Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
This chapter includes an accompanying lecture presentation that has been prepared by the authors: ![]() .
.
Alzheimer dementia is the most common form of dementia worldwide and is showing a steadily growing incidence, especially in developed countries.
Medical therapy is limited, and new treatment modalities are needed.
In a case report, chronic deep brain stimulation (DBS) of the fornix led to improvement in verbal, associative, and recollective memory in a cognitively nonimpaired individual.
Experiments in rodents suggest that DBS in the fornix circuit enhances memory and can exert certain neuroplastic and neuroprotective effects.
Results of preliminary studies in Alzheimer disease (AD) patients suggest that fornix-DBS may lead to a slowdown in cognitive decline and an increase in cerebral glucose metabolism in brain areas affected by AD in certain patients.
To date, fornix-DBS appears safe and well tolerated in this elderly and cognitively impaired patient population, based on a pilot study and a randomized controlled double-blind phase 2 study.
DBS of the nucleus basalis of Meynert (NBM) as the main cholinergic forebrain nucleus has been shown to be safe in pilot studies for Parkinson disease (PD)–associated dementia and mild AD.
New techniques such as focused ultrasound (FUS) may play a crucial part in the future treatments of AD to open the blood-brain barrier to clear amyloid or to selectively steer drugs to desired brain regions.
Alzheimer disease (AD) is the most common form of dementia and one of the leading health care challenges for the 21st century. It is estimated that currently more than 40 million people globally have dementia, with its prevalence expected to double every 20 years. Affected patients experience a relentless decline in memory and cognition that eventually ends in death within 3 to 9 years. The histopathologic hallmark is the presence of widespread formation of cortical extracellular neuritic (or “senile”) plaques consisting of amyloid β [Aβ] aggregates surrounded by reactive microglia and astrocytes as well as intracellular tangles. The cause and exact pathogenesis of AD are still unknown. Proponents of the amyloid hypothesis posit that Aβ-42 peptide initiates a cascade that eventually leads to failure of synaptic transmission and neuronal loss. Consequently, several therapeutic strategies have been developed either to decrease the production or to enhance amyloid clearing. However, none of these amyloid-based therapeutics have demonstrated compelling efficacy in randomized controlled trials. Development of effective treatment strategies for AD remains one of the biggest unmet needs in clinical neuroscience.
In this chapter we review the history of neurosurgical interventions for AD, insights into the pathophysiologic processes, results of neuromodulation trials, and the rationale for treating the network dysfunction in AD.
A variety of neurosurgical interventions for the treatment of AD has been studied for decades ( Table 131.1 ). Silverberg and colleagues used ventriculoperitoneal shunts in patients with AD to putatively enhance amyloid and protein tau clearance. The initial promising results from the pilot study were not replicated in the large-scale randomized controlled trial, which was stopped because of the futility of interim results. Goldsmith transposed the peritoneal omentum to the cortical surface on the premise that this could possibly improve cerebral hypoperfusion present in dementia. There is no controlled evidence of efficacy of this intervention. Vagus nerve stimulation has also been evaluated in small case series, and the efficacy of this intervention still remains to be evaluated in the setting of randomized trials.
| Intervention | Site | Proposed Mechanism | Outcome | Reference |
|---|---|---|---|---|
| Ventriculoperitoneal shunt placement | Intraventricular | Enhanced clearance of CSF amyloid and tau | Trial stopped because of futility of treatment | Silverberg et al. |
| Omental transplantation | Cortical surface | Enhanced cerebral perfusion to salvage “at risk” neuronal tissue | Small case series with unclear benefit | Goldsmith |
| Human mesenchymal stem cell injection | Bilateral hippocampi and right precuneus | Paracrine effects of stem cells to reduce amyloid plaques | No major adverse events in feasibility study | Kim et al. |
| Growth factor infusion (NGF and G M1 ganglioside) | Intraventricular | Rescue from ongoing cholinergic neuronal loss | Uncertain benefit with significant side effects | Eriksdotter et al. Augustinsson et al. |
| Gene therapy with CERE-110 | Nucleus basalis of Meynert | Rescue from ongoing cholinergic neuronal loss | Phase 2 clinical trial, ongoing (NCT00876863) | Rafii et al. |
| Vagus nerve stimulation | Cervical vagus nerve | Enhanced cognition | Some benefit in small case series No controlled trials |
Merrill et al. |
Several researchers have attempted to mitigate ongoing neuronal loss with the injection of growth factors, cellular grafts, and, more recently, gene therapy. The strategy of intraventricular growth factor injection (nerve growth factor [NGF] and G M1 ganglioside) showed promise but was not pursued further because of its uncertain efficacy and undesirable side effects such as pain and weight loss. Gene therapy for targeted NGF delivery to the nucleus basalis of Meynert (NBM) was found safe in the phase 1 trial. , A phase 2 trial of NGF gene therapy was conducted, but results have not been published yet. The injection of umbilical cord stem cells for reversal of amyloid deposition in the bilateral hippocampi and right precuneus is also in the initial stages of investigation and has been shown to be safe.
