Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
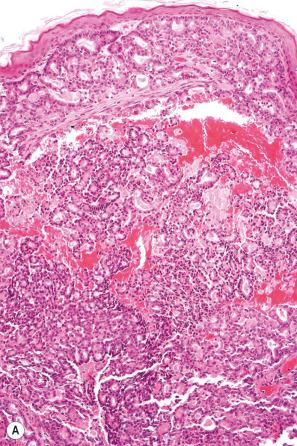
Secondary involvement of the integument by a malignant tumor may represent a metastatic phenomenon (such as following vascular or lymphatic emboli) or occur as a direct consequence of contiguous spread through tissue spaces or lymphatic and vascular channels ( Figs 30.1 and 30.2 ). In addition, tumor deposits may be accidentally implanted during surgical procedures. Rarely, cutaneous metastasis may be the presenting feature of a visceral malignancy.
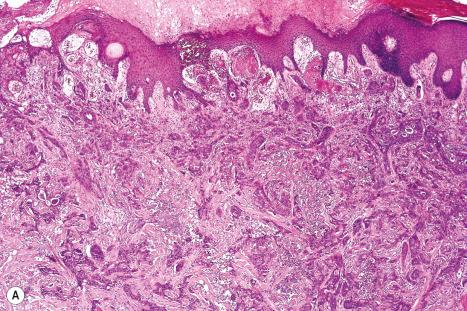
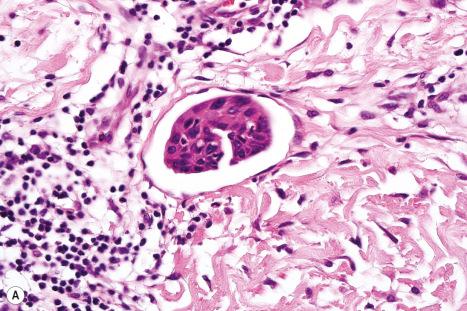
On histologic grounds alone, the differential diagnosis between metastatic tumors in the skin and primary cutaneous tumors can be challenging. This discrimination is of a paramount importance due to the profound implications for prognosis and subsequent therapy. Detection of cutaneous metastases usually indicates disseminated disease and a poor prognosis.
Determining the origin of the tumor is often very difficult and sometimes impossible, although careful use of ancillary techniques – particularly immunohistochemistry and, very rarely, electron microscopy – can give the pathologist some useful pointers in the right direction.
When unusual intradermal tumors are encountered, particularly in elderly patients, a high index of suspicion is always necessary, since the unwary can easily mistake a small deposit of metastatic breast duct carcinoma for an adnexal tumor. Care with tissue specimens, by both the clinician and the pathologist, is always mandatory. Poorly fixed lymphomatous infiltrates, for example, may be misdiagnosed morphologically as cutaneous neuroendocrine carcinoma, while artifactual crushing may result in confusion with metastatic small cell carcinoma. Ancillary studies such as immunohistochemistry should be engaged when histologic detail is compromised.
The more frequent sites for secondary tumor deposits are lymph nodes, liver, lungs, adrenals, brain, bone, ovaries, and kidneys with some variation based on primary tumor type. The skin is a relatively uncommon site for presentation of metastasis; recorded frequencies have varied from 0.7% to 9% of all cases of metastatic malignant disease. A meta-analysis including over 20 000 patients with carcinoma, and excluding melanoma, found the incidence of cutaneous metastases to be 5.3%. Considering the fact that the skin is the largest organ in the body, it is unclear why this relatively low incidence of metastasis from internal malignancy is observed. As initially described in 1889 by Stephen Paget in his ‘seed and soil’ hypothesis, the tumors preferentially metastasize to those organs with an intrinsically favorable environment. It is therefore possible that the skin may provide the proper environment for colonization and survival of only a few types of tumors. The role of the unique microenvironments composed of various immune and stromal cells present at different body sites in the metastatic process is the topic of much research.
Cutaneous metastases present most commonly a few months or years after the primary tumor has been diagnosed. There are rare reports of metastatic tumors arising decades after the diagnosis of the primary neoplasm. Cutaneous metastases develop most commonly at the same time as internal metastases. Less frequently, a metastasis is diagnosed concurrent with the primary tumor or represents the initial manifestation of the disease. In a study by Brownstein and Helwig, skin metastases were the presenting sign of the disease in 37% of men and in 6% of women. This discrepancy may reflect the fact that although metastatic breast carcinoma represents the most common skin metastasis in women, currently breast cancer is usually diagnosed fairly early in the course of the disease as opposed to many other internal malignancies. Cutaneous metastases from some primary sites appear more likely to be the first sign of the disease. These include lung cancer (60% of cases), renal cancer (53% of cases), and ovarian cancer (40% of cases), but the incidence of skin metastasis as the initial presenting symptom is likely falling dramatically from these historical levels with earlier clinical detection of internal disease in contemporary medicine. A more contemporary study suggests that where cutaneous metastases do occur, they can be the initial sign of metastases beyond the regional lymph nodes. Although cutaneous metastases may present at any age, the greatest incidence is in the fifth to seventh decades.
