Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
This chapter includes an accompanying lecture presentation that has been prepared by the authors: . ![]()
In adults, meningioma is the most common benign brain tumor and glioblastoma is the most common malignant brain tumor.
The most important prognostic factor in patients with infiltrating gliomas (astrocytoma, oligodendroglioma, and glioblastoma) is the presence or absence of an IDH1 or IDH2 mutation.
Combined IDH1 / 2, ATRX, and TP53 mutations is the molecular signature of lower grade astrocytomas.
The diagnosis of “oligodendroglioma, IDH-mutant, 1p/19q codeleted” requires testing for IDH1 / 2 mutation and 1p/19q co-deletion.
TERT promoter mutations, EGFR amplification, and combined complete gain of chromosome 7 with complete loss of chromosome 10 (+7/−10) are molecular alterations characteristic of glioblastoma.
Circumscribed gliomas (e.g., pilocytic astrocytoma, ganglioglioma, pleomorphic xanthoastrocytoma) have a low risk of recurrence after complete surgical resection, whereas infiltrating gliomas (diffuse astrocytomas, oligodendrogliomas, glioblastomas) cannot be cured by surgery.
Pediatric high-grade gliomas are molecularly and biologically different from their counterparts in adults.
The most common tumors in children are pilocytic astrocytoma (benign, grade I) and medulloblastoma (malignant, grade IV).
Medulloblastomas can be classified histologically (classic, desmoplastic/nodular, medulloblastoma with extensive nodularity, or large cell/anaplastic) and genetically (WNT-activated, SHH-activated, and non-WNT/non-SHH).
The historical category of primitive neuroectodermal tumor of the central nervous system (cPNET) has been eliminated from the current World Health Organization 2016 classification. The majority of the supratentorial small round cell tumors are currently classified, by a combination of histology and genetic alterations, as embryonal tumors or high-grade gliomas.
The current WHO Classification of Tumours of the Central Nervous System, revised 4th edition (WHO 2016), lists more than 120 types of brain tumors. Classification of brain tumors is an evolving process, with obsolete entities being discarded and newly recognized tumors added with each successive revision. In the past, classification relied heavily on recognition of morphologic patterns and immunohistochemical identification of differentiation antigens, but with the discovery a decade ago of the association between the translocation and subsequent deletion of chromosomal arms 1p and 19q , and the responsiveness of anaplastic oligodendroglioma to treatment, a new era of molecular classification of brain tumors began. Current advances in molecular methodologies, particularly in the fields of genomics, transcriptomics, and proteomics, have revolutionized brain tumor classification. In recent years, histology is increasingly being complemented by genetic characterization of neoplasms. For some entities, specific genetic alterations have become a defining feature, superseding histologic characteristics. In this chapter we present an overview of the current histopathologic and molecular classification of brain tumors based on World Health Organization (WHO) 2016 criteria.
Brain tumors that exhibit purely astrocytic differentiation can be separated into two major categories: diffuse (or infiltrating) and circumscribed. Complete surgical resection is rarely attained with diffuse astrocytoma because of the inherently infiltrative nature of the neoplastic cells, which extend well beyond the apparent gross margin of the tumor.
Diffuse astrocytoma (WHO grade II) is a mildly hypercellular infiltrative neoplasm composed of well-differentiated, spindle- to stellate-shaped astrocytes with minimal nuclear atypia and separated by a loosely fibrillary (and sometimes microcystic) background. Nuclei are characteristically irregular, angulated, and hyperchromatic. Mitotic figures are either absent or rare, and there is no vascular proliferation or necrosis. Glial fibrillary acidic protein (GFAP) may be highly or weakly expressed. Two morphologic subtypes are recognized: protoplasmic and gemistocytic. Protoplasmic astrocytomas are composed of small tumor cells with scant GFAP expression and are frequently associated with a myxoid or microcystic background. Gemistocytic astrocytomas consist of a predominant population of large rounded neoplastic astrocytes with a copious amount of glassy eosinophilic cytoplasm that is strongly GFAP positive and has an eccentrically located nucleus. Despite their benign morphologic appearance, diffuse astrocytomas have an intrinsic tendency to recur, spread extensively, and undergo anaplastic progression to a higher grade. The time to recurrence and progression after initial clinical evaluation varies from case to case but ranges from months to several years.
Anaplastic astrocytoma (WHO grade III) is a hypercellular glioma composed of poorly differentiated astrocytes consisting of a mixture of pleomorphic fibrillary and gemistocytic cells with significant nuclear atypia and prominent mitotic activity. These malignant tumors may develop as a result of anaplastic progression from a preexisting, low-grade diffuse astrocytoma or may arise de novo. The mean age of patients with anaplastic astrocytoma at initial diagnosis is approximately 41 years, which falls between the age means for patients with low-grade diffuse astrocytoma and glioblastoma multiforme (GBM).
GBM (WHO grade IV) is unfortunately both the most common glioma and the most malignant primary brain tumor arising in adults. Most commonly GBMs are solitary tumors of the cerebral hemispheres, but they may develop at almost any site within the neuraxis, including the cerebellum and the spinal cord. In many cases they infiltrate across the corpus callosum or arise directly within it, with bilateral extension (butterfly tumor). Multifocal tumors are observed in about 2% of patients and are often mistaken for metastatic disease on preoperative neuroimaging studies. The necrotic tumor mass may be partially delineated on gross examination, but infiltrating glioma cells can easily be identified microscopically well beyond the apparent gross tumor boundaries. The cellular morphology of GBM cells is highly variable, with the spectrum ranging from small, tightly packed, round or elongated cells to giant bizarre and multinucleated forms, all of which are frequently encountered within a single tumor. Mitotic figures are typically readily identified, and corresponding proliferation marker indices, such as the Ki-67 antigen, show elevated levels. Positive immunostaining for GFAP is characteristic but highly variable, and can be absent in some instances. Microvascular proliferation and foci of necrosis are histologic hallmarks of GBM ( Fig. 133.1 ). The presence of necrosis is not required for a diagnosis of GBM; vascular proliferation, in conjunction with pleomorphism and increased mitotic activity, is sufficient according to WHO 2016 criteria.