The classical “staging” of AD is based on the examination of pathologic changes (with regard to tau protein and Aβ) distribution in postmortem brains. The severity of pathologic changes correlates with the overall cognitive function of the individual. Longitudinal follow-up of affected patients with serial structural and amyloid positron emission tomography (PET) imaging has provided insights into the pathologic progression of disease in individual patients over time (intraindividual progression). According to a current hypothesis, the underlying pathologic process may involve two independent proteinopathies (tau and Aβ). , Deposition of tau protein is first detected in brainstem nuclei (e.g., locus caeruleus, raphe nuclei), and hypothalamus before amyloid can be detected in the cortex. The accumulation of Aβ is a slow process that can precede the appearance of hippocampal and cortical atrophy by years or decades. At a cellular level, abnormal protein accumulations produce oxidative stress and aberrant reentry of mature neurons into the G 1 and G 2 phases of the cell cycle. The consequent neuronal injury is believed to be responsible for failure of synaptic transmission and for producing the profound clinical symptoms of AD. ,
Certain brain networks are especially susceptible to dysfunction in AD. For example, the degree of dysfunction and atrophy are well correlated in the medial and lateral temporal regions. In contrast, the posterior cingulate gyrus and precuneus (elements of the default-mode network) have marked hypometabolism with relative structural preservation. In certain individuals at increased genetic risk for developing AD (apo E4 allele carriers), the evidence of suppressed glucose metabolism in the default-mode network can be detected several years before the onset of any clinical symptoms. This functional disconnection (network dysfunction) of structurally preserved brain regions is implicated in the marked cognitive and memory disturbance that characterizes AD. Restoring function in these activity-impaired but relatively anatomically preserved areas could be a useful approach in treating AD.