The relative frequencies of the underlying primary neoplasms largely reflect the incidence of the various tumors that occur in humans. In Western communities, the most common sources of cutaneous metastases in males include the lungs (24%), large intestine (19%), melanoma (13%), squamous carcinoma of the oral cavity (12%), kidney (6%), stomach (6%), and esophagus (3%) ( Fig. 30.3 ). In females, the primary tumor site is most often the breast (69%), while other important sources include the large intestine (9%), melanoma (5%), ovaries (4%), and uterine cervix (2%) ( Fig. 30.4 ). With the increased incidence of cigarette smoking in females, carcinoma of the bronchus is of growing importance. Much less common primary sites have been reported including thyroid, adrenal, endometrium, urinary bladder, and pancreas. Prostate carcinoma, despite its high incidence in men, is among the least common primary site to result in cutaneous metastases, perhaps a practical demonstration of the seed and soil hypothesis discussed above.
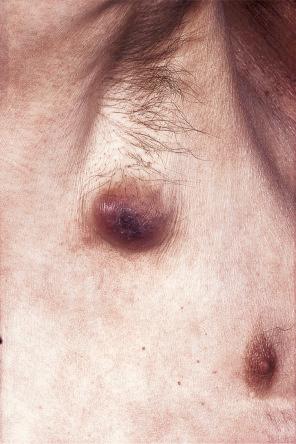
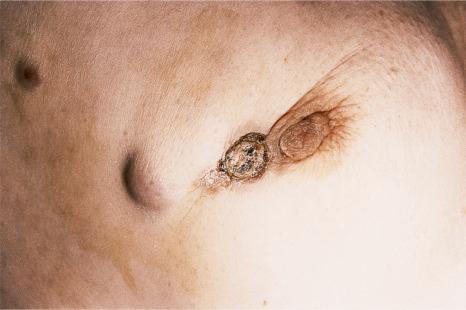
In a study by Lookingbill, melanoma was the most common source of metastatic disease in the skin in men and the second most common source for women ( Fig. 30.5 ). A study from India has shown that the most common primary sites of cutaneous metastasis are the lung and esophagus in men and the breast and ovaries in females.
With the exceptions of leukemia and lymphoma, cutaneous metastases from underlying malignancies in children are very rare. However, around 50% of malignant cutaneous tumors in children represent metastatic disease. Interestingly, and in contrast to adults, cutaneous metastases represent the first manifestation of the disease in up to 84% of cases.
The most common childhood tumors metastasizing to the skin are rhabdomyosarcoma and neuroblastoma, reflecting the incidence of these tumors in this age group ( Fig. 30.6 ). Neuroblastoma is most often seen in neonates. In this age group, neuroblastoma presents with skin metastases in up to 32% of cases.
Rare tumors metastasizing to skin in children include osteosarcoma, choriocarcinoma, Ewing sarcoma, malignant rhabdoid tumor, melanoma, paraganglioma, and nasopharyngeal carcinoma. Choriocarcinoma presenting as a metastatic tumor in a neonate is associated with a primary placental tumor, which can also spread to the mother. As with adults, central nervous system tumors only exceptionally spread to the skin in children. In the vast majority of children with metastatic carcinoma in the skin, the source of the primary tumor is almost invariably determined after thorough and comprehensive investigation. An unknown primary is truly exceptional.
Cutaneous metastases in young adults are rare, reflecting the general low incidence of cancer in this age group.