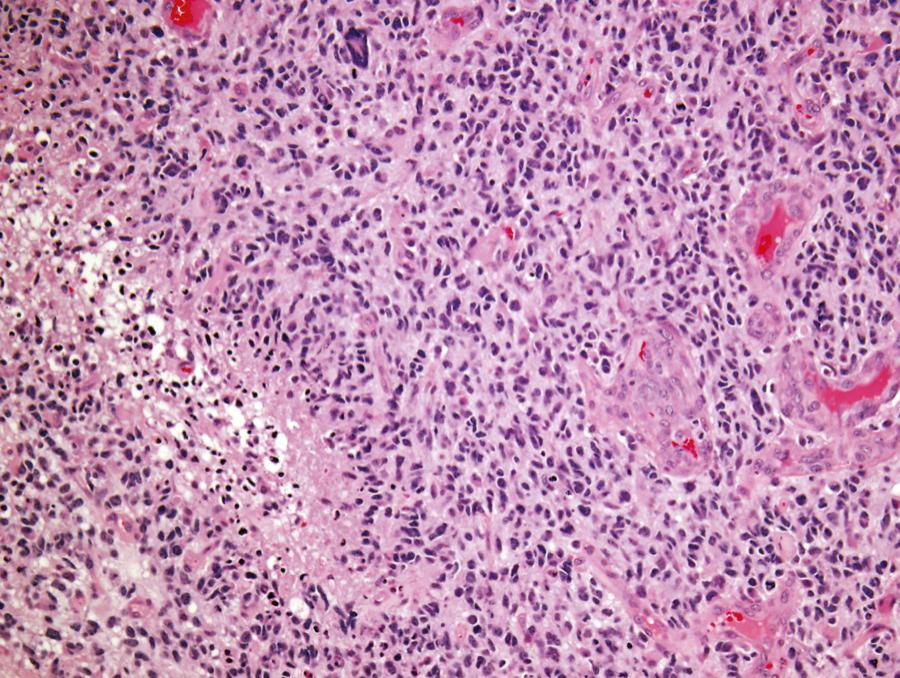
The typical peripheral, ring-like zone of contrast enhancement seen on computed tomography and magnetic resonance imaging (MRI) corresponds to the highly vascularized peripheral area of the neoplasm. Vascular proliferation is defined as the presence of blood vessels with multilayered vessel walls (more than two cell layers thick). So-called glomeruloid vascular proliferation constitutes the most obvious and characteristic example of the florid microvascular proliferation seen in GBM. Large areas of necrosis are typical of GBMs, which also frequently exhibit multiple small serpiginous zones of necrosis surrounded by densely crowded tumor cells in a pseudopalisading pattern. The hypoxic perinecrotic apoptotic cells strongly express vascular endothelial growth factor, which in turn induces the characteristically prominent vascular proliferation.
Two subsets of GBM, primary and secondary, have been described based on clinical and genetic data. Primary GBM arises de novo (i.e., without evidence of a preexisting lower grade astrocytoma), typically in patients in the sixth decade of life and beyond. In contrast, secondary GBM tends to develop in younger adults (<45 years old) by malignant progression from a diffuse astrocytoma of WHO grade II or III ( Table 133.1 ).
| Diagnosis | Common Mutations | Additional Characteristics |
|---|---|---|
| Astrocytomas | ||
| Diffuse astrocytoma, IDH-mutant (WHO grade II) |
|
Usually younger patients (40–50 years old); present as lower gliomas; most glioblastomas in this group are secondary |
| Anaplastic astrocytoma, IDH-mutant (WHO grade III) | ||
| Glioblastoma, IDH-mutant (WHO grade IV) | ||
| Diffuse astrocytoma, IDH-wildtype (WHO grade II) |
|
Usually older patients (>55 years old); present as high-grade gliomas; most glioblastomas in this group are primary |
| Anaplastic astrocytoma, IDH-wildtype (WHO grade III) | ||
| Glioblastoma, IDH-wildtype (WHO grade IV) | ||
| Diffuse midline glioma, H3 -K27M-mutant |
|
Midline location, infiltrating on histology, H3.3 p.K27M mutation |
| Oligodendrogliomas | ||
| Oligodendroglioma, IDH-mutant, 1p/19q codeleted (WHO grade II) |
|
ATRX is wild-type (ATRX mutations are mutually exclusive with 1p/19q codeletion), strong correlation with perinuclear clearing histology |
| Anaplastic oligodendroglioma, IDH-mutant, 1p/19q codeleted (WHO grade III) | ||
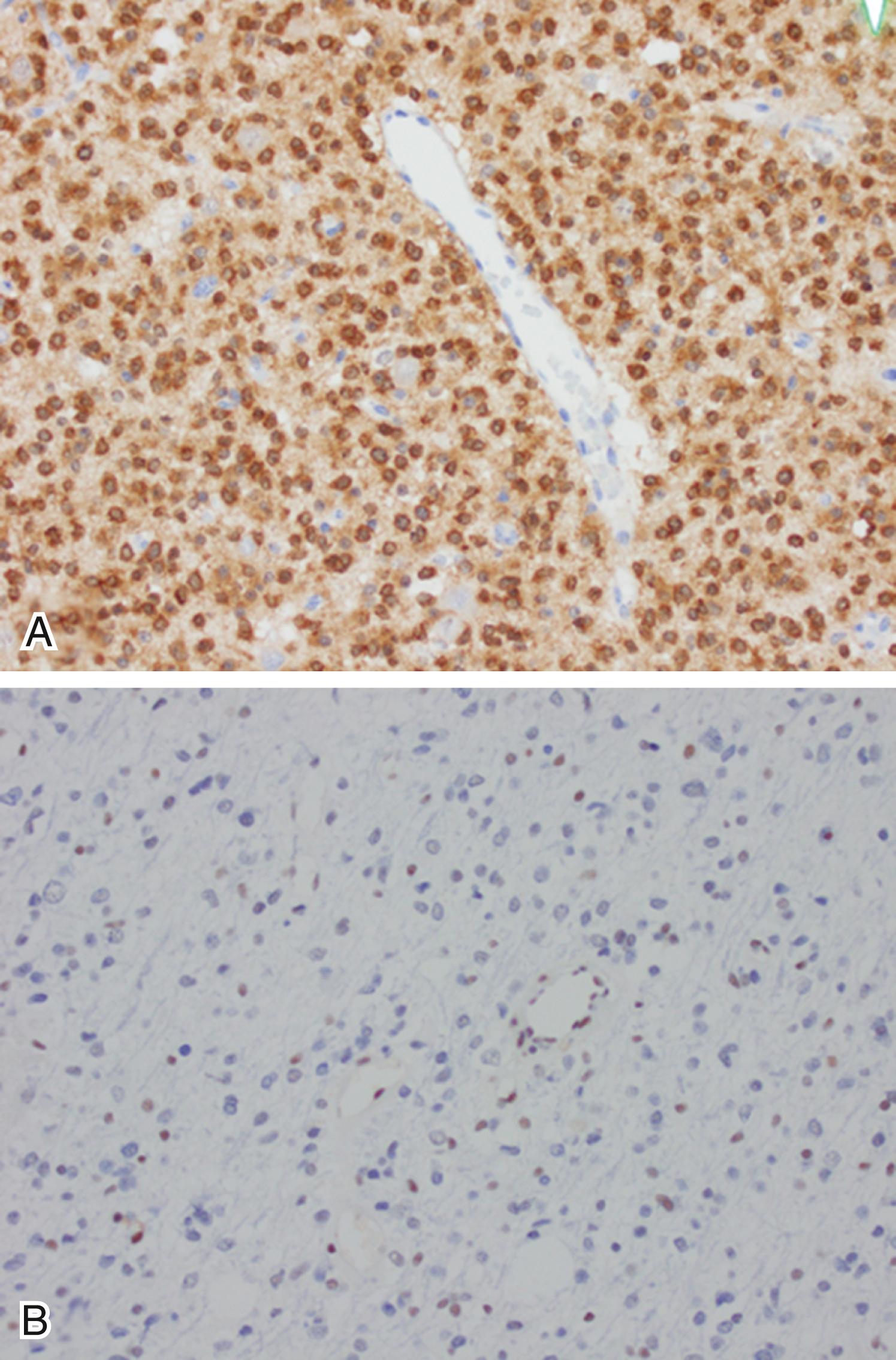
