linearly increases from 0 to 2π during one period T (e.g., triggered whenever the signal crosses some threshold). To refer directly to the parametrization of the motion on a closed orbit, we first have to (re)construct this orbit in a 2D state space from the observed (scalar) signal s .
This can be done in several ways. The first temporal derivative
can be computed from s(t) (using finite differences, for example) and combined with the signal to reconstruct the state y (t) = [y 1 (t),y 2 (t)] = [s(t),ṡ(t)] moving on a closed orbit in the s(t) – ṡ(t) plane. Or, a so-called delay embedding y (t) = [y 1 (t),y 2 (t)] = [s(t),s(t – τ)] can be used where the second component is given by the time-delayed signal s(t – τ). , For a sinusoidal signal s(t) = sin(ωt), a delay of τ = T/4 = π/2ω would result in an optimal circle-shaped reconstruction [sin ω(t), cos(ωt)] of the closed orbit. This feature can be achieved for arbitrary periodic signals by shifting the phases of all its Fourier components by 2π/4 = π/2, as it is done by the Hilbert transformation. This motivates the third approach of 2D state-space representation [y 1 (t),y 2 (t)] = [s(t),s H (t)] combining s(t) with its Hilbert transform s H (t).
The 2D state-space reconstruction can now be used to introduce a phase variable describing the motion on the closed orbit. To do so, select a point of reference (r 1 ,r 2 ) within the circle and compute the angle ϕ(t) = arctan2(y 2 (t) – r 2 ,y 1 (t) – r 1 ) as illustrated in Fig. 37.2C . An alternative, often used in practice, is to subtract the mean from s(t) before state space reconstruction because, in this case, the origin is usually a good point of reference (r 1 ,r 2 ) = (0,0) for introducing the phase variable ϕ. Having computed the phases ϕ( x ,t) at time t at each location x (in one or the other way), their values can be plotted to obtain an alternative visualization of the rotating wave (see Fig. 37.2D ). As can be seen, the spiral tip is a singular point; following a circle around it shows that the phase attains all possible values (i.e., its values fill the full interval [0,2π]). At the tip of the spiral, the amplitude of s(t) is zero; therefore the phase is not defined there. In other words, a phase singularity (PS) occurs at the center of the rotor (or spiral tip). To detect it, compute the line integral over the gradient of the phase ∇ϕ( x ,t) along a closed path δD
where n and m are the numbers of clockwise and counterclockwise rotating PSs enclosed by the boundary δD of D, respectively. To detect a single PS, the integration path δD should be a short as possible, limited practically by the spatial discretization in numerical simulations and spatial resolution of measurement devices. The value of the line integral ( Eq. 37.1 ) is also called the topological charge of the PS. It indicates the direction of rotation of the rotating wave and obeys conservation laws analogous to electrical charges. Although there are various processes by which spiral waves are created or annihilated, the conservation of topological charge requires that PS can only be created or annihilated in pairs with opposite topological charge. Li and colleagues presented an alternative method for estimating the topological charge (without explicitly computing phases and the line integral). They introduced the so-called Jacobian-determinant method, which is also based on delay embedding.
So far, we have considered the ideal case of a spiral wave rotating periodically at a fixed location in space, such that the tip of the spiral is a fixed point. In general, however, spiral waves may exhibit complex motion, for example, drifting or meandering while rotating. In this case the signal s(t) measured at location x is in general no longer periodic, but quasiperiodic (with at least two incommensurate frequencies whose ratio is an irrational number) or chaotic. As long as the previously introduced methods for (2D) state-space reconstruction yield trajectories that resemble noisy limit cycles, an approximative phase variable can still be introduced, but this approach fails as soon as no clear “hole” is visible in the reconstructed orbit that could guide the selection of a good point of reference needed to define the phase (angle). Although well defined for periodic spiral waves, the concept of phase variables may thus lose its meaning when it comes to very complex wave patterns with chaotic breakup of spiral waves.
Further challenges for detecting and tracking PS are measurement noise and limited spatial resolution. Bray and coworkers suggested identifying the position of PS through an image analysis method using spatial convolution. However, the method was found to produce a large number of false-positive PSs. , To overcome this limitation, Tomii and colleagues introduced an algorithm for detecting spiral tips using the variance of the phase values, which is larger around PS. Although most methods for detecting PS rely on local information, Marcotte and Grigoriev suggested a global topological approach where PSs are associated with intersections of level sets of two variables representing the fast and slow dynamics. An extension of this approach was presented by Gurevich and colleagues , ; it is based on a single observable and can be applied to experimental data.
