Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
This chapter includes an accompanying lecture presentation that has been prepared by the authors: ![]() .
.
This chapter includes an accompanying lecture presentation that has been prepared by the authors: ![]() .
.
Anti–vascular endothelial growth factor (VEGF) therapy (bevacizumab [Avastin]) has been approved by the US Food and Drug Administration (FDA) for the treatment of recurrent glioblastoma and is useful to pharmacologically “debulk” the mass effect—vasogenic edema of large, deep, multicentric or inoperable tumors—yet converts the angionecrotic phenotype to a diffuse, infiltrative, “gliomatosis” tumor that does not yield to surgery or conventional chemotherapy.
An intense effort is underway to identify genetic, epigenetic, molecular, serum, or radiographic markers to identify which patients could best respond to antiangiogenic therapy. These efforts have led to the development of the Response Assessment in Neuro-Oncology (RANO) and more recently the immunotherapy (iRANO) and the modified (mRANO) criteria.
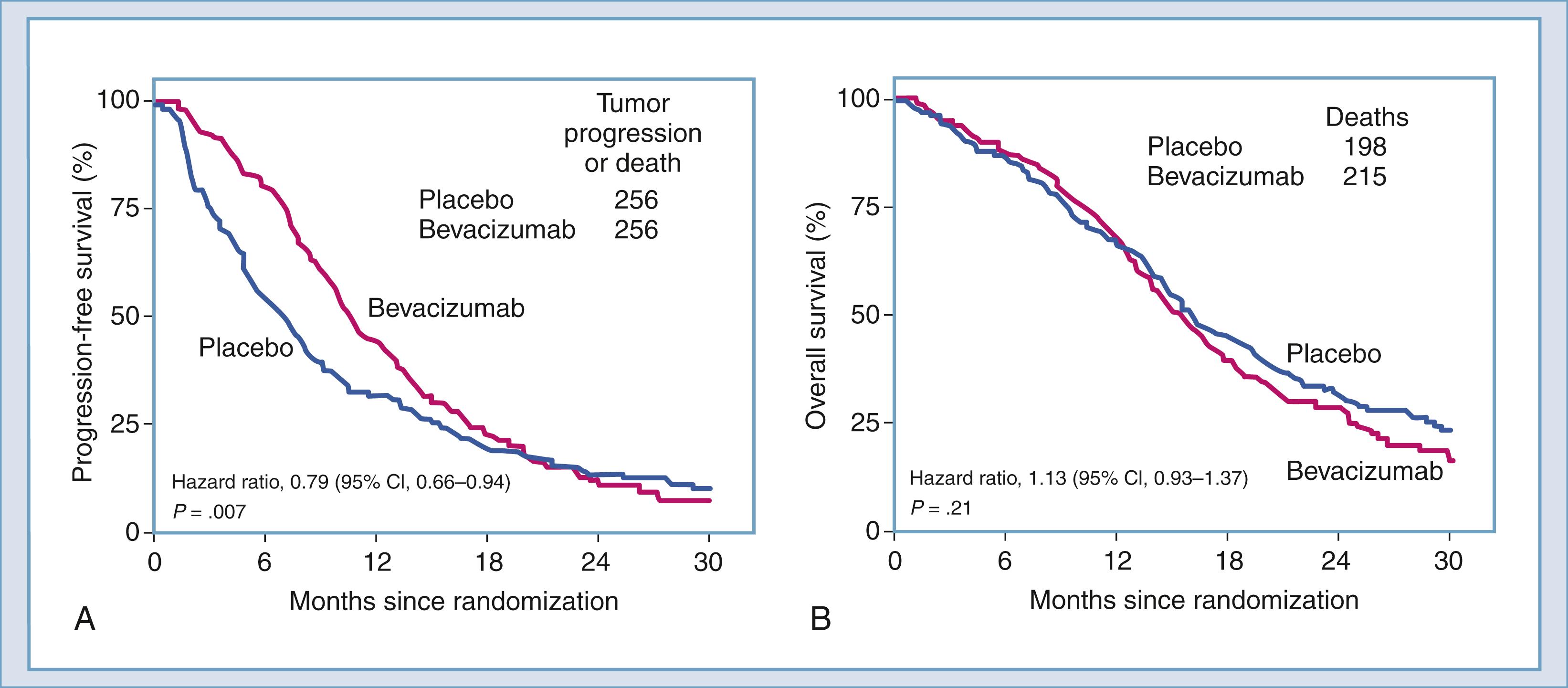
Two large, independent, prospective, randomized clinical trials (AVAglio and RTOG 0825) failed to show an increase in overall survival (OS) when adding bevacizumab to the temozolomide and radiation therapy for newly diagnosed glioblastoma. Furthermore, a more recent, randomized controlled trial failed to show a survival advantage when chemotherapy (lomustine) was added to bevacizumab for recurrent glioblastoma.
Bevacizumab has a clinical role to increase progression-free survival (PFS), reduce edema, and improve symptoms, and is particularly useful in the management of radiation-induced necrosis for brain metastases.
Because VEGF is essential for wound healing, bevacizumab should be withheld 4 weeks before planned surgery or 4 weeks after surgery, unless surgery is performed on an emergency basis.
The molecular and cellular mechanisms for antiangiogenic resistance and escape include vascular co-option, vascular mimicry, endothelial and stem cell transformation, the perivascular niche, multiple proangiogenic factors, mutational heterogeneity, and alternative signaling pathways (e.g., c-MET).
Bevacizumab can play an important role in radiation-refractory, inoperable acoustic neuromas (e.g., neurofibromatosis type 2 [NF2]), but has been unsuccessful in the treatment of pituitary tumors, meningiomas, brain metastases, medulloblastomas, ependymomas, and pediatric brain tumors, except for anecdotal reports.
Antiangiogenic therapy shows promise in the development of novel immunotherapeutic approaches, as the microvasculature is a key component of the tumor microenvironment.
Clinically, angiogenesis remains a key step in malignant progression from low-grade to anaplastic astrocytoma to glioblastoma, and efforts to block angiogenesis could potentially lead to a sustained state of glioma dormancy.
Antiangiogenesis therapy remains a major treatment option in neuro-oncology during the current era of molecular medicine and represents a fertile area for development of precision medicine and translational therapy. Treatment with angiogenesis inhibitors is now a cornerstone of modern anticancer therapy, and currently bevacizumab (Avastin), the prototype of an angiosuppressive drug, is approved for (1) metastatic colorectal cancer; (2) first-line treatment of nonsquamous, non–small cell lung cancer (NSCLC); (3) stage III or IV ovarian cancer after primary surgery; (4) cancer of the cervix; (5) metastatic colorectal cancer; (6) metastatic renal cell cancer; and (7) recurrent glioblastoma. For newly diagnosed glioblastoma, two multicenter, randomized, phase 3 clinical trials (AVAglio [A Study of Bevacizumab (Avastin®) in Combination With Temozolomide and Radiotherapy in Participants With Newly Diagnosed Glioblastoma] and Radiation Therapy Oncology Group [RTOG] 0825) showed a significant increase in progression-free survival (PFS), but disappointingly, not overall survival (OS) , , ( Table 140.1 ). Although survival remains the “gold standard” by which efficacy is measured, clinical benefit such as neurocognitive preservation or quality of life can also be used as a criterion for US Food and Drug Administration (FDA) approval. The first generation of clinically useful angiogenic inhibitors focused on vascular endothelial growth factor (VEGF) and the targets in the VEGF pathway; the next generation of angiogenesis-directed therapeutics will target other angiogenic pathways and cellular subpopulations. The current chapter updates the scientific basis of angiosuppressive therapy; results of recent randomized trials; therapeutic applications for glioblastoma and other CNS tumors; treatment of radiation-induced necrosis; potential toxicity; mechanisms of escape and resistance and conversion to an invasive phenotype; neuroimaging and biomarkers to monitor therapeutic response; and future directions, including the use of antiangiogenesis combined with other therapeutic strategies such as immunotherapy.