Although the histologic grading criteria to distinguish among diffuse astrocytoma WHO grade II, anaplastic astrocytoma WHO grade III, and glioblastoma WHO grade IV are still utilized in the WHO 2016 classification system, the increased understanding of the molecular alterations that drive astrocytomas challenges the prognostic value of grading on histologic grounds alone. Mutations in the isocitrate dehydrogenase 1 and 2 genes ( IDH1 and IDH2 ) have been recognized as key genetic alterations in diffuse and anaplastic astrocytomas. The presence or absence of IDH1/DIH2 mutations in an astrocytoma is a more powerful predictor of tumor behavior than grading based on histologic features alone. IDH1 or IDH2 mutations in astrocytomas frequently coexist with mutations in TP53 and ATRX . Therefore, the molecular signature of low-grade astrocytomas, which have better prognosis, is IDH1 or IDH2 mutant, TP53 mutant, and ATRX mutant ( Fig. 133.2 ). In contrast, IDH-wildtype astrocytomas have worse prognosis and frequently have mutations in the promoter of the TERT gene, EGFR amplification, and PTEN mutations. IDH-mutant astrocytomas occur in younger patients and typically present as grade II or III astrocytomas, with later progression to GBM. In contrast, IDH-wildtype astrocytomas present later in life, and frequently as grade IV tumors. Therefore, in molecular terms, IDH-wildtype GBM is the equivalent of primary GBM and IDH-mutant GBM is the equivalent of secondary GBM. Although rarely, IDH-mutant GBM will present de novo. Under the WHO 2016 classification system, diffuse astrocytoma, anaplastic astrocytoma, and GBM must be further classified as IDH-mutant or IDH-wildtype. The most common mutation in IDH-mutant astrocytomas is IDH1 p.R132H, and it is present in approximately 95% of IDH-mutant tumors. There is an antibody that recognizes the IDH1 p.R132H mutant protein (see Fig. 133.2A ) and is routinely used in clinical practice. In addition, an antibody that recognizes the ATRX protein is frequently used to aid in the diagnosis of lower grade astrocytomas. The majority of astrocytomas with an ATRX mutation will demonstrate loss of protein expression that can easily be demonstrated with immunohistochemistry (see Fig. 133.2B ). Large-scale genomic studies have shown that ATRX mutations and 1p/19q co-deletion are mutually exclusive events. Therefore the presence of an ATRX mutation or loss of ATRX expression, demonstrated by immunohistochemistry in a diffuse glioma, essentially excludes the diagnosis of oligodendroglioma.
Molecular alterations of GBM include TERT promoter mutations, combined whole chromosome 7 gain and whole chromosome 10 loss (+7/−10), and EGFR amplification, among others. Given the evidence that molecular alterations correlate better with patient prognosis than histologic grading, it is possible to make a diagnosis of “diffuse astrocytoma with molecular alterations of glioblastoma” in a patient with an infiltrating astrocytoma without the presence of vascular proliferation or necrosis, if the critical genetic alterations ( TERT promoter mutation, EGFR amplification, or +7/−10) can be demonstrated.