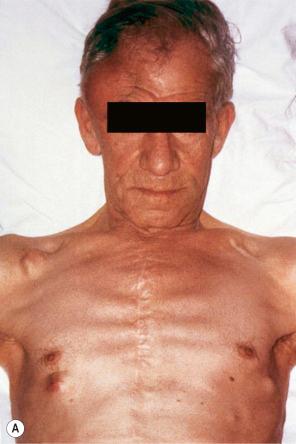
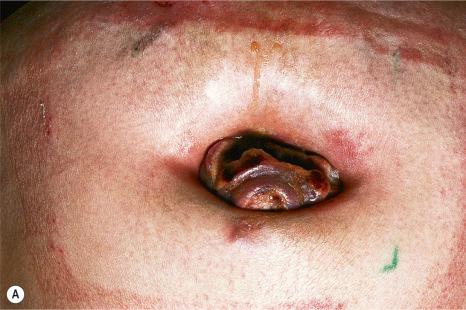
Although not always the rule, the location of a cutaneous metastasis often reflects the site of the underlying tumor. The anterior chest, abdomen, head, and neck regions are the most common sites for metastatic tumors. Multiple sites of cutaneous metastases (involving two or more anatomic regions) have also been reported. Metastases to the skin of the face and neck are most often associated with a squamous carcinoma of the oral cavity, although lung, kidney, and breast may also represent rare sources of such metastases. Carcinoma of the breast most frequently spreads to the anterior chest wall, while bronchial and lung tumors tend to metastasize to the chest wall and upper extremities ( Fig. 30.7 ). Gastrointestinal tumors usually involve the anterior abdominal wall but may present in other areas such as the face and scalp ; pelvic neoplasms show a predilection for the perineal region. Primary tumors of the prostate, bladder, and colon can spread to the penis or scrotum. Intra-abdominal tumors, including those of the stomach, colon, ovary, endometrium, urinary bladder, fallopian tube, prostate, cervix, and pancreas, sometimes metastasize to the skin of the umbilicus to form the so-called Sister Mary Joseph nodule ( Fig. 30.8 ). Rarely, a metastatic breast carcinoma or mesothelioma can present as a Sister Mary Joseph nodule. Umbilical metastases represent the first manifestation of disease in about 14% of cases.
Metastases to the limbs are uncommon, most often resulting from spread of primary cutaneous melanoma. Sometimes, these present in a subungual location. Subungual metastases can also complicate carcinoma of the stomach, lung, esophagus, kidney, colon, and breast. Subungual metastases from a choriocarcinoma have also been reported.
Melanoma and neoplasms associated with vascular invasion (e.g., renal cell carcinoma, thyroid follicular carcinoma, and choriocarcinoma) lack such a tendency towards regional localization ( Figs 30.9 and 30.10 ).
Some tumors are characterized by a predilection to metastasize to certain sites. For example, ocular melanoma selectively spreads to the liver. In addition to skin, lung, bowel, and brain are also common sites for melanoma metastasis.
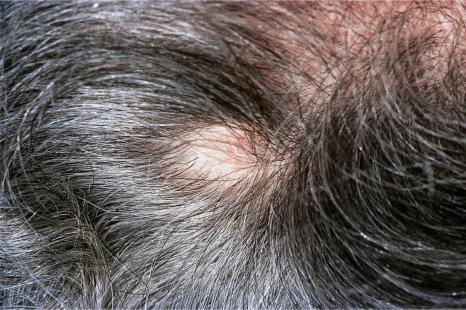
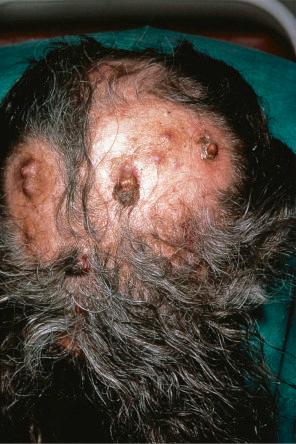
Involvement of the scalp is not uncommon, accounting for up to 5% of all metastases. Sometimes, it may represent a primary manifestation of the underlying malignancy. In a large series of malignant tumors of the scalp from Taiwan, metastatic disease accounted for 12.8% of cases. The most common primary site in both men and women in this series was the lung. Tumors most likely to spread to the scalp include those of the breast, lung, colon, stomach, and kidney ( Figs 30.11 and 30.12 ). Scalp metastases are particularly common in renal cell carcinoma. A plaque of alopecia is the usual manifestation, but occasional cases of metastatic breast carcinoma without alopecia have also been described. Amongst sarcomas, leiomyosarcoma shows a particular propensity for scalp metastases.
