Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Despite advances in understanding the pathophysiology of acute kidney injury (AKI) and chronic kidney disease, treatment for kidney disease remains unsatisfactory. In this chapter, we briefly recount the importance of acute kidney injury and chronic kidney disease, then provide a brief description of labile iron, and, finally, summarize the role of labile iron in acute and chronic kidney disease. The availability of iron chelators provides new therapeutic tools to prevent and/or treat kidney disease.
Acute kidney injury is an independent risk factor for morbidity and mortality. A modest increase (0.3 mg/dL) in serum creatinine is associated with high in-hospital mortality, and AKI is associated with post-hospital discharge mortality and progression to end-stage kidney disease (ESKD).
Chronic kidney disease (CKD) affects approximately 10-15% of the adult population worldwide, is an independent risk factor for cardiovascular disease, and carries a high economic cost. There is a worldwide increase in the incidence and prevalence of diabetes, which is the most common cause of CKD. These observations, coupled with the increasing incidence of end-stage kidney disease despite the use of angiotensin receptor blockers (ARBs) and the multiplier effect of CKD on cardiovascular disease, indicate a major importance of CKD as a global public health problem. The failure of recent studies to halt diabetic nephropathy highlights an urgent need for new therapeutic modalities to halt progression of kidney disease.
Iron is the most abundant transitional metal in the body. The term “labile iron pool” was first used to denote a transient iron pool of low-molecular-weight, weakly chelated iron that passes through the cell. Methodological approaches for detection of this pool of iron ions are based on the use of metal chelators. Critical to iron’s importance in biological processes is its ability to cycle reversibly between its ferrous and ferric oxidation states. This precise property, which is essential for its functions, also makes it very dangerous, because free iron can catalyze the formation of free radicals that can damage the cell. Thus, from a pathophysiological standpoint, the broadest definition of a labile iron pool is that it consists of chemical forms that can participate in redox cycling, and is therefore often referred to as catalytic iron.
The catalytic iron pool is estimated to be less than 100 mg compared to the total iron in the body, which is approximately 4 g. In most cells iron homeostasis consists of iron uptake, utilization, and storage. The process of iron uptake is carried out by a transferrin receptor (TFR) and a divalent metal transporter 1 (DMT1, also called DCT1; NRAMP2), whereas ferritin is an intracellular, iron-sequestering protein. Studies are beginning to yield information on the pathways of iron transport, its export from the cell via the divalent iron ion exporter ferroportin 1, and its regulatory mechanisms including hepcidin. Since uptake and storage of iron is carried out by different proteins, the pool of accessible iron ions constitutes a crossroad of metabolic pathways of iron-containing compounds.
Studies using a variety of methods have begun to define intracellular distribution of labile iron (for reviews see Kruszewski and Esposito ). Using several techniques including laser scanning microscopy, the concentration and distribution of chelatable iron has been estimated to be about 5.0 to 15 μM in the cytoplasm and subcellular organelles including mitochondria and nuclei. In vivo , most of the iron is bound to heme or non-heme protein and does not directly catalyze the generation of hydroxyl radicals or a similar oxidant. The bleomycin-detectable iron assay measures catalytic iron and is based on the observation that the anti-tumor antibiotic bleomycin, in the presence of catalytic iron, binds to and degrades DNA with the formation of a product that reacts with thiobarbituric acid to form a chromogen. Thus the assay detects iron complexes capable of catalyzing free-radical reactions in biological samples. The binding of the bleomycin-iron complex to DNA makes the reaction site-specific and antioxidants rarely interfere. The bleomycin assay detects only “free” iron and not iron bound to specific transport proteins or to enzymes.
The ability of iron to participate in redox cycling makes it potentially hazardous by enabling it to participate in the generation of powerful oxidant species such as hydroxyl radical (metal-catalyzed Haber-Weiss reaction, below) and/or reactive iron-oxygen complexes such as ferryl or perferryl ion. In several systems, the amount of free-radical generation is related to the amount of labile iron present ( Figure 89.1 ).