GBM is highly resistant to therapy, largely because of its invasive properties, which preclude surgical cure; the presence of a population of neural stem cell–like tumor cells that may harbor resistance mechanisms distinct from those of most nonneural stem cell–like tumor cells; and retention of abundant DNA repair mechanisms that abrogate the effectiveness of chemotherapy and radiotherapy. In 2005, a randomized phase 3 trial of temozolomide and radiation therapy in patients with newly diagnosed GBM determined that patients whose tumors exhibited methylation of the MGMT gene promoter survived longer after treatment than did those without evidence of promoter methylation. MGMT is a ubiquitous cellular enzyme largely responsible for resistance to the cytotoxic action of DNA alkylating agents such as temozolomide, carmustine, lomustine, and procarbazine. Inactivation of MGMT impairs the ability of the cell to repair DNA damage. To date, MGMT promoter methylation status is the single most important prognostic marker for predicting benefit from chemotherapy with alkylating agents.
Gliomatosis cerebri has historically been considered to be a unique glial tumor entity, but mounting evidence from molecular studies supports that gliomatosis cerebri is a pattern of exceptionally diffuse involvement of the central nervous system (CNS) that can be seen with any of the diffuse glioma subtypes, including oligodendroglioma.
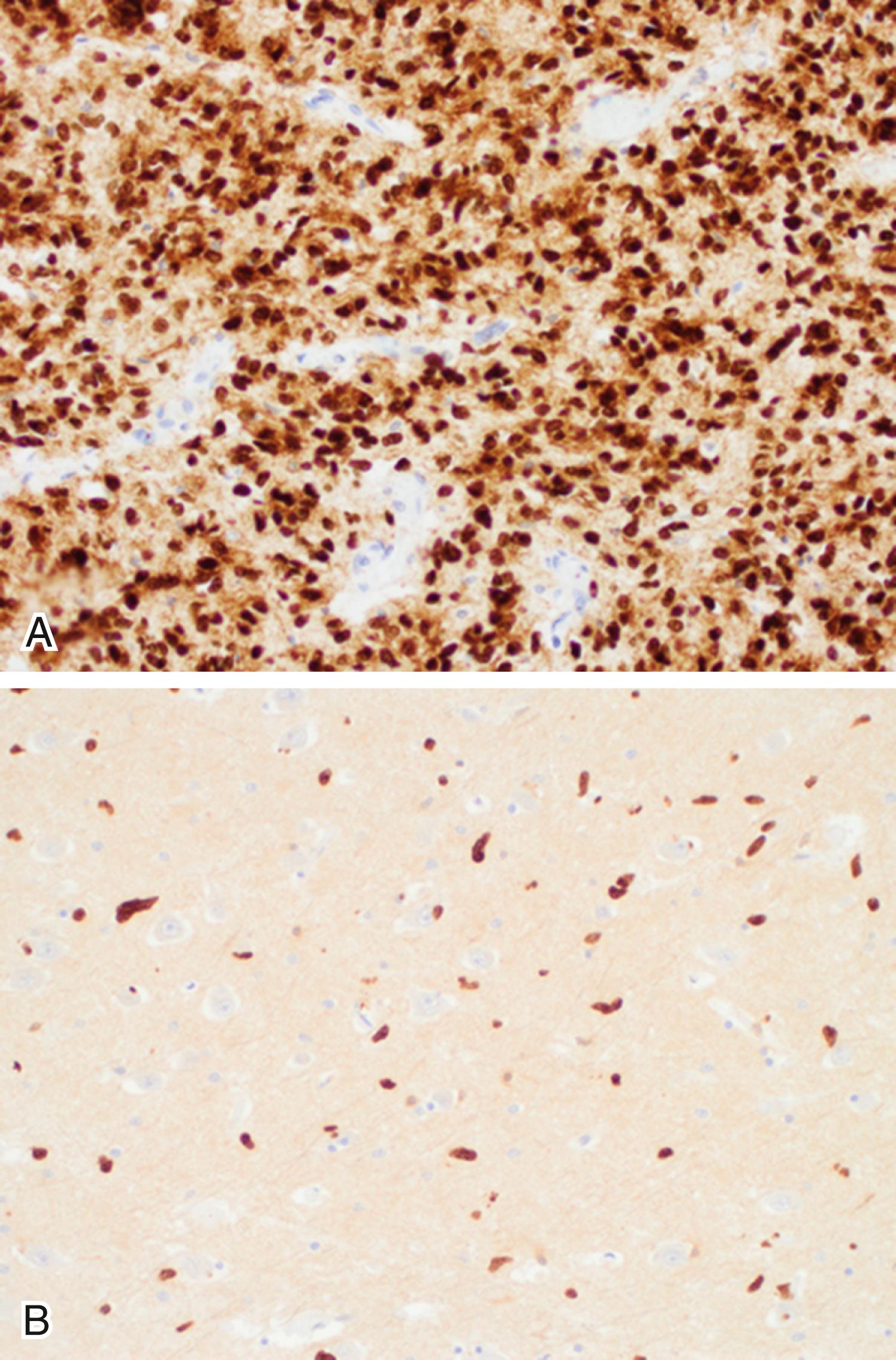
The “diffuse midline glioma, H3 K27M-mutant” (DMG-K27M) is a newly recognized entity defined in the WHO 2016 classification system as a highly malignant infiltrative midline glioma with a K27M mutation in either H3F3A or HIST1H3B/C genes. The H3 K27M mutation is found in the majority of diffuse intrinsic pontine gliomas and nearly half of diffuse gliomas arising at any other midline location of the CNS (mostly the thalamus, brainstem, and spinal cord). DMG-K27M tumors are most common in children, but they can occur at any age. These tumors may present a variety of imaging findings as well as histologic appearances at biopsy, including bland hypocellular tumors with features of low-grade gliomas. Although K27M mutations have also been described in the context of other histologies such as pilocytic astrocytoma, ganglioglioma, and ependymoma and in tumors away from the midline, their highly aggressive behavior is closely associated with DMGs. Therefore only the midline infiltrative gliomas bearing the K27M mutation qualify for the DMG-K27M diagnosis. The mutation can be readily demonstrated by immunohistochemistry with antibodies targeting the mutant protein ( Fig. 133.3 ). Another mutation in the H3 histone family, the H3F3A G34R/V mutation, has been recently described in hemispheric high-grade gliomas in children and young adults.