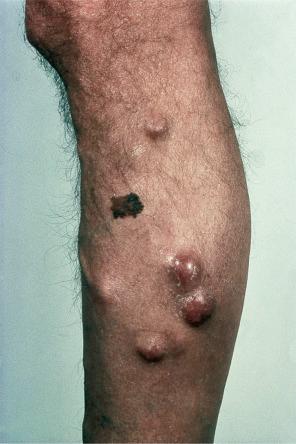
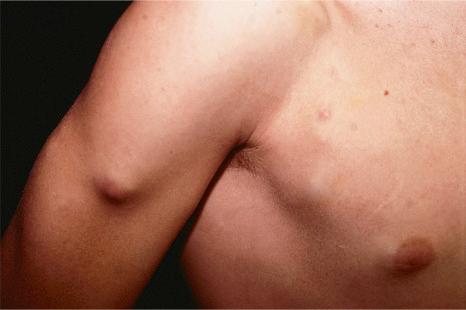
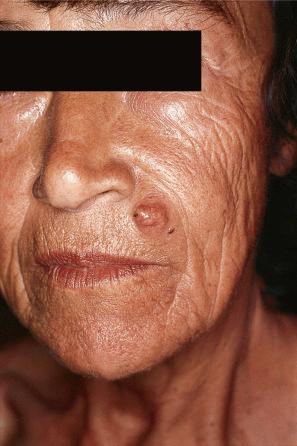
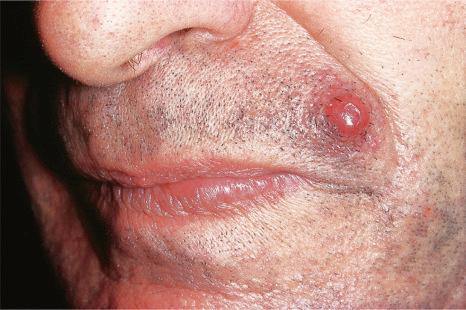
Of the various patterns of cutaneous metastasis, the most common is development of a nodule or group of nodules (frequently in a surgical scar), typically painless, rapidly growing, freely mobile, and often flesh colored ( Figs 30.13–30.16 ). Most lesions are less than 3 cm in greatest diameter. Unless traumatized, they rarely ulcerate. Other patterns of cutaneous metastases include micropapules, plaques, and lesions simulating scars. Scarlike lesions can be seen in metastases from the breast, stomach, lung, and kidney.
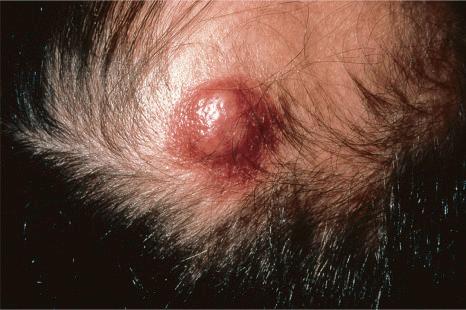
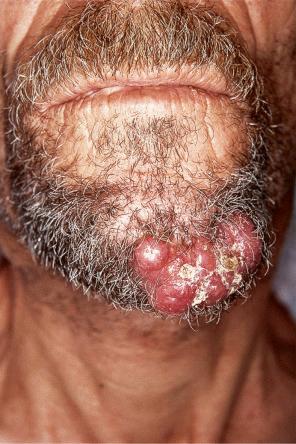
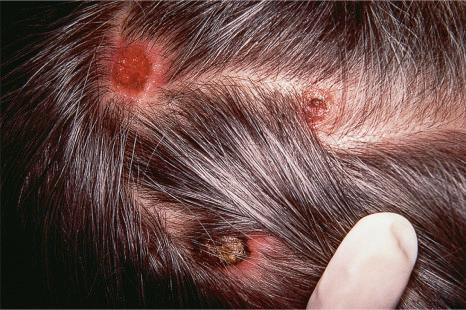
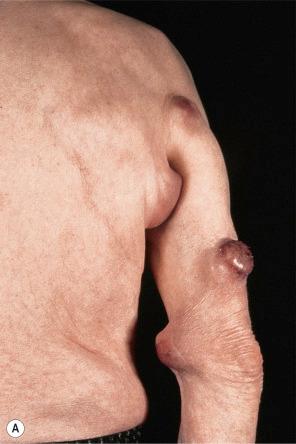
The clinical appearance of some metastases mimics a number of benign dermatological conditions such as erythema annulare centrifugum, contact dermatitis, cellulitis, kerion, chancre, follicular cyst, condyloma, and even a cutaneous horn. Other clinical presentations include zosteriform and elephantiasiform lesions, facial lymphedema ( Fig. 30.17 ), and hemorrhagic blisters. Zosteriform metastases have occurred with many malignancies including melanoma, squamous cell carcinoma, porocarcinoma, angiosarcoma, and carcinomas of the breast, ovary, colon, bronchus, tonsil, prostate, and bladder, and transitional carcinoma of the renal pelvis.
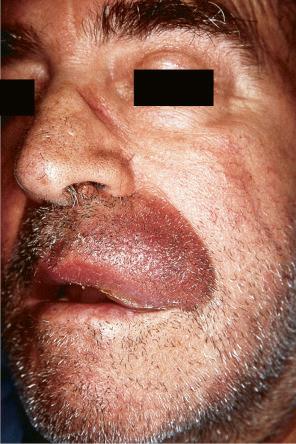
Cutaneous metastasis can also mimic primary cutaneous tumors such as pyogenic granuloma, cylindroma, keratoacanthoma, blue nevus, Kaposi sarcoma, angiosarcoma, and melanoma.