| Author | Trial | Patients (n) | Median PFS (mo) | 6M-PFS (%) | Median OS (mo) | Comments |
|---|---|---|---|---|---|---|
| Friedman et al., 2009 | BRAIN (rGB) BEV |
85 | 4.2 | 42.6 | 9.2 | Landmark paper showing that bevacizumab ± irinotecan is well tolerated and active in recurrent glioblastoma |
| BEV + IR | 82 | 5.6 | 50.3 | 8.7 | ||
| Kreisl et al., 2009 | NCI (rGB) | 48 | 4.0 | 29 | 7.8 | Phase II prospective study showing significant biologic and antiglioma activity of single-agent bevacizumab in recurrent glioblastoma Early MRI response (first 96 hours and 4 weeks) predictive of long-term PFS |
| Wick et al., 2017 | BELOB (rGB) BEV |
50 | 3 | 18 | 8 | Phase 2 randomized prospective trial that shows promising efficacy of the combination of lomustine with bevacizumab for recurrent glioblastoma |
| Lomustine | 46 | 2 | 11 | 8 | ||
| BEV + lomustine | 44 | 11 | 41 | 11 | ||
| Chinot et al., 2014 | AVAGlio (nGB) | Improved PFS and maintenance of baseline QOL and performance status were observed with BEV; however the rate of adverse events was higher in the BEV group than in the placebo control group | ||||
| BEV + TMZ/RT | 458 | 10.6 a | NR | 16.9 | ||
| TMZ/RT | 463 | 6.2 | NR | 16.8 | ||
| Gilbert et al., 2014 | RTOG 0825 (nGB) BEV + TMZ/RT |
312 | 10.7 b | NR | 15.7 | Although there was a similar increase in PFS, over time an increased symptom burden, a worse QOL, and a decline in neurocognitive function were more frequent in the bevacizumab group |
| TMZ/RT | 309 | 7.3 | NR | 16.1 |
More than 100 years ago, Paul Ehrlich introduced the concept of targeted therapy in the form of a “magic bullet”—a drug that would go straight to its intended target. He coined the term chemotherapy, and described: (1) specific cellular receptors, (2) the blood-brain barrier, (3) neurotoxicity of drugs, (4) rationally “designed” drugs, and (5) animal models as part of a systematic approach to drug discovery—thereby creating the foundations of experimental therapeutics in oncology.
A century later, the pioneering work of Judah Folkman ( Fig. 140.1 ) developed the field of angiogenesis research that revolutionized ophthalmology, oncology, and angiogenesis-dependent diseases. To date, hundreds of thousands of patients have benefited from inhibitors of the angiogenic protein VEGF. In 2004 the FDA commissioner announced that in addition to the traditional three modes of cancer therapy—surgery, radiotherapy, and chemotherapy—“antiangiogenic therapy can now be considered the fourth modality of cancer treatment.” , We use the terms antiangiogenic therapy, angiosuppression, and angiotherapy , , interchangeably.

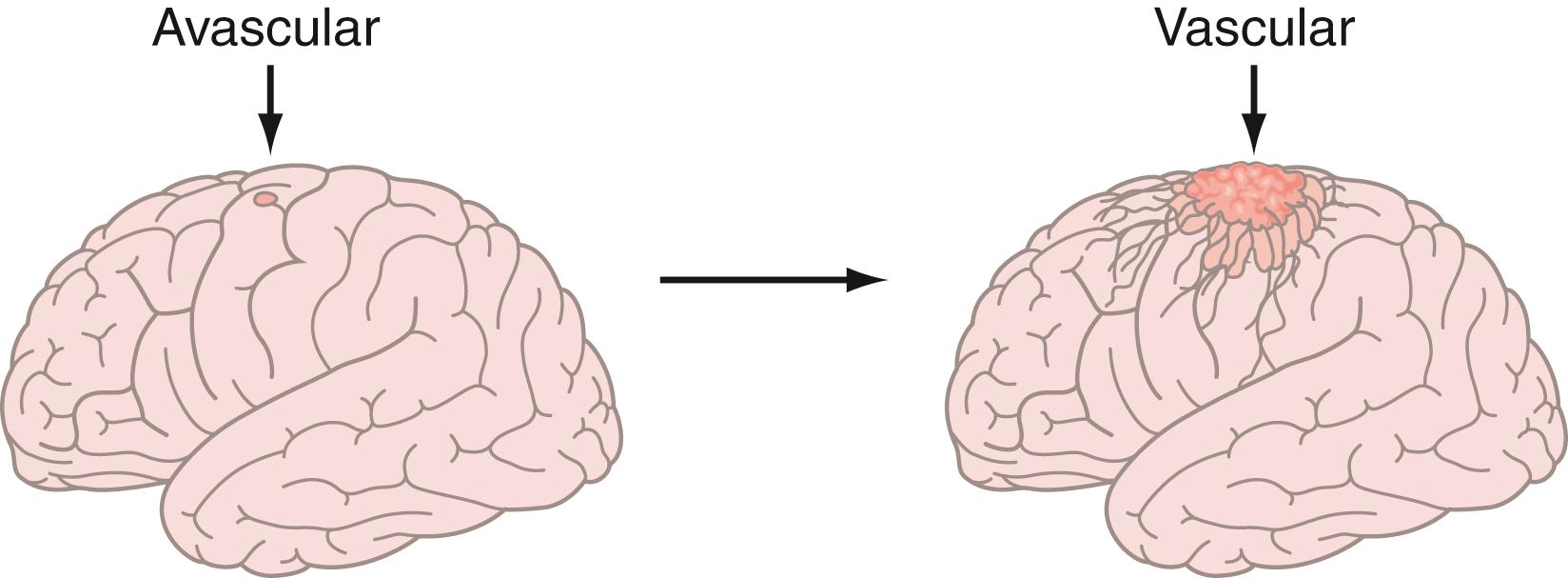
In Folkman’s seminal paper, he hypothesized that (1) solid tumor growth is dependent on angiogenesis; (2) tumors secrete a diffusible angiogenic factor; and (3) blockade of the factor would render tumors harmless, existing in a persistent state of dormancy. , These concepts were supported by observation of tumors, including brain tumors, transplanted into the avascular vitreous body. , , Once tumors acquired a blood supply, there was a 19,000-fold burst in tumor volume, supporting the concept of an angiogenic switch ( Fig. 140.2 ). The first quantitative, translational study of angiogenesis , compared microvascular density in a range of human neoplasms and predicted that glioblastomas would be uniquely susceptible to angiosuppression because of the extremely high degree of neovascularization.
Proof-of-principle that angiogenesis could be controlled by naturally occurring, endogenous inhibitors was demonstrated by Judah Folkman and Henry Brem within cartilage, , and shortly thereafter by Steven Brem within the vitreous humor of the eye. , These seminal discoveries provided the impetus for the discovery of additional endogenous and pharmacologic stimulators and inhibitors of angiogenesis. The use of anti-VEGF molecules revolutionized ophthalmology, with FDA approval of pegaptanib and ranibizumab (a fragment of bevacizumab) for the treatment of age-related macular degeneration and diabetic macular edema.
The pivotal discovery of a protein termed vascular permeability factor by Harold Dvorak , and vascular endothelial growth factor by Napoleone Ferrara led to the development of a monoclonal antibody that decreases angiogenesis, capillary permeability, and growth of tumors, including glioblastoma. Inhibition of VEGF signaling by a variety of knockout techniques halts the growth of experimental gliomas. , In patients with gliomas, VEGF is highly overexpressed in the tumor tissue and the cerebrospinal fluid (CSF), making VEGF an attractive therapeutic target. Phase 1 trials showed that bevacizumab was well tolerated and could be added to standard chemotherapy without exacerbating chemotherapy-induced toxicity. In a phase 2 trial in patients with metastatic colorectal cancer, bevacizumab added to chemotherapy (irinotecan, 5-fluorouracil, and leucovorin [IFL]) resulted in a significant improvement in OS, response rate, and time to disease progression. These findings were validated in a randomized prospective trial that provided class I evidence of increased survival with use of an antiangiogenic drug in human cancer.