Pilocytic astrocytoma (WHO grade I) typically arises in children and young adults. Pilocytic astrocytomas are macroscopically circumscribed and grow very slowly. In contrast to diffuse astrocytomas, pilocytic astrocytomas exhibit very little tendency for anaplastic progression. Pilocytic astrocytomas preferentially affect the cerebellum, brainstem, optic nerves, and third ventricular region. The tumor often manifests as a mural nodule within a cyst. Invasion of the overlying leptomeninges is common, but this feature does not constitute a negative prognostic factor. Microscopically, many pilocytic astrocytomas exhibit a biphasic architectural pattern consisting of compacted areas of elongated, piloid (hair-like) cells alternating with loosely textured and microcystic areas populated by scattered stellate cells. The neoplastic pilocytes of pilocytic astrocytoma are GFAP positive. Rosenthal fibers, which are brightly eosinophilic compacted masses of glial intermediate filaments with entrapped cytosolic proteins and eosinophilic granular bodies (EGBs), are histologic hallmarks of pilocytic astrocytoma, although they are neither a constant nor a pathognomonic feature. Hyalinized and glomeruloid vessels are frequently observed. Vascular proliferation is responsible for the contrast enhancement seen on preoperative MRI studies and can occasionally cause histologic diagnostic confusion with a high-grade glioma (e.g., GBM), particularly in small biopsy samples. In pilocytic astrocytomas, mitotic activity, vascular proliferation, and necrotic foci do not have the same ominous prognostic significance as when present in diffuse astrocytomas. In favorable anatomic locations, such as the cerebellum, surgical resection of pilocytic astrocytoma has the potential to be curative.
The majority of pilocytic astrocytomas, in particular those occurring in the cerebellum, show a characteristic tandem duplication in chromosome 7 (7q34) that results in a fusion between the BRAF gene and the poorly characterized gene KIAA1549. , The fusion results in a constitutively active BRAF kinase domain. Other genetic alterations in pilocytic astrocytomas include point mutations in BRAF (e.g., BRAF p.V600E), NRAS, and KRAS. This has led to the understanding that pilocytic astrocytomas occur as a result of dysregulation of the mitogen-activated protein kinase (MAPK) pathway.
Pilomyxoid astrocytoma is a histologic variant of pilocytic astrocytoma that has been found to be clinically more aggressive than ordinary pilocytic astrocytoma. It occurs most commonly in the hypothalamic or chiasmatic region. Distinctive histologic features include a monomorphic population of neoplastic pilocytes in a prominent myxoid background stroma. Unlike the situation with most pilocytic astrocytomas, in pilomyxoid astrocytoma, Rosenthal fibers and EGBs are either very inconspicuous or completely absent, although tumors with overlapping features or maturation into classic pilocytic astrocytoma over time have been reported. Because of the more aggressive clinical course, pilomyxoid astrocytomas were previously designated as WHO grade II. However, it is now believed that the more aggressive clinical course results from difficulties in obtaining complete surgical resections in the hypothalamic/chiasmatic region, and the grading of these tumors is now uncertain, although they remain in the low-grade category (I versus II).
Pleomorphic xanthoastrocytoma (PXA; WHO grade II) is another circumscribed variant of astrocytoma that typically arises in a superficial cerebral location in children and young adults with a history of seizures. The temporal lobe is a favored anatomic site. Histologically, PXA mimics GBM in its strikingly pleomorphic tumor cells, but the paucity of mitotic figures and absence of vascular proliferation and necrosis are clues to the correct diagnosis, and the characteristic presence of EGBs, which are also seen in other circumscribed tumors (such as pilocytic astrocytoma and ganglioglioma), is an even stronger signature of PXA. The pleomorphic, giant, and often multinucleated cells may display a variable xanthomatous change in their cytoplasm because of intracellular accumulation of lipids. Most of these pleomorphic cells are GFAP positive, but neuronal marker proteins can also be detected in some tumors, indicative of dual differentiation. In addition to prominent EGBs, dense intercellular and pericellular reticulin and perivascular lymphocytic infiltrates are also typical microscopic features of PXA. As with pilocytic astrocytoma, invasion of the overlying meninges is characteristic and does not constitute a negative prognostic factor. PXAs generally exhibit indolent behavior; however, approximately 15% will recur and undergo anaplastic progression to high-grade diffuse astrocytoma. The majority of PXAs have a characteristic mutation in the BRAF gene (BRAF p.V600E). Recently, the most frequent molecular alterations in anaplastic PXAs have been described as BRAF p.V600E, CDKN2A deletion, and TERT promoter mutations. BRAF p.V600E is not specific to PXAs; it can be present in pilocytic astrocytomas, gangliogliomas, and GBMs. Specifically, the BRAF p.V600E mutation can be seen in approximately 50% of GBMs with epithelioid morphology (i.e., epithelioid GBM subtype).
Subependymal giant cell astrocytoma (WHO grade I) is an intraventricular tumor composed of large cells that resemble gemistocytes or ganglion cells, or both, morphologically and immunohistochemically. This tumor is almost invariably associated with tuberous sclerosis, although this condition is often not known at initial evaluation. Surgical resection is curative.