Hemorrhagic cutaneous metastatic nodules usually imply a primary renal cell carcinoma, thyroid follicular carcinoma, or choriocarcinoma. Pigmented metastases can mimic a melanoma. The pigmented nature of the tumor is most likely due to epidermotropic metastasis that causes damage to the epidermis and subsequent pigmentary incontinence.
Exceptionally, metastases to benign cutaneous lesions such as nevi have been documented. Unusual presentation of metastases includes spread to skin with radiation changes and to sites of surgical procedures. The latter include scars following removal of abdominal organs or mastectomy sites, percutaneous biopsies or fine needle aspirates, pericardiocentesis, thoracocentesis, and sites of implantation of biliary catheters.
There are other modes of presentation of cutaneous metastases which are particularly, but not exclusively, related to breast cancer. Inflammatory cutaneous metastasis (carcinoma erysipelatoides) is fairly rare and results from massive lymphatic obstruction by tumor with associated edema. It presents as a tender, erythematous, warm plaque ( Fig. 30.18 ) located in most cases on the anterior chest wall (particularly in females with pendulous breasts). Clinically, it may be misdiagnosed as cellulitis or erysipelas, but theoretically, at least, it may be distinguished by the absence of fever and leukocytosis. Inflammatory cutaneous metastases have also been described with carcinoma of the prostate, lung, large bowel, pancreas, stomach, kidney, ovary, tonsil, parotid, uterus, esophagus, melanoma, and bladder. Also associated with a poor prognosis are breast metastases presenting on the anterior chest wall as violaceous telangiectatic papules, vesicles, or nodules resembling lymphangioma circumscriptum. This is known as carcinoma telangiectaticum, and it may also be seen rarely with lung carcinoma. The lesions are sometimes purpuric and hemorrhagic and resemble a vasculitic process. Other manifestations of breast cancer are peau d'orange , representing dermal edema due to obstruction of superficial cutaneous lymphatic vessels and carcinoma en cuirasse , an intensely sclerotic plaque of tumor ( Fig. 30.19 ).
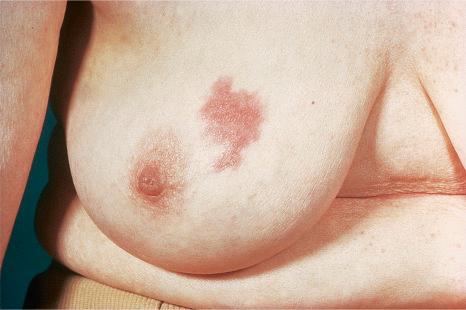

Metastatic breast carcinoma infrequently presents in the inframammary crease in a characteristic pattern mimicking dermatitis or resembling a primary epithelial tumor including basal cell and squamous cell carcinoma.
A clinical variant that sometimes causes diagnostic confusion is the sclerodermatous (eburneous) metastasis usually associated with tumors having a dense fibrous stroma, such as carcinoma of the breast and pancreas. The lesion is usually nontender and nonpruritic, and is most often located on the scalp with associated alopecia, so-called alopecia neoplastica. The latter may be confused with lichen planopilaris, pseudopélade, lupus erythematosus, alopecia areata, keloid, and morpheaform basal cell carcinoma. Alopecia neoplastica has also been described in association with metastatic colonic carcinoma to the scalp.
Neonates with metastatic neuroblastoma often present with multiple firm, blue-gray nodules in a typical appearance described as the ‘blueberry muffin’ syndrome. This presentation, however, is not specific and can also be seen in lymphoma, leukemia, vascular tumors, and congenital infections.