Bevacizumab was confirmed to be an effective agent, extending PFS, when used in combination with cytotoxic chemotherapy in patients with cancer of the breast or lung. The success of bevacizumab preceded FDA approval of other antiangiogenic compounds, notably sunitinib (Sutent); sorafenib (Nexavar), and lenalidomide (Revlimid), for a spectrum of cancer types. , ,
Bevacizumab, a humanized monoclonal antibody directed against VEGF (VEGF-A), received FDA approval in 2004 for metastatic colorectal carcinoma. , In view of compelling evidence of efficacy, measured by tumor responsiveness, specifically from the BRAIN trial (A Study to Evaluate Bevacizumab Alone or in Combination With Irinotecan for Treatment of Glioblastoma Multiforme) , and the National Cancer Institute (NCI) trial, , bevacizumab was endorsed by the National Comprehensive Cancer Network (NCCN) brain tumor panel for recurrent glioblastoma. Accelerated FDA approval soon followed on May 5, 2009, based on durable objective responses (independent radiologic review and stable or decreasing corticosteroid use). In the light of these groundbreaking observations, initial enthusiasm for a new approach to finally control the growth of human glioblastoma was soon tempered by the realization that malignant glial cells use several mechanisms to resist and escape anti-VEGF therapy. Two highly anticipated, randomized, multi-institutional trials, AVAglio and RTOG 0825, 13 evaluated bevacizumab for newly diagnosed glioblastoma. These studies each showed that bevacizumab led to an increase in PFS, but not OS ( Fig. 140.3 ).
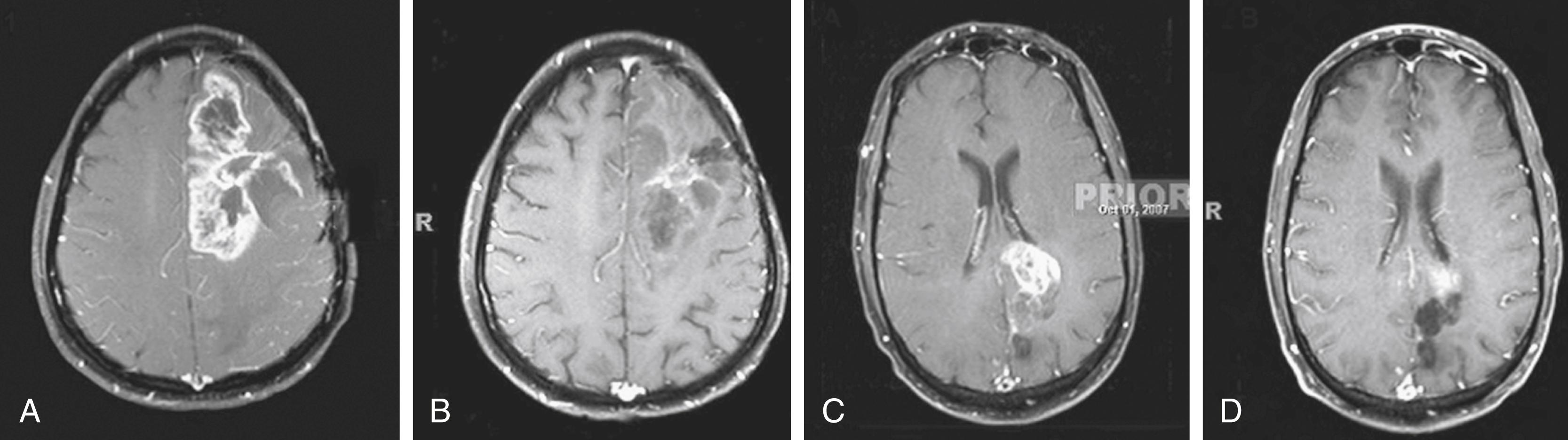
The efficacy of the bevacizumab to rapidly and dramatically decrease tumor volume is readily and reproducibly apparent in terms of tumor response ( Fig. 140.4 ). As predicted by the Folkman hypothesis of tumor dependency on angiogenesis and validated in tumor models, there is a rapid, reproducible clinical response frequently observed at the initiation of bevacizumab, a “pharmacologic debulking” (see Fig 140.4 ). We performed a meta-analysis of 548 patients with malignant gliomas from 15 published reports and found radiographic response rates of 55%, with 6% complete responses and 49% partial responses—striking results when compared with the rate in historical controls of 9% response to cytotoxic chemotherapy for recurrent malignant gliomas. No difference was found in bevacizumab dose-response benefit between 5 mg/kg and 10 to 15 mg/kg. Historically, Wong et al. previously reported a 6-month progression-free survival rate (6M-PFS) of 21% for chemotherapy alone without angiosuppression using any of several chemotherapeutic regimens. Lomustine, for example, as a single agent (one of the choices for recurrent glioblastoma) shows only a 10% response, with a 6M-PFS of only 20%. By contrast, the average 6M-PFS of bevacizumab-treated patients was 45%. PFS is an important end point to evaluate therapy for recurrent high-grade gliomas. Based on FDA approval and NCCN endorsement, despite the absence of well-controlled trials, bevacizumab is widely used in the treatment of recurrent glioblastoma. Because of the increase in PFS, bevacizumab is currently approved for first-line therapy of newly diagnosed glioblastoma in Japan, but not in the United States, Canada, or Europe. In a long-term postmarketing surveillance study conducted in Japan, there were no new safety concerns in this patient population, with acceptable rates of bleeding, infection, and adverse drug events. There is an urgent need to identify which patients are most likely to benefit from bevacizumab therapy. , There is an ongoing phase 2 study of bevacizumab beyond progression for glioblastoma treated with key therapeutics (BIOMARK study), in which bevacizumab is continued even after disease progression, and a comprehensive genetic analysis is planned.
Responses to bevacizumab, or failures, are noticeable early, usually after four doses (two cycles) and at the first follow-up MRI examination. Responses occur even in patients heavily pretreated with chemotherapy, and irrespective of age, gender, location of the tumor, or extent of surgical resection. , Performance status is correlated with survival in certain reports but not in others. For a select population, in those patients who have early clinical deterioration and/or have partially resected tumor, adding bevacizumab up-front can contribute to superior outcome as measured by survival.
Angiotherapy is effective for patients with anaplastic glioma. , In a series of 54 recurrent grade III glioma patients, linking biomarkers to prognosis, only the p53 status (worse OS with positive p53 immunohistochemical staining) and the performance status were linked to survival; smaller tumor sizes and frontal lobe location were significantly more common in patients responding to bevacizumab. Paradoxically, other molecular biomarkers, such as MGMT promotor methylation, epidermal growth factor receptor (EGFR), Ki-67, 1p/19q codeletion, IDH1 mutation, and loss of ATRX expression failed to predict response to bevacizumab or affect outcome measured in survival. The radiographic response to bevacizumab can be more pronounced for patients with glioblastoma than anaplastic glioma, possibly owing to the higher VEGF levels in glioblastomas.