Desmoplastic infantile astrocytoma (WHO grade I) is a rare tumor of infancy with morphologic and clinical features that overlap those of another rare tumor termed desmoplastic infantile ganglioglioma. Both lesions share a superficial cerebral cortical location, large size, circumscribed growth pattern, and development during infancy. A marked desmoplastic meningeal reaction is characteristic. Surgical resection is the treatment of choice, and the prognosis is generally more favorable than would otherwise be suggested by the typically very large size of most of these tumors at diagnosis.
Oligodendroglioma (WHO grade II) is a well-differentiated, diffusely infiltrating tumor composed of cells resembling normal oligodendroglia. Most oligodendrogliomas arise in adults in the fourth and fifth decades. The preferential location of these tumors is the white matter of the cerebral hemispheres, from which tumor cells typically infiltrate the overlying cortex. As viewed macroscopically and on neuroimaging studies, oligodendrogliomas often appear somewhat more circumscribed than astrocytomas. They are composed of uniform round cells with cleared cytoplasm surrounding a central spherical nucleus (fried egg appearance). The perinuclear halo is a diagnostically useful fixation artifact present only in formalin-fixed, paraffin-embedded tumor tissue ( Fig. 133.4 ). Mitotic activity is inconspicuous. A branching network of small delicate blood vessels (chicken wire pattern) is a classic histologic feature of many oligodendrogliomas. Microcalcifications are also common; this feature can be appreciated in some cases as gyriform calcifications on imaging studies. Subpial tumor infiltration, perineuronal satellitosis, and perivascular satellitosis of tumor cells (secondary structures of Scherer) are characteristically seen in oligodendrogliomas that infiltrate gray matter. Oligodendrogliomas generally recur locally and ultimately undergo anaplastic progression.
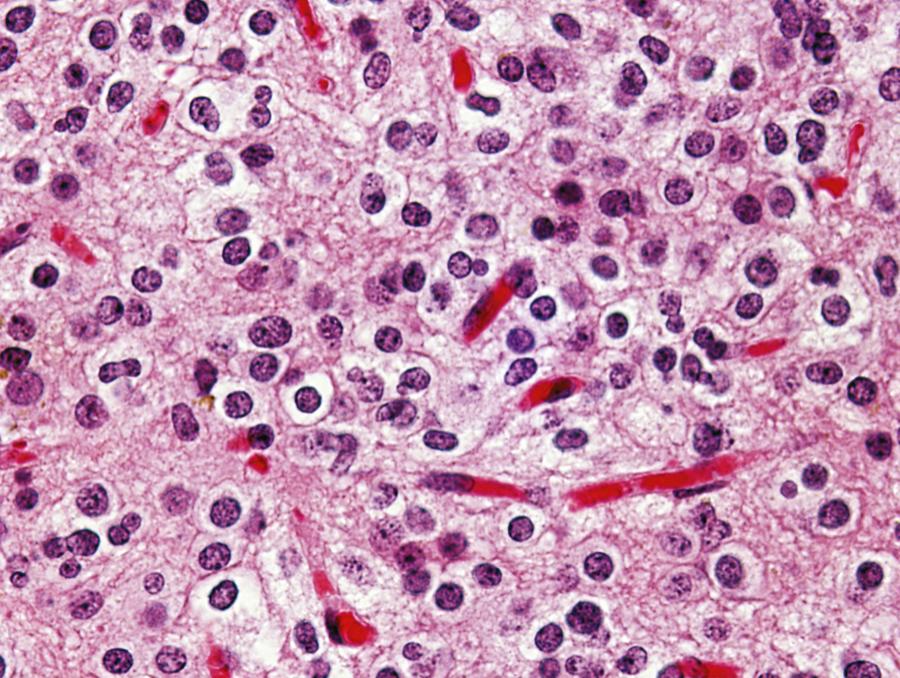
Anaplastic oligodendroglioma (WHO grade III) is an oligodendroglioma in which high-grade features, such as increased mitotic activity, microvascular proliferation, or necrosis (or any combination of such features), are present (see Table 133.1 ). Some tumors show florid vascular proliferation and necrosis that mimic what is seen in GBM. Varying numbers of cells with eosinophilic GFAP-positive cytoplasm that resemble miniature gemistocytes (mini-gemistocytes) or astrocytes (gliofibrillary oligodendrocytes) may be present.
The hallmark genetic signature of oligodendroglioma (low grade and anaplastic) is IDH1 or IDH2 mutations and combined whole-arm deletion of chromosomes 1p and 19q, which arises secondary to an initial translocation event and constitutes an independent prognostic marker, with 1p and 19q loss being associated with improved outcome regardless of the specific therapeutic regimen. Mutations in the promoter of the TERT gene are also frequent, although not a defining feature. The WHO 2016 classification system incorporates the defining genetic alterations in the name of oligodendrogliomas. As a result, the diagnostic entities are now “oligodendroglioma, IDH-mutant, 1p/19q codeleted, WHO grade II,” “anaplastic oligodendroglioma, IDH-mutant, 1p/19q codeleted, WHO grade III,” and “oligodendroglioma, NOS, WHO grade II or III.” The not otherwise specified (NOS) designation is used to indicate that the defining molecular alterations of oligodendrogliomas have not been evaluated and the diagnosis is based on histologic grounds alone. Although there is a strong correlation between oligodendroglial histology and the presence of IDH1/IDH2 mutations and 1p/19q co-deletion, the overlap between histologic features of astrocytomas and oligodendrogliomas makes the “oligodendroglioma NOS” diagnosis less certain.