Some tumors have only very exceptionally been reported with skin metastases. Many of these primary tumors are themselves quite uncommon. These include glioblastoma, glioma, eccrine porocarcinoma or hidradenocarcinoma, pheochromocytoma, ameloblastoma, parotid adenocarcinoma (including malignant pleomorphic adenoma), chordoma, malignant thymoma, seminoma, Leydig cell tumor, papillary serous carcinoma of the uterus and of the ovary, ovarian malignant Brenner tumor, gallbladder adenocarcinoma, adrenocortical carcinoma (including a case presenting with cutaneous metastasis 30 years after the primary), transitional bladder carcinoma, hepatocellular carcinoma, carcinoid tumor, neuroendocrine tumors, mesothelioma, anaplastic carcinoma of the thyroid, medullary thyroid carcinoma, Sertoli cell tumor, salivary adenoid cystic carcinoma, cholangiocarcinoma, paraganglioma, and pancreatic mucoepidermoid carcinoma. Other cases include metastases of myoepithelial carcinoma including one case in a child, well-differentiated fetal adenocarcinoma, adenocarcinoma of rete testis, malignant sacrococcygeal teratoma, prostatic small cell carcinoma, mucinous sweat duct carcinoma, adenocarcinoma arising in a longstanding nevus sebaceus of Jadassohn, small-cell neuroendocrine carcinoma of the uterine cervix, squamous cell carcinoma of the ureter, an exceptional case of acinic cell carcinoma of salivary gland presenting with cutaneous metastases 20 years after the primary, and a malignant blue nevus of the scalp with distant metastasis to the back.
Sarcomas mainly involve the skin as a result of direct extension from the underlying subcutis or deeper soft tissues. Cutaneous metastatic sarcoma, which represents less than 3% of all cutaneous metastases, has been described in association with leiomyosarcoma arising in different organs including the uterus and small intestine, epithelioid sarcoma, chondrosarcoma, osteosarcoma including a postirradiation lesion, undifferentiated pleomorphic sarcoma, giant cell tumor of bone, fibrosarcoma, rhabdomyosarcoma, gastrointestinal stromal tumor, synovial sarcoma, alveolar soft part sarcoma, Ewing sarcoma, myxofibrosarcoma, angiosarcoma, PEComa, and pleomorphic liposarcoma. In the authors' experience, metastatic leiomyosarcoma is most commonly encountered. A cutaneous metastasis derived from the chondrosarcomatous component of a metaplastic (sarcomatoid) breast carcinoma has also been described. A case of cutaneous metastasis from osteosarcoma has been reported as the first manifestation of disease. Atrial myxoma may embolize to the skin, particularly in the context of Carney complex.
The presence of cutaneous metastases is usually (although not invariably) an ominous sign. The average survival is about 6 months after diagnosis. However, some patients may have long survival. This reflects both improvement in cancer therapy and the biological behavior of the individual tumor. For example, neuroblastoma in children can mature or undergo complete regression, and patients with renal cell carcinoma can have a long survival following removal of the primary tumor and the metastases. In general, patients with cutaneous metastases from the upper digestive tract, upper respiratory tract, lung, and ovary have very poor survival. In a study involving 200 patients with cutaneous metastases, Schoenlaub and coauthors reported median survival times of 13.8 months for breast cancer, 2.9 months for lung cancer, and 6.5 months for all other cancers.
Histologic interpretation of a cutaneous metastasis depends upon a complete knowledge of the patient's clinical history (past and present), coupled with adequate experience of the wide range of histologic features of tumors.
The most challenging differential diagnosis of cutaneous metastases is with primary cutaneous tumors, especially with those of adnexal origin. On histologic grounds alone, several features are traditionally used to distinguish between these entities. One of the features in favor of a primary cutaneous tumor is continuity with the epidermis or growth into skin appendages (i.e., an in situ component). However, it is important to remember that tumors such as hidradenocarcinoma are strictly intradermal and do not connect with overlying epidermis, and also that epidermotropic metastases are not infrequent. Another useful clue for a primary cutaneous tumor is identification of the benign counterpart within the lesion (such as areas of benign spiradenoma within a spiradenocarcinoma). One finding that is seldom noted in the literature is the more frequent presence of entrapped, ‘passenger’ melanocytes within primary cutaneous lesions in contrast to metastatic tumors where they are rarely seen. The most useful histologic features in favor of a cutaneous metastasis include location in the deep dermis or subcutaneous tissue, multifocality, and more frequent presence of lymphovascular invasion. It is important to be aware that while these histologic findings may be useful in the diagnosis of cutaneous metastases they are not completely reliable. They should be used in combination with the clinical history and, in the majority of cases, immunohistochemical studies that may help in identification of the tumor origin. On some occasions, however, the finding of poorly differentiated adenocarcinoma or squamous carcinoma will permit only a list of the more likely sites of the primary lesion.