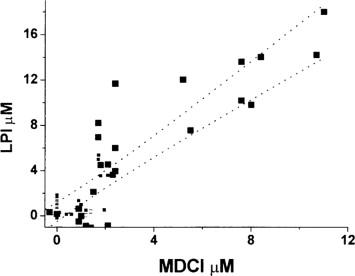
Iron also has a major role in lipid peroxidation, either directly or indirectly (through hydroxyl radicals or forming a perferryl ion) in which there is oxidative reaction of polyunsaturated lipids by removing hydrogen atoms from polyunsaturated fatty acids.
A major advancement in understanding the important role of iron in the pathophysiology of tissue injury is the recognition that iron plays a role even in the absence of systemic iron overload. It is now known that specific defects in cellular iron metabolism and/or an increase in catalytic iron may be important in several disease processes not associated with iron overload. In Friedreich’s ataxia, there is an improper processing of iron because of the deficiency of the iron-chaperone protein frataxin, resulting in accumulation of iron in the mitochondria. Deficiencies in pantothenate kinase, a key enzyme in coenzyme A synthesis, leads to iron depositions and brain damage. In addition to these specific defects in cellular iron, there is now overwhelming evidence that increased catalytic iron from subcellular or other sources participates in tissue injury in a wide variety of common disease states. This has been demonstrated in many disease states including acute and chronic kidney disease, acute myocardial infarction, and neurodegenerative disorders. In large part, the evidence consists of demonstrating an increase in catalytic iron and the ability of iron chelators to provide a protective effect, thus establishing a cause-effect relationship ( Table 89.1 ).
|
The first causative association of acute kidney injury with muscle injury (rhabdomyolysis) was described during the Battle of Britain. We now recognize that the spectrum of etiologies for rhabdomyolysis, myoglobinuria, and renal failure has been expanded with the recognition of both traumatic and nontraumatic causes. The most widely used model of myoglobinuric acute kidney injury is produced by subcutaneous or intramuscular injection of hypertonic glycerol.
There are a number of studies that indicate a role for catalytic iron in rhabdomyolysis. There is a marked and specific increase in catalytic iron content in myoglobinuric acute kidney injury ( Figure 89.2 ). In addition, studies from different investigators have shown functional and histological protection in models of myoglobinuric acute kidney injury ( Fig. 89.2 ).
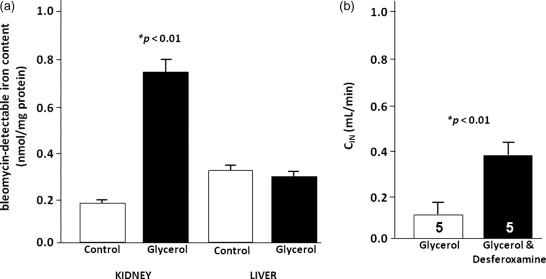
Because myoglobin is rich in heme iron, the prevailing dogma is that myoglobin from the muscle serves as an important source of iron in glycerol-induced acute kidney injury. However, several studies indicate that cytochrome P450 can also serve as a rich source of iron. Cytochrome P450 is a heme-containing enzyme that can generate reactive oxygen metabolites. Baliga et al . have shown that cytochrome P450 is a significant source of catalytic iron in glycerol-induced acute renal failure, and inhibitors of cytochrome P450 provide significant protection.