Although both astrocytomas and oligodendrogliomas have mutations in IDH1 or IDH2, ATRX mutations are characteristic of astrocytomas and 1p/19q co-deletion is characteristic of oligodendrogliomas. Also, ATRX mutations and 1p/19q co-deletion are mutually exclusive events (with extremely rare exceptions). In the setting of an infiltrating glioma, loss of ATRX expression, which can be evaluated by immunohistochemistry using an antibody against the ATRX protein, essentially rules out a diagnosis of oligodendroglioma. A caveat of 1p/19q analysis by fluorescence in situ hybridization (FISH) is that a small number of GMBs will give an apparent positive result due to partial deletions in chromosomes 1p and 19q. These partial deletions are distinct from the translocation that results in complete loss of the 1p and 19q chromosomes, which is characteristic of oligodendrogliomas, but are indistinguishable on the FISH assay. Therefore, an infiltrating glioma with 1p/19q co-deletion by FISH analysis, but without IDH1 or IDH2 mutations, is most likely an astrocytoma (very likely a GBM).
The oligoastrocytoma (mixed glioma), as a distinct entity, was eliminated in the WHO 2016 classification system since nearly all morphologically defined mixed gliomas can be categorized as oligodendrogliomas or astrocytomas based on the previously mentioned molecular features. It is important to recognize that pediatric gliomas with an oligodendroglial histologic phenotype (pediatric oligodendrogliomas) very rarely harbor an IDH1/IDH2 mutation or 1p/19q co-deletion, and their defining molecular alterations remain unknown.
Ependymoma (WHO grade II) is a slowly growing neoplasm of children and young adults that originates from the ependymal lining of the cerebral ventricles ( Box 133.1 ). A subtentorial location is the most frequent in children, whereas in adults most of these tumors are supratentorial. Ependymomas may occur outside the ventricular system in the brain parenchyma and also in the spinal cord. Ependymomas are grossly characterized by a sharply demarcated edge. As seen histologically, classic ependymomas are moderately cellular tumors composed of oval cells with monomorphic nuclei and tapering eosinophilic cytoplasm. Some ependymomas have a more glial appearance, whereas others are more epithelioid. Some ependymoma variants (cellular, tanycytic) mimic other primary tumors, although others (papillary, clear cell) may mimic secondary tumors. The histologic hallmarks of ependymoma are the perivascular pseudorosettes (perivascular collars of radiating tumor cell cytoplasmic processes) and, more elegantly but less frequent, the true ependymal rosette (tumor cells surrounding a central lumen) ( Fig. 133.5A ). GFAP and epithelial membrane antigen (EMA) immunopositivity is a frequent finding in ependymoma. GFAP reactivity is often strongest in the perivascular pseudorosettes, and EMA positivity takes the form of cytoplasmic dot-like and ring-like staining that can be very focal. Electron microscopy may be used in some cases to identify the ultrastructural features associated with ependymal cell differentiation (intercellular lumina filled with microvilli and sometimes cilia and prominent intercellular junctional complexes), although this approach is rarely used in clinical practice anymore.
Choroid plexus papilloma
Atypical choroid plexus papilloma
Choroid plexus carcinoma
Choroid plexus meningioma
Central neurocytoma
Ependymoma
Subependymoma
Subependymal giant cell astrocytoma
Chordoid glioma of the third ventricle
Metastasis
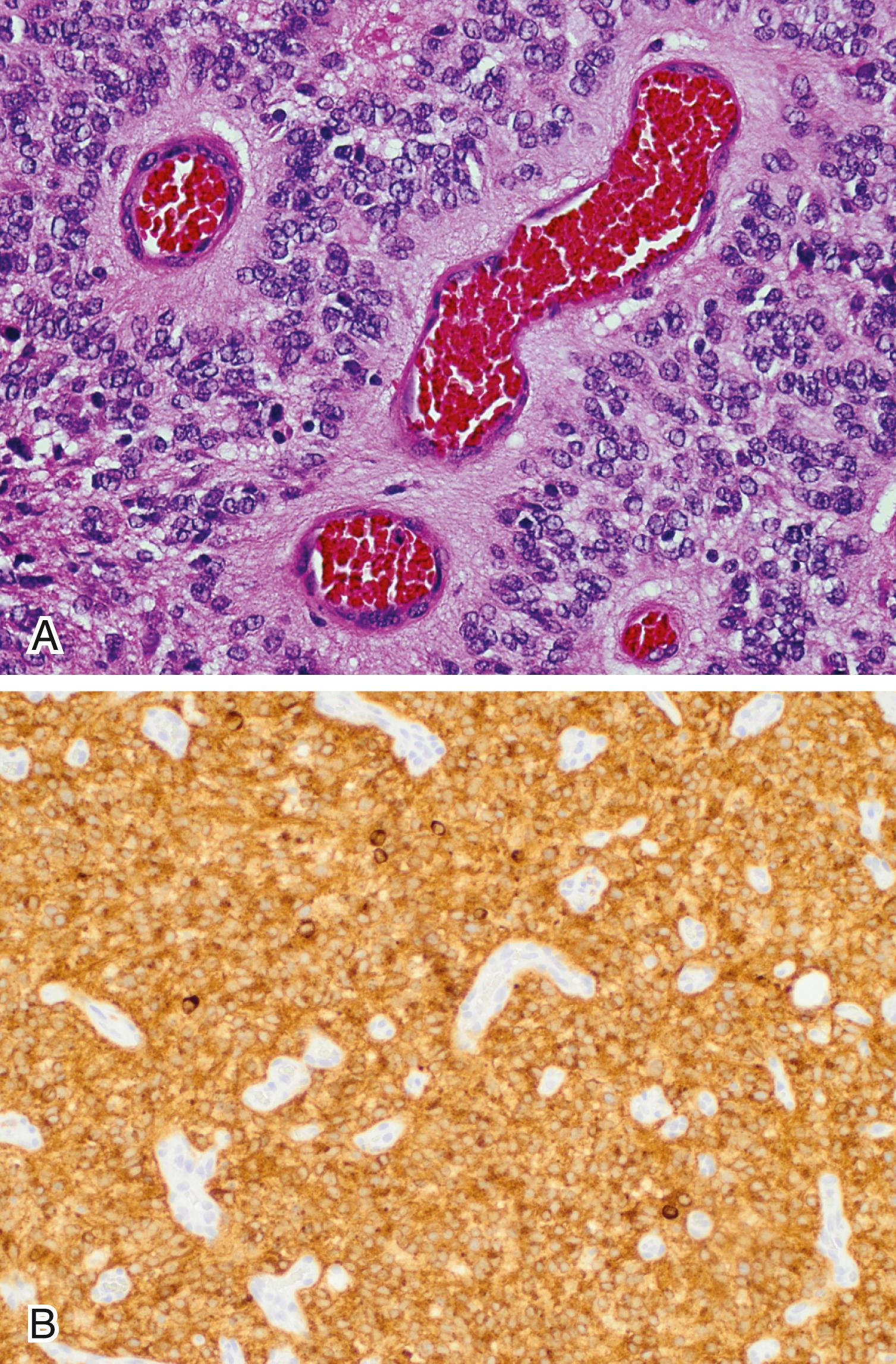
Anaplastic ependymoma (WHO grade III) typically shows increased mitotic activity and microvascular proliferation. However, the prognostic value of anaplasia in ependymomas is controversial.