Although the radiographic responses are impressive with anti-VEGF therapy, it soon became clear that the traditional use of contrast enhancement to define the target lesion as a mass, the Macdonald criteria, is insufficient, because the use of bevacizumab can also induce an infiltrative nonenhancing growth pattern (a pattern resembling “gliomatosis cerebri”) at disease progression. , The initial observations of response to angiosuppressive therapy led to revised criteria to evaluate the response to recurrent glioma. The revised Response Assessment in Neuro-Oncology (RANO) criteria give more emphasis to changes on T2-weighted or fluid-attenuated inversion recovery (FLAIR) images on MRI Use of the RANO criteria ( Table 140.2 ) reveals that the classical outcomes of 6M-PFS and the proportion of patients with an objective radiographic response are not ideal end points for trials using bevacizumab in recurrent glioblastoma. , ,
| Criterion | CR | PR | SD | PD |
|---|---|---|---|---|
| T1 gadolinium-enhancing disease | None | ≥50% ↓ | <50% ↓ but <25% ↑ | >25% ↑ b |
| T2/FLAIR | Stable or ↓ | Stable or ↓ | Stable or ↓ | ↑ b |
| New lesion | None | None | None | Present b |
| Corticosteroids | None | Stable or ↓ | Stable or ↓ | NA c |
| Clinical status | Stable or ↑ | Stable or ↑ | Stable or ↑ | ↓ b |
| Requirement for response | All | All | All | Any b |
a RANO, Response Assessment in Neuro‐Oncology, was developed, in part, to evaluate glioblastoma response in the era of antiangiogenic therapy. RANO accounts for the signal changes evident on T2/FLAIR series of the MRI.
b Progression occurs when this criterion is met.
c Increase in corticosteroids alone will not be taken into account in determining progression in the absence of persistent clinical deterioration.
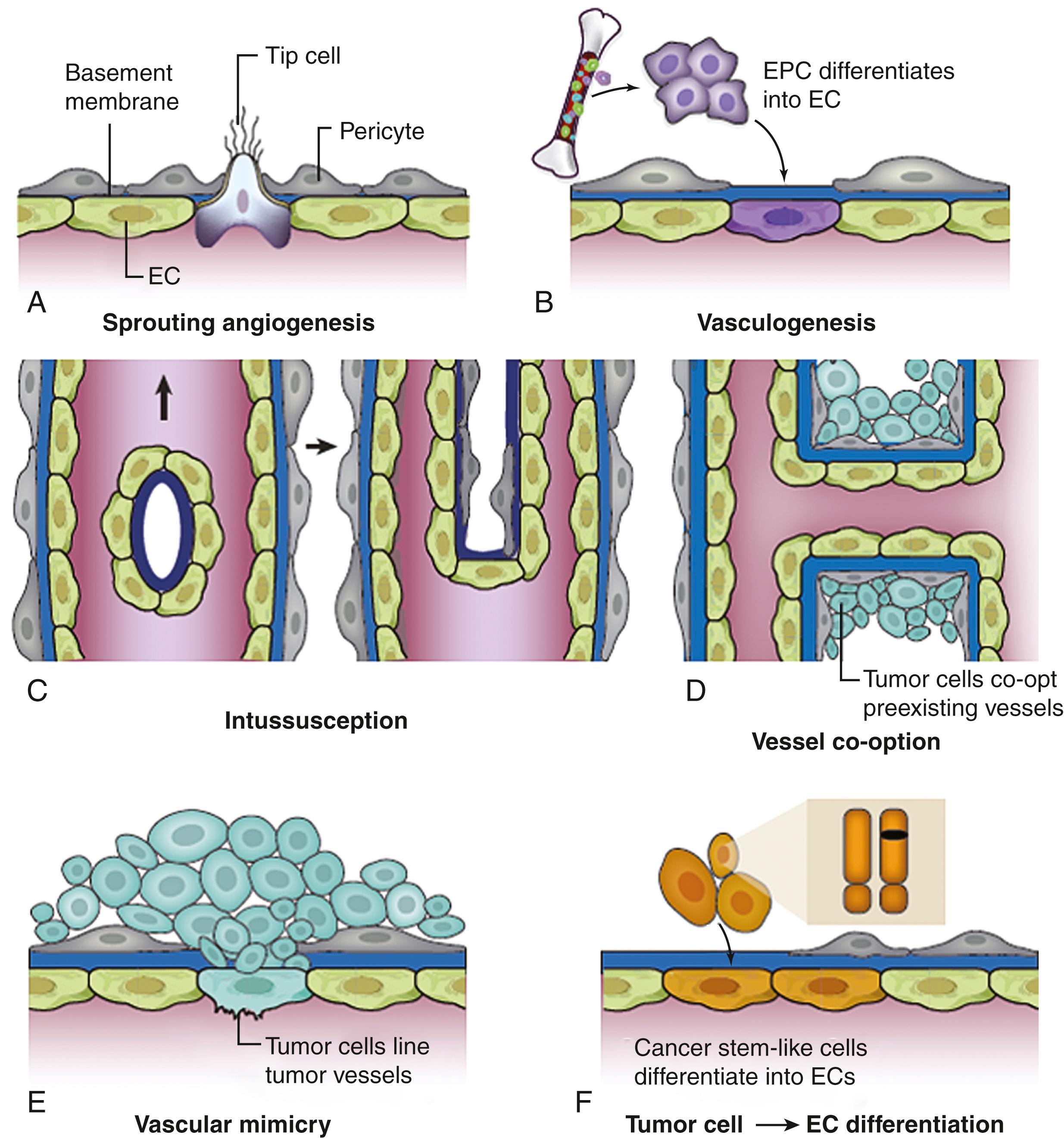
Our understanding of the fundamental biology of tumor angiogenesis has grown sharply in the past decade, , , , , as summarized in Fig. 140.5 .
Angiogenesis is recognized as a fundamental hallmark of cancer. During the development of malignancy from preneoplasia, including the transition from low-grade to high-grade gliomas, the “angiogenic switch” is expressed , , (see Figs. 140.2 and 140.6 )—a key event in multistage carcinogenesis with therapeutic implications. Angiogenesis is essential in the stepwise progression of low-grade gliomas to anaplastic gliomas and glioblastoma. The objective of therapeutic angiosuppression is to suppress the logarithmic growth associated with the onset of the angiogenic switch, and ultimately to convert large vascularized tumors such as glioblastoma into tiny, innocuous nodules, analogous to in situ cancer (i.e., “cancer without disease”).
Angiotherapy, by reducing tumor size (a pharmacologic “debulking”), could theoretically enhance other modes of therapy, including chemotherapy, radiation therapy, and immunotherapy.
Angiogenesis not only affects the growth potential of the malignant cell by affecting the microenvironment and permitting logarithmic growth (i.e., affecting the “soil”), but it also affects the “seed” of cancer. The transformed cancer cell undergoes oncogene activation, dependence, or “addiction.” For example, tumorigenicity of myc or ras oncogenes is angiogenesis dependent. Mutations relevant to glioma formation—including the PTEN, EGFR, or c- myc genes—can act as an “angiogenic switch” by stabilizing HIF-1α or one of its downstream targets (e.g., VEGF).
Human glioblastomas contain clones that vary in their angiogenic capacity. The angiogenic phenotype in tumors is malleable and can spontaneously revert to the nonangiogenic phenotype in a subpopulation of human tumor cells. Those that display the ”angiogenic switch” grow rapidly, produce vascularized tumors in immunodeficient mice, and are rapidly fatal; in contrast, those glioblastoma cells that are nonangiogenic are slow-growing and harmless to the host.