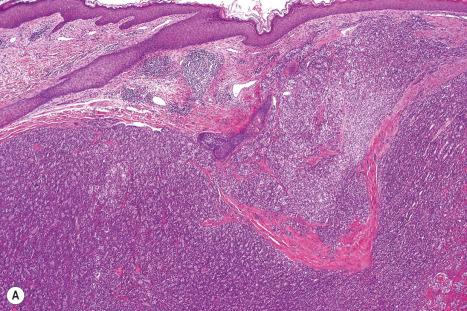
Immunohistochemistry is of particular value in distinguishing between various carcinomas, melanomas, lymphomas, and pleomorphic sarcomas ( Figs 30.20–30.24 ). With respect to metastatic adenocarcinoma, however, it is more difficult to accurately predict the likely site of the primary neoplasm. With a few specific exceptions, including thyroid (thyroglobulin), prostatic carcinoma (prostate specific antigen, prostatic acid phosphatase), and hepatocellular carcinoma (hepatocyte paraffin 1 and α-fetoprotein), if the precise diagnosis is not apparent after careful scrutiny of hematoxylin and eosin stained sections then it is less than likely that immunohistochemistry will be of definite help in resolving the problem.

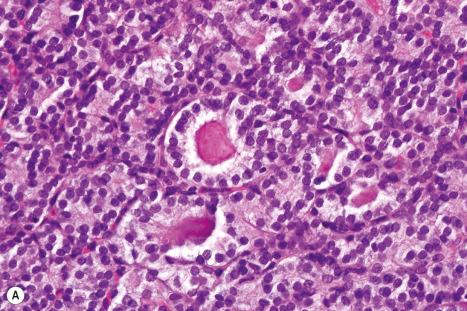
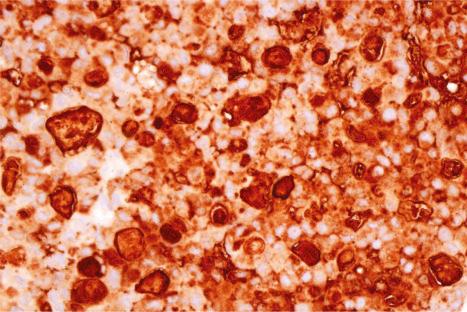
Immunohistochemistry can sometimes play an important role in the assessment of metastasis associated with an occult primary tumor. An enormous range of antibodies is now available, and a large proportion is suitable for use with routine, formalin-fixed, paraffin-embedded tissues. Difficulties, however, are often experienced due to lack of specificity or poor sensitivity. It is essential that the practitioner be aware of the often wide range of cell types that may express any particular antigen and that the use of panels of immunohistochemical studies is more helpful than single stains. Virtually, all new markers initially heralded as highly specific in an initial publication follow a natural history of decreasing specificity in subsequent reports.
Distinction between primary cutaneous and metastatic neuroendocrine carcinoma is usually made by the presence of positive dotlike perinuclear staining for low molecular weight cytokeratin (CK) 20 and neurofilament protein expression in the former and absence of staining in the latter. Apart from the skin, CK20 is only expressed consistently in neuroendocrine carcinomas of salivary gland origin. In contrast, bronchial neuroendocrine carcinoma expresses CK7. Although these findings are useful in the differential diagnosis in most cases, caution is recommended before basing a diagnosis on a single immunohistochemical result. Some cases of lung small cell carcinoma express CK20, and there are also reported cases of cutaneous neuroendocrine carcinoma (Merkel cell carcinoma) that are CK20-negative. The use of thyroid transcription factor 1 (TTF-1) can also be of help in the distinction between a primary cutaneous and a metastatic bronchial neuroendocrine carcinoma: TTF-1 is negative in the former but present in the latter. However, occasional cases of Merkel cell carcinoma have been shown to be positive for TTF-1. Although CK20 is most commonly used, other cytokeratins, such as CAM 5.2, also label the tumor cells of primary cutaneous neuroendocrine carcinoma. A further marker has been described as useful in the distinction between pulmonary small cell carcinoma and Merkel cell carcinoma. MASH-1 (raised against Achaete-scute complex-like 1) is an important protein in the development not only of the brain but also of the neuroendocrine system. MASH-1 tends to be positive in pulmonary small cell carcinoma in up to 83% of cases but appears to be uniformly negative in Merkel cell carcinoma.
Historically, gross cystic disease fluid protein 15 (GCDFP-15), estrogen receptor (ER), and progesterone receptor (PR) have been used as markers for breast differentiation. However, it is important to note that GCDFP-15, ER, PR, and Her2/neu have all been shown to be also expressed in cutaneous adnexal neoplasms, reflecting the close relationship between the sweat gland and the breast. For this reason, these markers should be used with caution if utilized in an attempt to distinguish primary cutaneous lesions from metastatic breast tumors.