A genetic alteration that drives a majority of supratentorial ependymomas (∼70% of supratentorial pediatric ependymomas) is the C11orf95 – RELA fusion. The translocation involves a poorly characterized gene ( C11orf95 ) and RELA, a critical component of the nuclear factor-κB complex. The “ependymoma, RELA fusion-positive” variant affects mostly children and has been incorporated as a new entity by the WHO 2016 classification system. Clear cell morphology and branching capillaries are often present. Immunohistochemistry utilizing both RELA (a.k.a. p65) and L1CAM antibodies, or FISH testing, can help identify this variant in routine diagnosis ( Fig. 133.5B ). Although not yet recognized by the WHO 2016 classification system, other molecularly and biologically distinct subgroups of ependymomas have been described; for example, the posterior fossa ependymoma (PF-EPN) subgroups A and B. The PF-EPN subgroup A, which is the most aggressive, can be identified by loss or reduction of the histone 3 lysine 27 trimethylation (H3K27me3) by immunohistochemistry.
Myxopapillary ependymoma (WHO grade I) is a distinct low-grade variant of ependymoma that arises almost exclusively from the caudal portion of the spinal cord of adults in the conus medullaris or filum terminale region ( Box 133.2 ). The tumor is usually well circumscribed and covered by an outer layer of investing leptomeninges (capsule). Layers of cuboidal or spindled tumor cells surround myxoid microcysts. Collars of myxoid material also surround blood vessels. Surgical resection of intact tumors can be curative in some cases; however, the presence of micrometastases, which are not visible to the unaided eye, or frank capsular breaching by the tumor can lead to diffuse dissemination among the nerve roots of the cauda equina and ultimately result in significant morbidity.
Ependymoma
Myxopapillary ependymoma
Paraganglioma
Subependymoma (WHO grade I) is an indolent intraventricular glioma of adults (rare cases arise in the spinal cord). Asymptomatic lesions are often discovered only as incidental findings at autopsy, but subependymomas occasionally produce ventricular obstruction of the lateral or fourth ventricles. Surgical resection is curative. As seen microscopically, the tumor is composed of clusters of small, uniform, benign-appearing tumor cell nuclei separated by extensive cell-free areas of finely fibrillary matrix. Lateral ventricular examples are prone to prominent microcystic change that can obscure the characteristic multinodular architecture. Cases of mixed subependymoma and classic ependymoma are rare but well documented; the clinical course of such tumors is similar to that of WHO grade II ependymoma.
Choroid plexus papilloma (WHO grade I), atypical choroid plexus papilloma (WHO grade II), and choroid plexus carcinoma (WHO grade III) originate from the epithelium of the choroid plexus of the cerebral ventricles (see Box 133.1 ). The lateral ventricles are more often involved in patients younger than 20 years. As viewed both macroscopically and microscopically, choroid plexus papilloma closely recapitulates the papillary architecture of normal choroid plexus, but the tumor cells tend to be more crowded and columnar, as opposed to the cuboidal morphology of normal choroid plexus epithelium. Most intraventricular choroid plexus papillomas are benign and can be cured with surgery, but dissemination via cerebrospinal fluid (CSF) can occur in up to 20% of cases.
In contrast, choroid plexus carcinomas are aggressive tumors that arise more frequently in childhood and show histologically frank features of malignancy, including effacement of the papillary pattern by solid areas of tumor, increased cellularity, nuclear pleomorphism, brisk mitotic activity, necrosis, frequent invasion of the adjacent brain parenchyma, and CSF dissemination. Choroid plexus carcinomas express cytokeratins, but the staining characteristics of S-100 protein, transthyretin, and GFAP, which are usually positive in choroid plexus papilloma, are inconstant and thus less helpful in diagnosing this tumor. Some cases of choroid plexus carcinoma require ultrastructural identification of the characteristic features of choroid plexus differentiation (microvilli, cilia, and intercellular junctional complexes).
Atypical choroid plexus papilloma is a recently codified entity that differs from choroid plexus papilloma in exhibiting increased mitotic activity. Curative surgery is still possible, but the probability of recurrence appears to be significantly higher.
The choroid plexus may also be involved by a variety of other neoplastic and nonneoplastic mass lesions, most prominently intraventricular meningioma, metastatic carcinoma (especially renal cell carcinoma), and xanthogranuloma (which is a reactive mass lesion characterized by cholesterol clefts and attendant multinucleated giant cell reaction).
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here