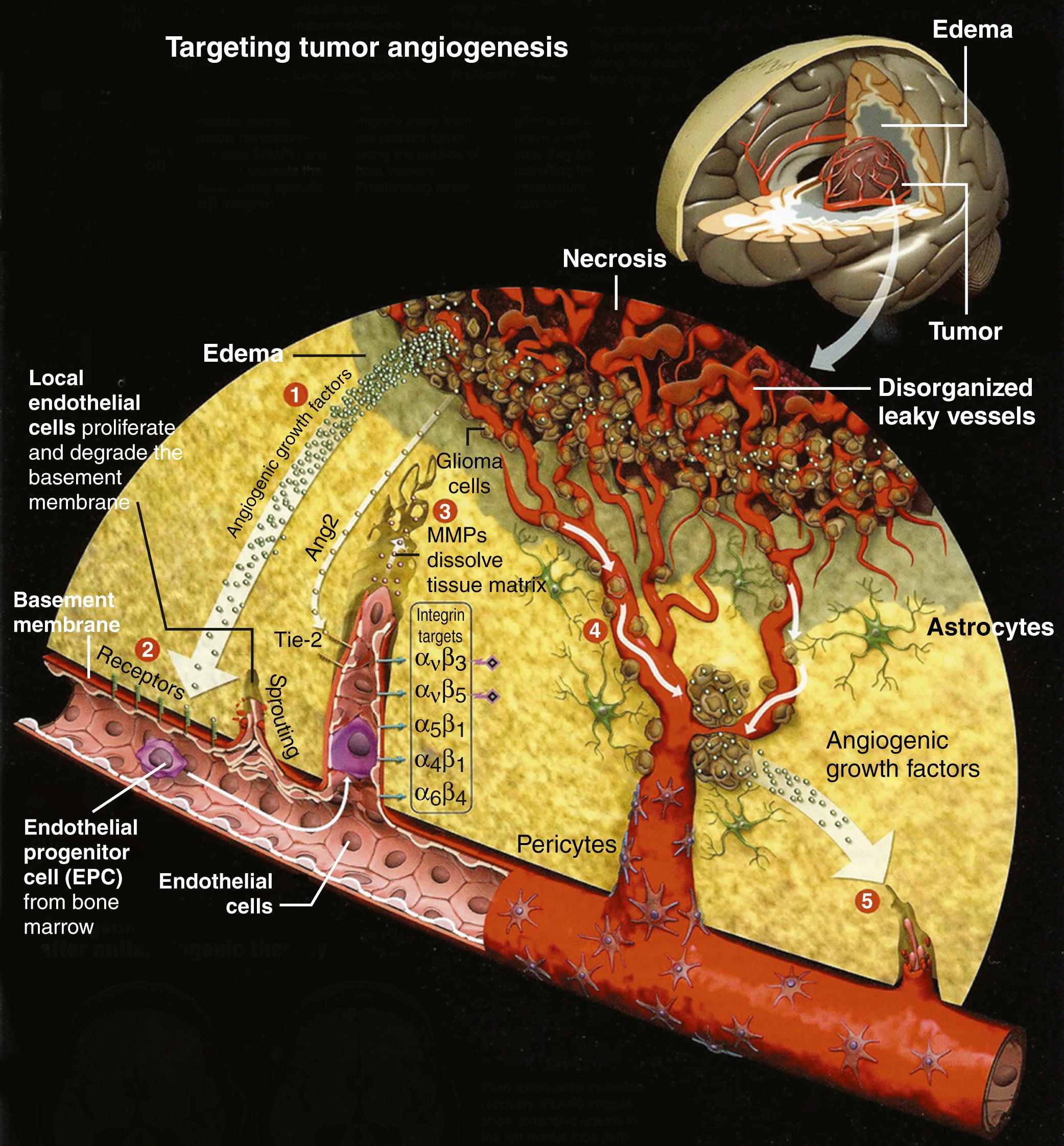
Key steps of the angiogenic switch (see Fig. 140.6 ) , in human glioblastoma are as follows. (1) Glioma cells, in response to gene mutations and/or hypoxia, release growth factors, such as VEGF (or bFGF), that activate brain endothelial cells during the switch to the angiogenic phenotype. (2) Growth factors (e.g., VEGF) bind to endothelial cell receptors and activate signal transduction pathways, leading to endothelial cell proliferation. (3) Sprouting vessels secrete urokinase and metalloproteases, and migrate toward the tumor using specific αβ integrins. (4) Glioma cells surround the host vessels by “co-option” and migrate along these vessels away from the main mass of the tumor to form a protected environment, or a “vascular niche.” (5) The glioma cluster reaches a critical size, approximately 5 × 10 5 cells, then recruit their own, additional vessels, perpetuating the process.
An important stimulus for the angiogenic switch in glioblastoma is hypoxia. , Within hours of exposure to hypoxia, C6 glioma cells upregulate the expression of VEGF messenger RNA and other proangiogenic peptides. The expression of VEGF is tightly regulated by hypoxia-inducible transcription factor (HIF) complex, including HIF-1α and HIF-2α. A subset of tumors initiate growth by co-opting existing host vessels, which then regress, leading to an avascular tumor that depends on angiogenesis for further growth. The ability of malignant cells to grow along preexisting cerebral microvessels (i.e., vascular co-option) also provides an important mechanism of resistance to antiangiogenic therapy and antiangiogenesis therapy (see Figs. 140.5D and 140.7C ).
The endothelial cells that form the vasculature of gliomas can arise from adjacent mature blood vessels or circulating endothelial precursor cells derived from the bone marrow. , , , Because the endothelial cells arise from nonmalignant cells, it was once thought that tumor endothelium carries a stable genome and therefore could surmount multidrug resistance. , Analysis of the endothelium derived from gliomas, however, indicates that these cells are genetically distinct from the normal quiescent endothelium, with mutations that indeed resemble those characteristic of gliomas. , Dynamic time-lapse analysis of endothelial-glioma cell interactions shows not only that the microvasculature provides a physical scaffold on which the glioma cells migrate, but also that glioma cellular mitosis occurs preferentially at microvascular branch points, suggesting that both proliferation and migration of glioma cells are guided by the microvasculature. The anatomic bifurcations are not only branch points, but also mitotic control points.
Numerous proangiogenic factors are implicated in glioma biology. , , ,
The key factors include VEGF, basic fibroblast growth factor (bFGF), CXCL12 (SDF-1α), hepatocyte growth factor–scatter factor (HGF-SF), placental-derived growth factor, and platelet-derived growth factor. bFGF is overexpressed in glioblastoma and has an autocrine, proliferative effect because glioblastoma cells have an elevated expression of FGF receptor 1. The binding of VEGF or bFGF to their cognate receptors activates the existing endothelial cells.
Other stimulators of angiogenesis include angiopoietin 1, angiotropin, angiogenin, epidermal growth factor, endothelin-1, profilin-1, transforming growth factor β1 (TGF-β1), granulocyte colony-stimulating factor, interleukin (IL)-1, IL-6, and IL-8. , , We found IL-6, an angiogenic factor and a component of a cellular senescence-associated gene signature, to be linked to age, glioma progression, and survival. Because of the availability of anti-IL-6R antibodies (e.g., tocilizumab), FDA approved for rheumatoid arthritis and to treat the cytokine storm in chimeric antigen receptor [CAR] T-cell therapy, IL-6 is an attractive target as a novel therapeutic approach using repurposed agents. In preclinical studies at our institution, we found that IL-6 targeting increased survival in a genetically engineered mouse model controlling macrophage alternative activation and polarization to an immunostimulatory state through HIF-2α. In preliminary experience in the context of a CAR T-cell trial, we found IL-6 targeting, using siltuximab, prescribed to prevent cytokine storm, to be well tolerated in patients with recurrent glioblastoma.
Angiogenesis is physiologically suppressed by one or more of the known endogenous inhibitors, including angiopoietin 2, angiostatin, brain-specific angiogenesis inhibitor 1 (BAI1), endorepellin, endostatin, interferon-α (IFN-α), kringle 5, pigment epithelium–derived factor, platelet factor 4, prolactin (16-kD fragment), tissue inhibitors of metalloproteinase (TIMP-1, TIMP-2, and TIMP-3), troponin I, tumstatin, and thrombospondin 1. , The ideal inhibitor of angiogenesis would be a naturally occurring molecule, present in the microvasculature of the brain, that is nontoxic and blocks both the angiogenic and infiltrative phenotype. We found a specific fragment of the parent Aβ peptides to suppress angiogenesis, and with use of this approach, glioma growth and vascularization were inhibited in orthotopic animal models.
