Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Necrotic liver tissue, whether before or after liver transplantation, is a serious condition that requires immediate attention, because it can result in considerable morbidity or mortality. Although the most effective treatment for a massively necrotic liver is liver replacement, the indications, the sequence, and the timing of procedures require considerable medical and surgical skills to achieve favorable outcomes.
Liver infarction typically results in a sudden increase in liver enzyme levels. When the amount of liver tissue involved is significant, a septiclike picture can ensue, with fever, leukocytosis, hemodynamic instability, or shock. The progression can be gradual or with an initial stable phase followed by abrupt deterioration. The liver failure can result in encephalopathy, lactic acidosis, which can further exacerbate the shock, and renal failure. This picture can be reversed by an urgent liver transplant. The septic picture is caused by massive cytokine release into the circulation from the necrotic liver. In this situation the shock can be temporarily reversed by emergency hepatectomy, which allows limited waiting time for a liver allograft. Imaging can show areas of low density or a small, collapsed liver on computed tomography scan, or a coarse echotexture on ultrasonography. Liver infarction can be localized. This can result in atrophy of a liver segment or lobe or liquefaction of the involved area, with formation of bilomas or abscesses. Localized necrosis can be amenable to surgical resection, drainage, or conservative treatment.
Emergency total hepatectomy is indicated in selected cases of acute liver necrosis from fulminant hepatic failure of the native liver or necrosis if the patient is a candidate for urgent liver transplantation. Patients considered for emergency hepatectomy have liver failure and a toxic syndrome with hemodynamic instability and severe refractory metabolic acidosis because of massive necrosis. In this situation, removal of the necrotic liver can result in temporary improvement in acidosis and hemodynamic status and other aspects of multiorgan failure; this makes transplantation feasible. However, the anhepatic state, which requires aggressive administration of fluid, coagulation factors, vasopressors, aggressive continuous venovenous dialysis, and other agents, can be sustained up to a maximum of 48 to 72 hours, beyond which a secondary deterioration occurs, this time refractory, leading to circulatory failure and death from herniation or multiorgan failure. Continuous venovenous dialysis effectively controls the intracranial pressure for 2 to 3 days; the intracranial pressure sharply rises thereafter, as a terminal event. The timing of emergency hepatectomy is assessed on a case-to-case basis. Because it brings only temporary improvement, it is best to perform the hepatectomy when there is a donor organ available and retransplantation is feasible in a reasonable time frame. At times, emergency hepatectomy is performed as a desperate attempt to buy extra time to find a liver donor.
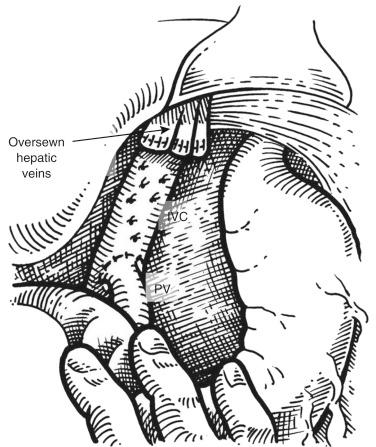
The surgical approach is similar to the first part of a liver transplant, with caval-sparing hepatectomy. Great care should be exercised to avoid bleeding, because consumption coagulopathy is extremely hard to treat in an anhepatic patient. The dissection in the hilum includes ligation of the branches of the proper hepatic artery, adjacent to the liver parenchyma. The bile duct is ligated and divided. The portal vein is dissected into the hilum, and its branches are ligated and divided. The liver is then dissected off the inferior vena cava (IVC), with ligation of the small veins from the right and the caudate lobe, up to the origin of the hepatic veins. The liver is then removed, and the stumps of the hepatic veins are oversewn. The portacaval shunt is created to decompress and drain the portal venous system: the end of the portal vein stump is anastomosed to the anterior side of the vena cava ( Fig. 65-1 ). Although every attempt to obtain hemostasis should be attempted, packing of the abdominal wound is frequently necessary. Depending on the donor liver availability, the patient can be then maintained in the operating room or, if a longer wait is contemplated, in the intensive care unit.
Primary nonfunction (PNF) is a condition characterized by immediate posttransplant liver failure with demonstrable blood flow into and out of the liver allograft. PNF occurs in up to 5.8% of liver transplants. Its outcome is fatal if a second transplant is not performed. Delayed graft function, estimated at 15% of grafts, is associated with poor initial liver function followed by recovery, unless other major complications occur. Risk factors for PNF include donor factors, such as donor age (below 1 year old or elderly), liver macrosteatosis, hemodynamic instability, warm ischemia from donation after cardiac death, cardiac arrest in the brain dead donor, poor organ flush and cooling after cross-clamp; organ preservation issues, including long cold ischemia time, inappropriate preservation solution or storage temperature; recipient issues with poor vascular inflow from portal vein thrombosis or embolization, poor arterial flow, or both, long warm ischemia from prolonged implantation, retransplantation of the liver, or cardiac arrest after allograft reperfusion. The cause of PNF cannot be determined in all cases.
PNF typically presents with unresponsiveness and coma after transplant, hypothermia, hemodynamic instability, coagulopathy, hypoglycemia, and lactic acidosis. The coagulopathy is not reversible with factor replacement. Acute renal failure secondary to liver failure further complicates the picture. Doppler ultrasonography shows patent vasculature. The arterial resistive index (defined as the peak systolic minus end-diastolic velocity divided by peak systolic velocity) is typically high initially, due to increased stiffness of the parenchyma; a subsequent decrease in the index—due to low resistance in the now wholly necrotic parenchyma, with loss of vascular tonus—is an ominous sign. If the sonogram findings are equivocal, urgent reexploration of the abdomen is indicated. The PNF liver is at times pink and slightly swollen. An early liver biopsy can underestimate the degree of liver necrosis, because the initial ischemic changes can be subtle. Therefore the decision to list the patient for a second liver transplant should not be delayed based on the biopsy result. If surgery is not contemplated, a transjugular liver biopsy can be performed, because the usual percutaneous approach is not safe due to coagulopathy.
PNF is typically not responsive to resuscitation and requires urgent retransplantation. Retransplantation is associated with good patient survival if PNF occurs after a primary transplant; results are much inferior with retransplantation after a second or third liver. Plasmapheresis, attempted in accelerated rejection, donor-recipient ABO-incompatible transplants, and fulminant hepatic failure, was used in situations labeled as PNF, resulting in liver allograft recovery. However, this approach remains controversial. Given the donor liver shortage, it is important to differentiate between PNF and delayed graft function, which is reversible.
Hepatic ischemia and necrosis are frequently associated with vascular thrombosis. The presentation is with elevated transaminase levels and liver allograft dysfunction. Although the liver has dual vascular inflow, thrombosis of either the hepatic artery or the portal vein has severe consequences for the allograft.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here