The modulation of memory networks during deep brain stimulation (DBS) surgery was discovered by serendipity during hypothalamic stimulation for treatment of morbid obesity.
Targeting the ventromedial and lateral hypothalamus to modulate appetite and hunger, intraoperative test stimulation in the awake patient produced a striking recall of autobiographic memories (flashback). This effect was time locked during active stimulation at 130 Hz for 90 microseconds and with an amplitude of 5 V or more. The trajectory of the electrode revealed to be anterior and parallel to the fornix was an important structure in the memory network. With subthreshold settings the cognitively nonimpaired patient showed unexpected large improvements in verbal, associative, and recollective memory during a 12-month follow-up period, which implied enhanced hippocampus-mediated memory function.
This sentinel observation was the basis for and led to a phase 1 study of fornix-DBS in six patients with mild to moderate AD ( Fig. 131.1 ). The bilateral target chosen was 2 mm anterior and parallel to the fornix column, so as to avoid injury to the fibers of the fornix ( Fig. 131.2 ). The cognitive outcomes were assessed with the Alzheimer’s Disease Assessment Scale–Cognition subscale (ADAS-Cog) and the Mini Mental State Examination (MMSE). At 12 months, the ADAS-Cog score increased (worsened) compared with baseline; however, the increase was smaller than the average annual worsening of AD patients. This indicates a slowdown of cognitive decline, and one patient even exhibited improved ADAS-Cog scores. Furthermore, the patients with less severe preoperative cognitive impairment had milder decline in ADAS-Cog score after 12 months of stimulation. Others investigators were able to recreate similar effects in a single patient case report.
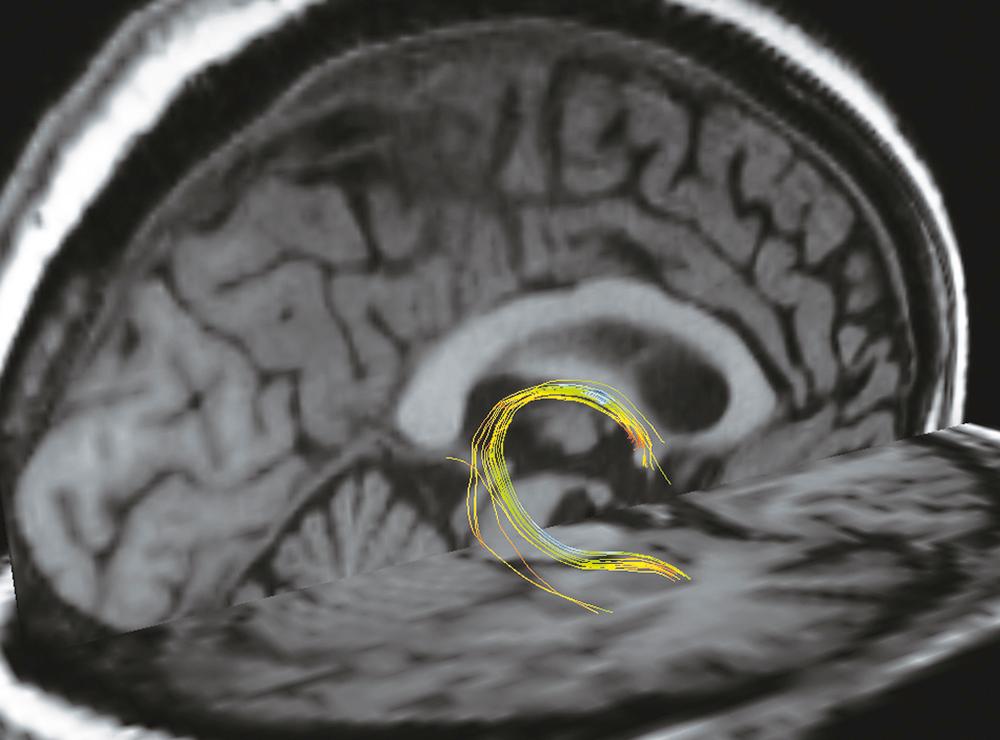
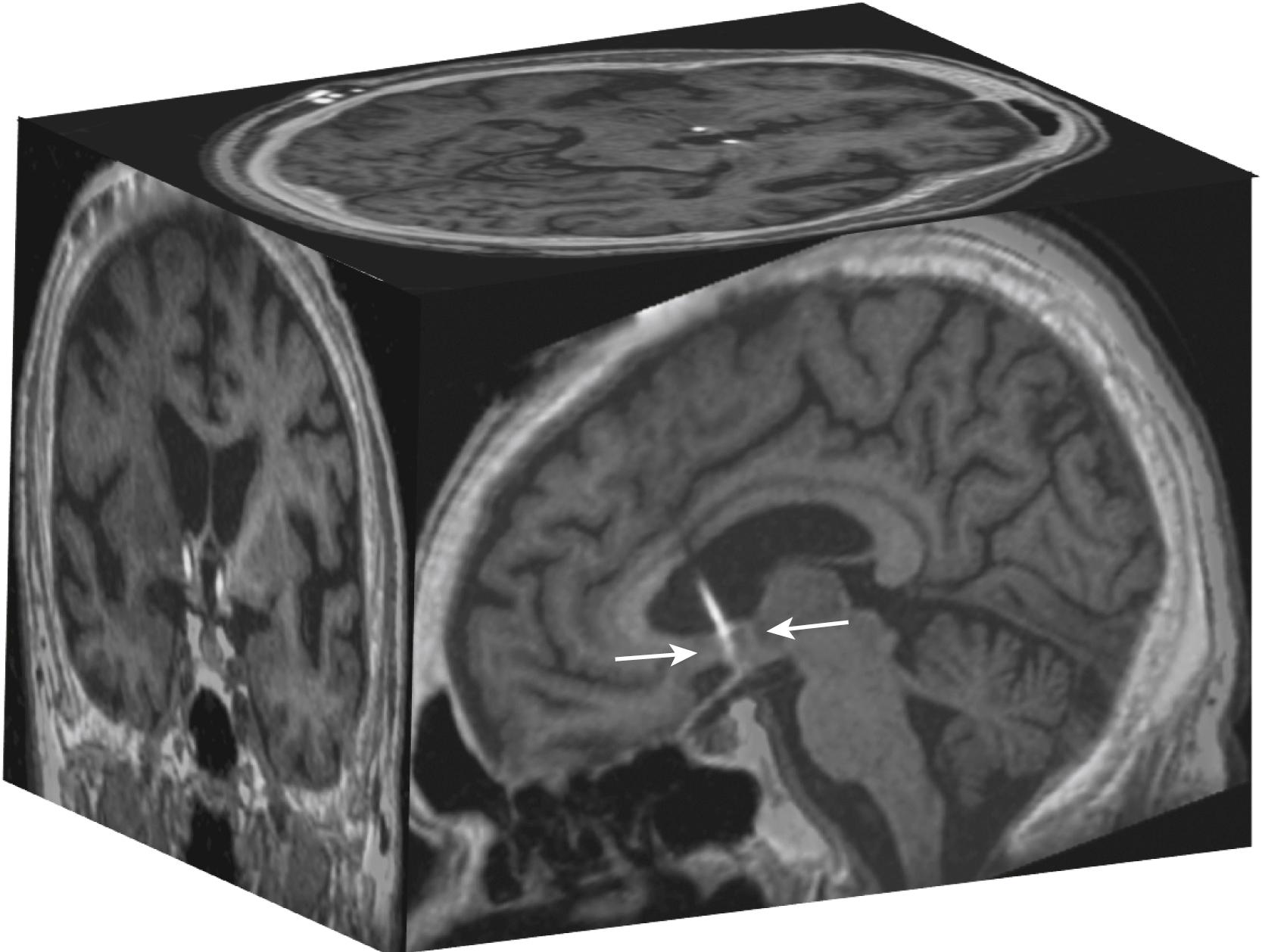
This preliminary work also suggested a possible trend toward a slower rate of cognitive decline in patients with AD who receive fornix stimulation, which warranted a multicentric prospective randomized controlled double-blind phase 2 follow-up study (Advance I trial) ( Box 131.1 ).
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here