The detailed physiologic studies of Jain and colleagues of tumor-microvascular interactions, including measurements of oxygenation, blood flow, and interstitial pressure of brain tumors, led to the normalization hypothesis. , , , This model provides the basis for restoring a physiologic balance of angiogenic stimulators and inhibitors. The optimal “normalization window” varies with each specific drug, dose intensity, duration of treatment, and duration of response. The normalization concept helps interpret findings in preclinical models of angiogenic inhibitors used for drug discovery. , , For example, in Fig. 140.7B , the tumor vascular bed been exposed to an angiogenic inhibitor (e.g., bevacizumab or penicillamine), thereby “normalizing” the vessels (compare with Fig. 140.7A ), restoring blood flow, enhancing oxygenation, improving delivery of chemotherapeutic drugs, and increasing radiosensitivity. , Whereas normalization of the vascular supply to the glioma should theoretically improve the efficacy of radiation therapy and chemotherapy, complex interactions with multimodality therapy can occur, such as the upregulation of other proangiogenic pathways (e.g., angiopoietin) when VEGF-A is targeted (by bevacizumab), or unexpected effects of the combination of radiation therapy with antiangiogenic therapy. Specifically, glioma cells have been found to form a microtubular system that interconnects as a method of resisting radiation therapy , and possibly antiangiogenic therapy. These distinct, ultra-long, highly functional membrane protrusions of glioma cells could explain, in part, resistance of gliomas to multiple forms of therapy (radiation, chemotherapy, antiangiogenesis) and form a novel target to attack treatment-resistant gliomas. , The complex interactions of vascular normalization with chemotherapy and antiangiogenic therapy can be studied in preclinical models to assess the side effects of glioma microtubular formation and glioma cell invasiveness to optimize treatment combinations, dosing, and scheduling.
Another target for bevacizumab and angiotherapy is the perivascular niche where glioma progenitor cells resembling stem cells (stem cell–like glioma cells [SCLGCs]) reside (see Fig. 140.8 ). , The stem cell hypothesis proposes that cancers arise from a small fraction of cancer cells that constitute a reservoir of self-sustaining cells that maintain the tumor. Expanding the number of blood vessels results in an increase in the fraction of SCLGCs; by contrast, depletion of blood vessels from gliomas ablates self-renewing cells from tumors and arrests tumor growth. When compared with other glioma cells, the SCLGCs consistently secrete elevated levels of VEGF, further amplified by hypoxia; bevacizumab suppresses growth of xenografts derived from SCLGCs but shows limited efficacy against xenografts derived from a matched non-SCLGC population. Targeting glioma stem cell–deprived pericytes can disrupt the blood-tumor barrier (BTB) and enhance chemotherapy, demonstrated by using ibrutinib, a drug that enhances effusion and activity of drugs with poor BTB penetration. Endothelial cells can accelerate the growth of glioblastoma stem-like cells by paracrine secretion of IL-8 in a tumor perivascular niche.
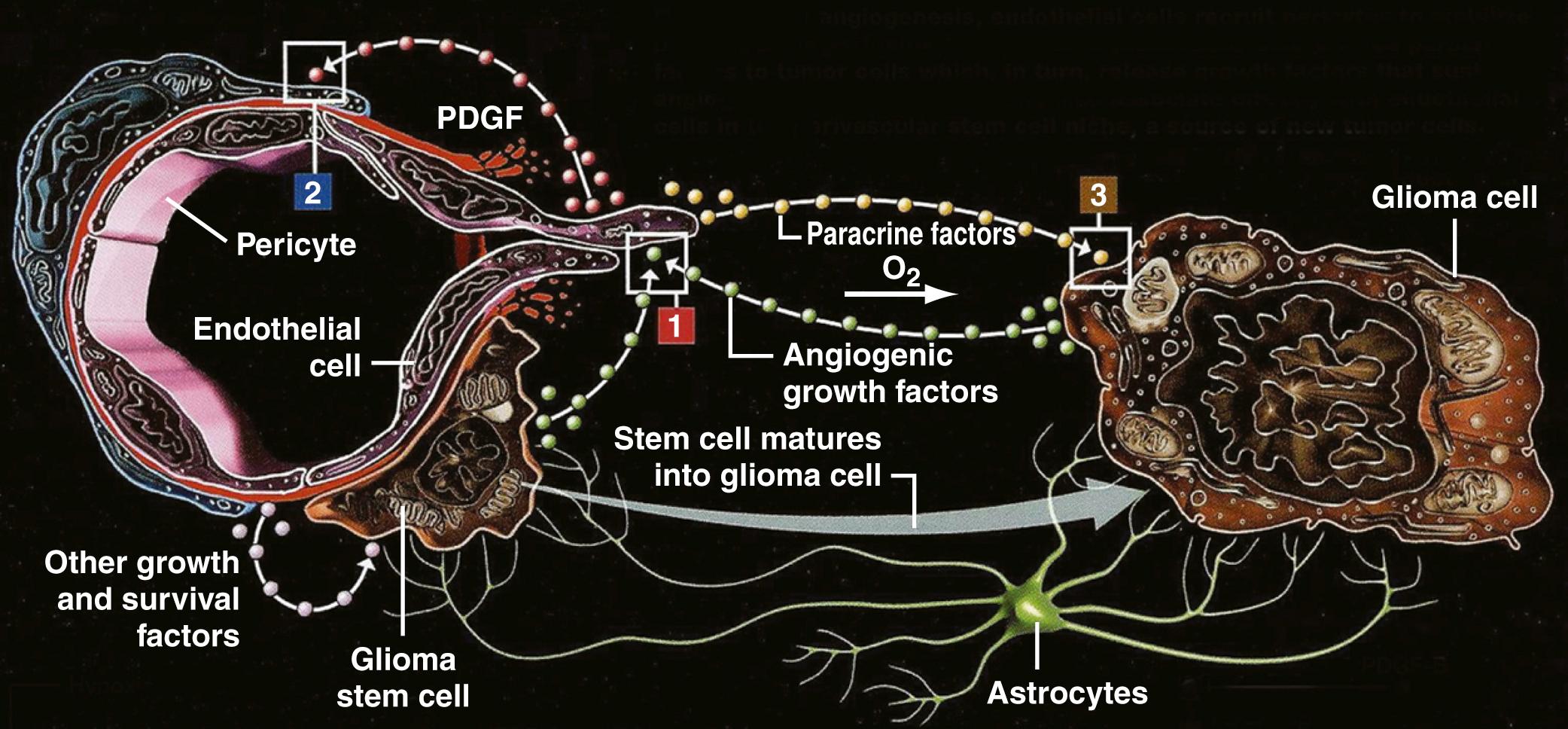
The disruption of such a vascular niche could result in the loss of “stem-ness” qualities, such as intrinsic drug and radiation resistance, providing another mechanism by which angiosuppression would be a chemosensitizer or radiosensitizer. In a rat glioma model, the combination of targeted antiangiogenic therapy and cytotoxic chemotherapy significantly reduced the fraction of tumor sphere-forming units (a measure of stem cell activity), whereas neither modality tested alone was effective. These findings explain why bevacizumab, in combination with chemotherapy, could be more effective than antiangiogenic monotherapy for many types of human cancers; also, it explains in part why it may not be necessary to have a significant reduction in tumor volume to achieve an increase in survival, if the stem-cell vascular niche is selectively vulnerable. If indeed the SCLGCs are the cellular target of bevacizumab, then the existence of angiogenesis-independent but invasive clones of glioma cells can explain the shrinkage of vascularized tumors and the emergence of highly invasive glioma cells. Engineering models of the tumor microenvironment can provide a discovery platform for the relationship of stem cells to the endothelium and enable drug screening for preclinical testing and precision medicine.
Furthermore, the close interrelationship between glioma stem cells and the vasculature has been underscored by the work of four independent groups showing that glioma stem cells can contribute to angiogenesis by differentiating into endothelial cells or pericytes, a phenomenon best termed vascular mimicry (see Fig. 140.5D ). , This work is important because it indicates that the progeny of glioma stem cells is not limited to neuroepithelial lineage. Using human, neurosurgical specimens, these investigators , found that endothelial cells lining tumor vessels contained the same genetic alterations (e.g., amplification of EGFR or mutation of TP53 ) found in the adjacent malignant glioma cells, presaging poor prognosis. Taken together, these studies point to the use of stem cell–driven angiogenesis as a target for innovative approaches to the treatment of glioblastoma ; the disruption of vascular mimicry, either through the specific targeting of tumor-derived endothelial cells , or through pericytes, slows tumor growth.