A PS represents the core of a rotational pattern, for example, a spiral wave pattern of electrical excitation in a 2D excitable medium. In contrast, in a 3D excitable medium a line of PS or a PS filament represents a rotational core of a 3D scroll wave or rotor pattern. The development of sophisticated imaging techniques, in particular, optical mapping, signal analysis, and numerical simulations, has shaped our current understanding of rotors and their role in cardiac fibrillation. Despite this progress, however, there is still debate about the precise mechanisms underlying fibrillation (e.g., a small number of driving sources or rotors vs. multiple wavelets , ). These mechanisms are inherently associated with 3D waves inside the heart muscle.
Filaments of 3D scroll waves can be either I-shaped, U-shaped, or O-shaped and may evolve in time as they interact with each other and the anatomic substrate (see Fig. 37.2E ). The interplay of stabilizing and destabilizing effects may result in intermittent complexity fluctuations, as shown in simulations by Zaritski and coworkers. Their simulations show that even in a homogeneous excitable medium, filament interaction may result in the self-organization of unstable filaments into weakly bound states that then decay into a complex, disorganized state again, as indicated by alterations of the total filament length. These intermittent temporal complexity fluctuations correspond to the observation of the transition between coarse and fine VF in clinical ECG, hypothesized to be related to the degree of synchronization of activation and the defibrillation threshold. Pathmanathan and Gray focused on the spatial organization of filaments during VF using a high-resolution biventricular heart model to study. Their simulations show that 3D filament interaction results in a transmural activity that is much more complex than what would be expected from surface observations alone. This suggests that much of the complexity of VF is hidden inside the ventricular wall, unlikely to be recovered using optical mapping alone.
To investigate the dynamics of electrical excitation and mechanical contraction experimentally, we have developed an ex vivo multimodal fluorescence imaging system, which allows for the measurement of membrane voltage, intracellular calcium, and mechanical strain on the surface of the heart. Intact isolated whole hearts are kept under physiologic conditions in a Langendorff-perfusion system , and allows studying the electrical and mechanical cardiac function of excised hearts in a controlled environment using optical and acoustic imaging techniques. Optical mapping uses fluorescent dyes to measure excitation wave dynamics on the heart surface with sensitive high-speed cameras at high spatial and temporal resolutions ( Fig. 37.3A ). Different dyes allow the recording of specific properties, such as the electrical membrane voltage or intracellular calcium Ca 2+ concentrations. The widely used potentiometric dyes di-4-ANEPPS and di-4-ANBDQPQ embed themselves into the cell membrane and exhibit a fast spectral shift in response to transmembrane voltage changes caused by the action potential. , Using optical long-pass filters to extract the fluorescent light beyond the emission spectrum maximum, the temporal change of the voltage is recorded as an approximately linearly proportional change (decrease) in filtered emission light. The fractional change is defined as the ratio of fluorescence intensity change over background intensity. The dynamics of intracellular Ca 2+ can be imaged using dyes that internalize within the cell, such as fluo-4 and rhod-2. Simultaneous mapping of membrane potential and intracellular calcium has been introduced by Efimov and colleagues and Choi and Salama. Simultaneous multiparametric optical mapping with a single camera can be achieved by frame-interleaved switching of excitation light wavelengths and multiband optical filters, as demonstrated by Lee and coworkers.