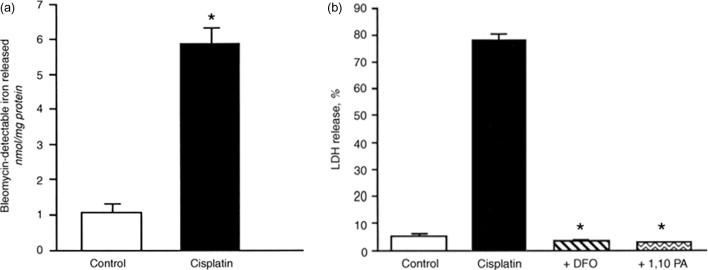
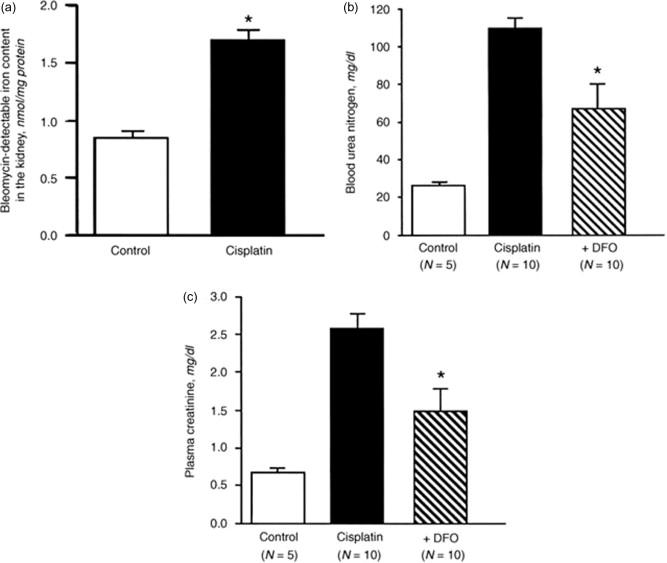
The mechanism of cisplatin nephrotoxicity is not well known. Baliga et al. have shown that the exposure of LLC-PK 1 cells to cisplatin results in a significant increase in catalytic iron released into the medium ( Figure 89.3 ). In addition, two iron chelators significantly protected against cisplatin-induced cytotoxicity ( Fig. 89.3 ). In an in vivo study, rats treated with cisplatin have a marked increase in catalytic iron ( Figure 89.4 ). An iron chelator provided marked functional (as measured by blood urea nitrogen and creatinine) ( Fig. 89.4 ) and histological protection against cisplatin-induced acute kidney injury.
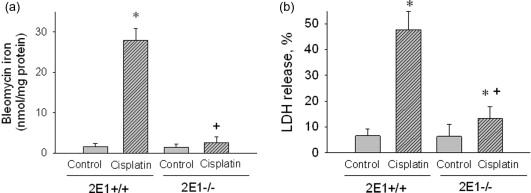
Baliga et al. have also examined potential sources of catalytic iron in cisplatin-induced nephrotoxicity. Using CYP2E1null (CYP2E1 -/- ) mice, Liu and Baliga have demonstrated a pivotal role of CYP2E1 in cisplatin-induced nephrotoxicity ( Figure 89.5 , Table 89.2 ). Incubation of CYP2E1 -/- kidney slices with cisplatin results in a significant decrease in the generation of reactive oxygen metabolites and attenuation of cytotoxicity as compared to that of wild-type mice (CYP2E1 +/+ ). CYP2E1-null mice had marked functional and histological protection against cisplatin-induced renal injury, thus demonstrating the importance of cytochrome P450 2E1 in cisplatin nephrotoxicity. Taken together, these data support a critical role for iron in mediating tissue injury via hydroxyl radical (or a similar oxidant) in this model of nephrotoxicity.
| Blood Urea Nitrogen mg/dL | Serum Creatinine mg/dL | Creatinine Clearance mL/hour | Catalytic Iron nmol/mg Protein | |
|---|---|---|---|---|
| CYP2E1+/+ | ||||
| Sham ( N =6 to 9) | 17±2 | 0.27±0.02 | 7.21±0.9 | 11.47±0.18 |
| Cisplatin ( N =8 to 14) | 69±2 a | 1.55±0.23 a | 1.02±0.2 a | 38.98±3.88 a |
| CYP2E1−/− | ||||
| Sham ( N =6) | 15±3 | 0.27±0.02 | 7.71±0.5 | 6.54±0.96 |
| Cisplatin ( N =10) | 32±4 a,b | 0.47±0.06 b | 6.24±0.8 b | 12.08±1.33 b |
a P < 0.05 compared to the respective control.
b P < 0.05 compared to CYP2e1+/+ mice treated with cisplatin.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here