An important mechanism, often overlooked, is the potential benefit of anti-VEGF angiotherapy on the immune response to glioblastoma. Cell-mediated immunity is a key host response to human brain tumors with broad therapeutic implications , , , ; however, the normal antitumor immune response is insufficient to eradicate malignant gliomas, which could be achieved if sufficient immune effector cells reached the tumor bed. Immune escape is a critical gateway to malignancy that affects tumor dormancy, progression, invasion, and ultimately, clinical outcomes. The connections between signaling pathways that control immune tolerance and those controlling proliferation, invasion, and angiogenesis remain largely unexplored, offering a fertile area for scientific investigation, , , particularly as immunotherapies are emerging as promising new approaches for treatment of glioblastoma. , ,
The immune response to brain tumors is normally suppressed, mediated by cytokines such as TGF-β, STAT3, granulocyte-macrophage colony-stimulating factor (GM-CSF), IL-6, and IL-10 and a profound suppression of T4-helper lymphocytes. , Standard therapies, such as corticosteroids, radiation therapy, and chemotherapy, further suppress the immune response, lowering the CD4 counts. Normally the endothelium-astrocyte complex abets immune escape by sequestration of antigens; VEGF blockade, however, normalizes vascular permeability and enhances perfusion, thereby exposing the antigens to blood-borne immune effector cells. ,
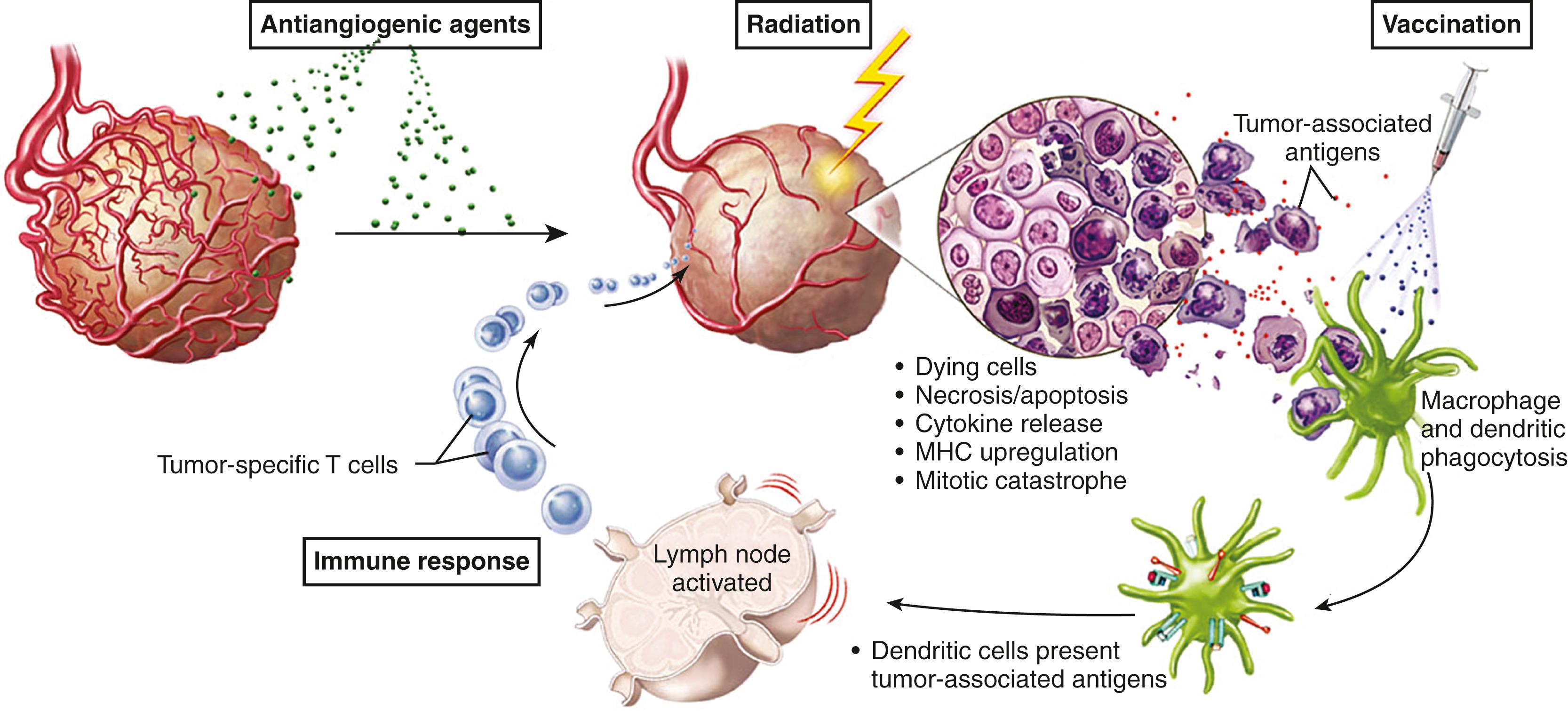
VEGF also contributes to immune escape of tumors by blocking dendritic cell (DC) differentiation and function. An antibody to VEGF, combined with DC-based immunotherapy, results in a prolonged and enhanced antitumor effect. The inhibitory function of VEGF on DC function is most likely mediated by the VEGF receptor Flt-1. , Bevacizumab, given to cancer patients, has the beneficial effects of (1) increasing the DC population of peripheral blood, (2) enhancing the allostimulatory capacity of DC and T-cell proliferation against recall antigens, and (3) decreasing the accumulation of immature myeloid cell progenitor cells. Because of the promise of current DC vaccines for patients with glioblastoma, , there is a compelling rationale for the combination of immunotherapy with anti-VEGF angiotherapy and radiation therapy (see Fig. 140.9 ). Vascular normalization can reprogram the tumor microenvironment from immunosuppressive to immunosupportive. ,
A major cause of morbidity and mortality from brain tumors is the peritumoral, vasogenic edema that can cause mass effect and midline shift and is responsible for much of the morbidity and mortality of brain tumors. The discovery of high-dose corticosteroids over 55 years ago is regarded as one of the great advances in neurosurgery and a revolution in brain tumor management. Although corticosteroids truly are key to controlling peritumoral edema, the long-term toxicities are problematic, and include Cushing syndrome; mood swings; weight gain; fluid retention; tremor; myopathy; immunosuppression with increased risk of infections such as Pneumocystis pneumonitis; hyperglycemia and insulin resistance; osteoporosis and pathologic fractures; capillary fragility; and bowel perforation. , In addition, corticosteroids counteract in vitro the effect of temozolomide and in vivo increase the expression and activity of multidrug resistance transporter proteins, thereby adding to the general resistance of the blood-brain barrier to cytotoxic chemotherapy. Furthermore, in a retrospective analysis of patients with newly diagnosed glioblastoma undergoing radiation therapy, dexamethasone use was associated with a worse prognosis unless bevacizumab was administered. The clinical and biologic shortcomings of dexamethasone offer an opportunity for a new class of vascular targeting agents, including bevacizumab, to be incorporated in the treatment of glioblastoma. Consequently there has been a search for less toxic alternatives to corticosteroids that could be used for long-term control of vasogenic edema. Because VEGF is a potent inducer of hyperpermeability, key advantages of anti-VEGF therapy are to normalize the blood-brain barrier; alleviate peritumoral vasogenic edema; and lessen the need for corticosteroids. In animal models, bevacizumab produces a rapid decrease in peritumoral edema, detected with dynamic MRI, and is more effective than dexamethasone, a drug that has serious negative consequences for immunosuppression and immunotherapy.