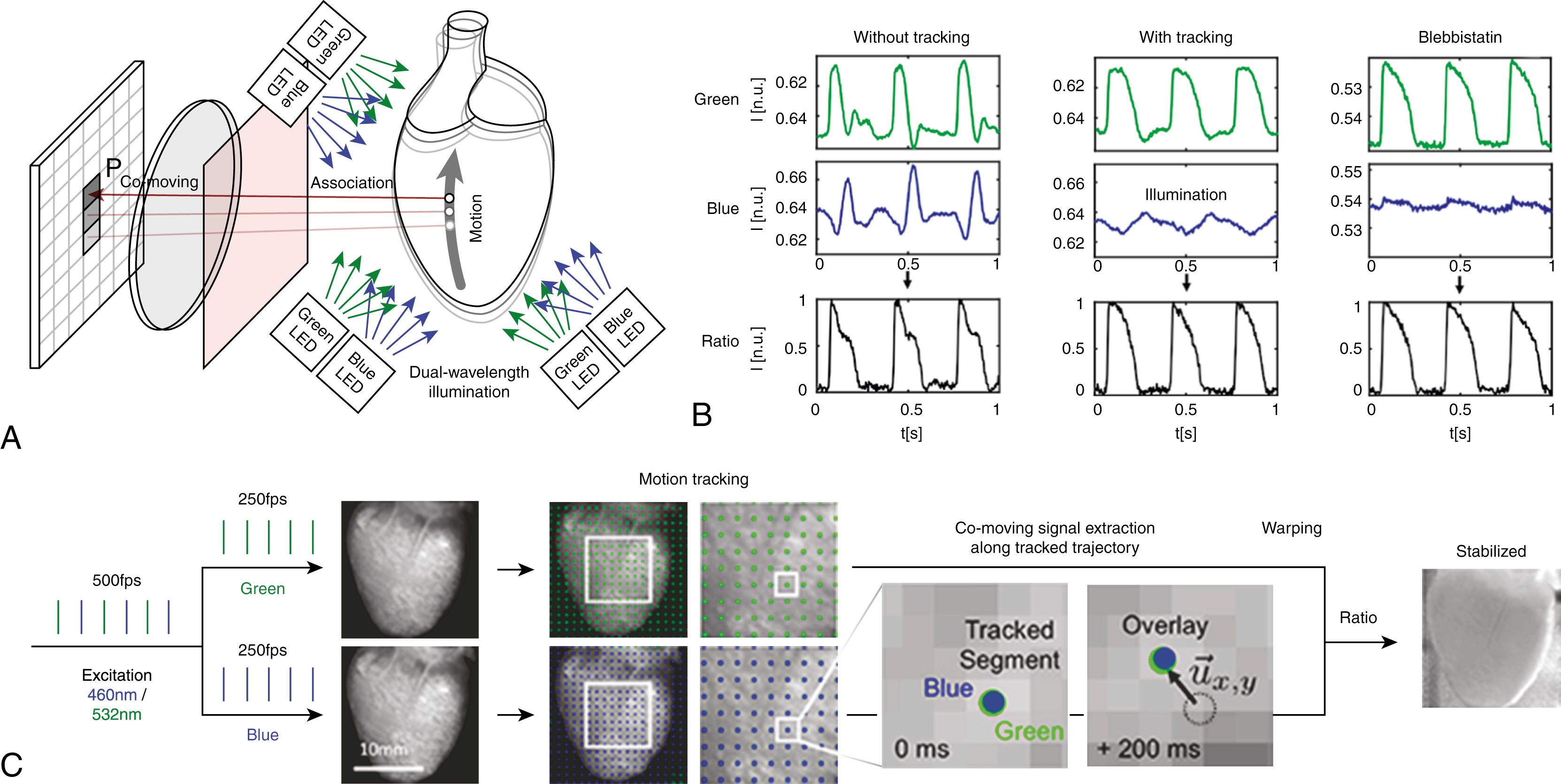
Motion artifacts are distortions in the fluorescence signal that occur when the contracting heart is in motion relative to the stationary imaging sensor. Two main mechanisms contribute to motion artifacts. First, fluorescence emitted from a single point or region on the moving heart surface will be imaged not on a single point or pixel on the image sensor but imaged along a trajectory. Vice versa, a single pixel of the image sensor records fluorescence signals from different regions of the heart surface as it moves relative to the sensor. Other regions of the heart may have very different dye staining and therefore different background fluorescence intensities. This background intensity variation can be significantly larger than the typical fractional change of the fluorescent dye. Thus a motion-induced change of background intensity can obscure the actual physiologic signal. Second, the incident excitation light is, in general, spatially nonuniform. As the heart contracts, it moves within a nonuniform excitation light field, which will modulate the fluorescence emission correspondingly. Consequently, fluorescence recorded from the moving heart entangles signals from electrophysiologic processes (e.g., transmembrane voltage and intracellular calcium, with signals representing the mechanical motion ).
Two types of strategies are pursued to cope with the effect of motion in optical mapping studies. In the first approach, the heart’s movement is suppressed to avoid or at least reduce motion-induced artifacts. The signal distortion caused by cardiac motion can be reduced mechanically, for example, by pressing the heart surface against a glass plate. This approach is simple and effective if the region of interest is small and the applied force is sufficiently small to avoid impaired perfusion and ischemia. Alternatively, pharmacologic motion suppression is widely used. An example of an excitation-contraction uncoupler is blebbistatin, which effectively inhibits motion. However, the effect of uncouplers on cardiac electrophysiology has to be carefully considered.
In the second approach, the heart is allowed to contract unhindered. The effect of motion is then compensated by imaging techniques, such as ratiometric imaging, or estimated and removed by signal possessing (e.g., motion tracking, or a combination of both). With the advancement of sensors, light sources, and signal processing, the interest in cardiac motion in optical mapping experiments shifts from the cause of signal distortion and artifacts that must be corrected for to a physiologic observable that shall be obtained from measurements.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here