Staining for other low molecular weight keratins such as CK7 is of limited value in the differential diagnosis of metastatic carcinoma. Although this marker is absent from many carcinomas – including those originating in prostate, colon, kidney, and thymus – it is present in the majority of carcinomas arising in other organs, and therefore its specificity is very limited. CK7 has been shown to be expressed in both primary cutaneous adnexal neoplasms and cutaneous metastases, mainly from the lung and breast. However, the pattern of CK7 labeling is distinctly different between the two categories: only focal in primary adnexal neoplasms, present in the inner luminal cells lining tubules and cysts, whereas there is strong and diffuse staining in metastatic adenocarcinomas. However, using CK7 and CK20 in combination with GCDFP-15 can be useful in determining whether a case of extramammary Paget disease is primary or metastatic. Primary perianal and extramammary Paget disease tends to be positive for CK7 and GCDFP-15 and negative for CK20, while metastatic rectal adenocarcinoma is usually negative for the former two markers and positive for the latter. The transcription factor GATA3 is seen in over 80% of both breast and urothelial carcinomas.
Gastrointestinal metastasis to the skin can often be suspected by the presence of ‘dirty’ necrosis (intraluminal necrotic debris), commonly seen in primary and metastatic intestinal adenocarcinoma. The pattern of strong CK20 expression and lack of CK7 expression favors a metastasis from an intestinal primary tumor. Carcinoma originating in the hepatobiliary or upper gastrointestinal tract may be also CK7-positive and/or CK20-negative. CDX2, a homeobox gene encoding an intestinal epithelial transcription factor, initially considered a specific marker for gastrointestinal tract origin, has also been reported in other tumors, including ovarian mucinous tumors of the intestinal type, pulmonary mucinous adenocarcinoma, bladder adenocarcinoma, and endometrial carcinoma, and rare tumors of the lung (non-mucinous), bladder (urothelial), and head and neck. Villin is an additional marker that may help to distinguish between colorectal (positive) and ovarian metastatic deposits (negative).
Cutaneous metastases from the lung often express CK7 and lack CK20. Additionally, approximately 75% to 85% of primary pulmonary adenocarcinomas express TTF-1, but is also seen in thyroid carcinomas. Napsin A is expressed in up to 80% of pulmonary adenocarcinomas as well as ovarian clear cell and renal cell and papillary carcinomas.
The great majority of adrenocortical neoplasms will coexpress Melan A/MART1, inhibin, and calretinin. Prostatic carcinoma will usually express PSA, PAP, androgen receptor (AR), and the transcription factor NKX3-1. The transcription factor PAX8 is a sensitive marker for carcinomas of the ovary, thyroid, and kidney.
Formalin-fixed, paraffin-embedded tissue may be reprocessed for electron microscopy, and ultrastructural investigation of such material may throw light on the likely source of the primary neoplasm. Traditionally, if possible, a small portion of fresh tissue was processed for electron microscopy. Identification of cellular markers, such as desmosomes, intracytoplasmic mucin, melanosomes, and membrane-bound neurosecretory granules, may be helpful in pointing to the correct diagnosis. Use of this technique to identify tumors of uncertain differentiation has become exceedingly rare with the wide availability of numerous immunohistochemical markers.
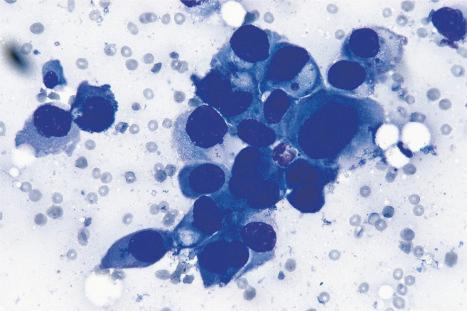
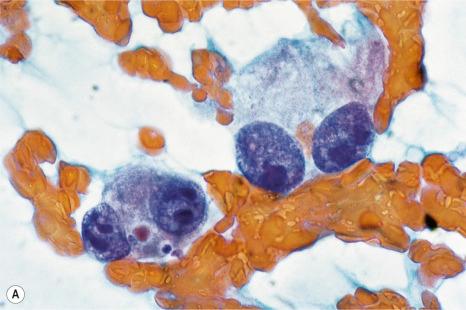
Over the past few decades, the enthusiasm for fine needle aspiration cytology has grown. The technique in many circumstances has proven to be a highly satisfactory alternative to more conventional excisional, punch, or tru-cut biopsy specimens. In the dermatopathology setting, it can be of particular value in assessing potential metastatic tumors, especially if the patient is unfit or unwilling to undergo further operative procedures ( Figs 30.25 and 30.26 ). Fine needle aspiration cytology is a fast, inexpensive, and effective method of confirming the diagnosis in the majority of cases. Where appropriate, the technique also permits the application of immunohistochemical investigations, fluorescent in situ hybridization (FISH), and polymerase chain reaction (PCR)-based techniques.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here