The majority of patients treated with bevacizumab demonstrate approximately 50% reduction in corticosteroid dose. , In the BRAIN trial, there was alleviation of edema in 50% of the patients receiving bevacizumab, and more than half of these patients were able to reduce the dosage of corticosteroids by 59%. , , The reduction in vasogenic edema is apparent on FLAIR MRI after one or two doses of bevacizumab , and apparent after the first dose of other antiangiogenic agents (e.g., cediranib) using sophisticated neuroimaging techniques to detect vascular normalization ( Fig. 140.10 ). The steroid-sparing effect of cediranib persists until the drug is discontinued because of tumor progression.
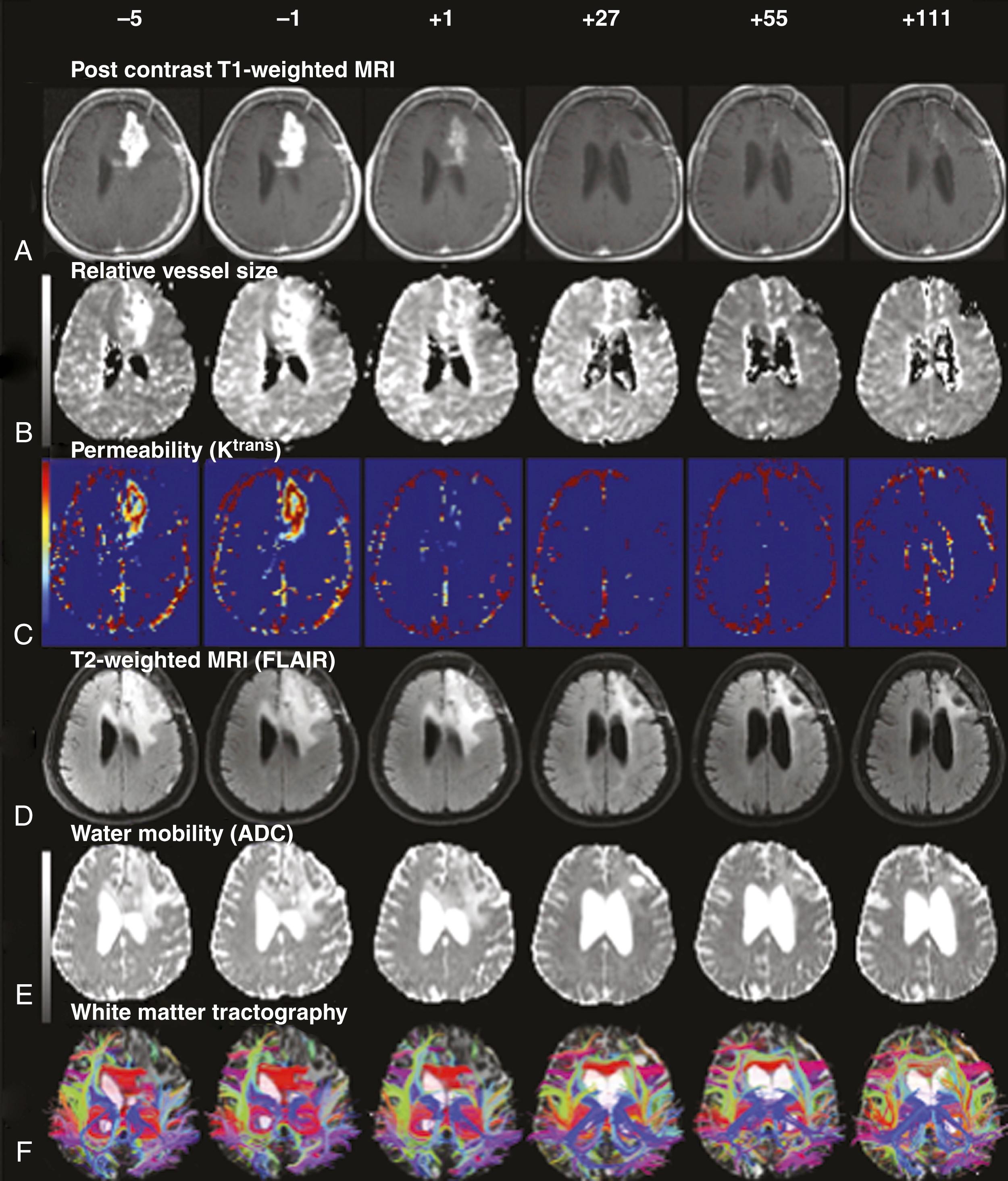
The clinical toxicity of radiation therapy of brain tumors includes radiation-induced necrosis, with peritumoral vasogenic edema. Heretofore, the mainstay of therapy has been corticosteroids, with surgical decompression if the patient became steroid dependent or steroid toxic, or if there were symptoms of mass effect despite corticosteroids. Treatment with hyperbaric oxygen, vitamin E, and other antioxidants yields only modest benefits. The molecular response of malignant cells to ionizing radiation is the secretion of VEGF and metalloproteases, which then promote angiogenesis and tumor invasion. , The multiple immunosuppressive forces in the tumor microenvironment represent an important barrier to successful tumor elimination ; local irradiation can transform tumors into in situ vaccines, with the potential to ignite tumor immune recognition by generating neoantigens.
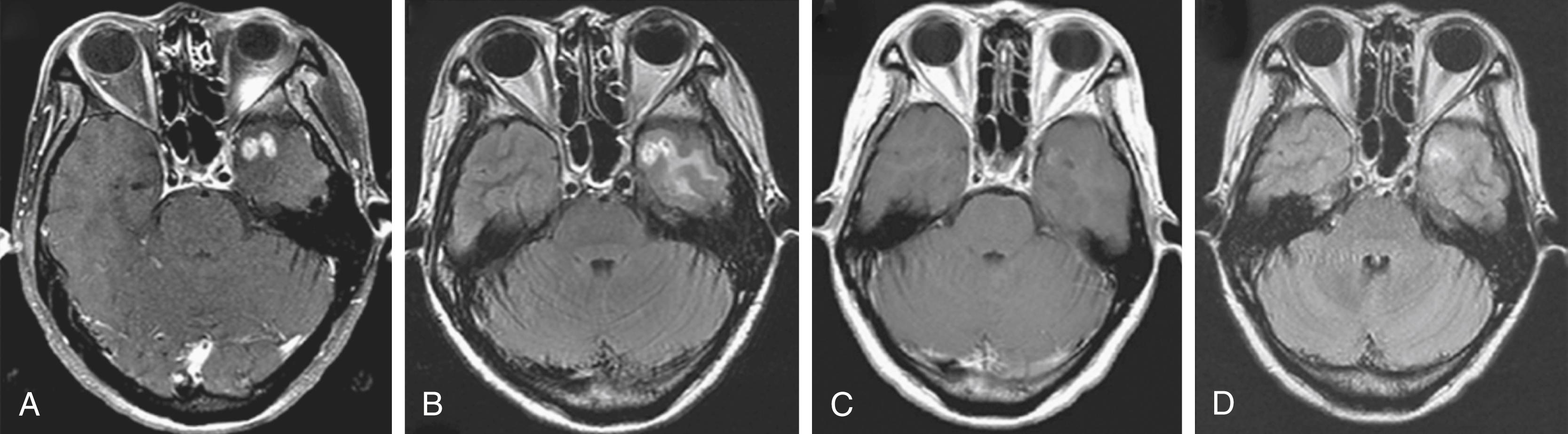
Bevacizumab has been discovered to provide a consistent benefit in patients with radiation-induced necrosis, , with a striking 50% to 60% reduction in vasogenic edema, detected on FLAIR images, and a 50% to 80% reduction in the size of the necrotic area, revealed by contrast enhancement ( Fig. 140.11 ), leading to a substantial reduction in the daily dexamethasone requirement and neurocognitive improvement. To treat radiation necrosis, the median number of cycles of bevacizumab is four (range, one to eight cycles); the average dosage is 7.5 mg/kg, and the time between cycles is 2 weeks. Bevacizumab is equally effective whether the radiation necrosis is induced by stereotactic radiosurgery or by fractionated radiotherapy.
Angiotherapy, by normalizing the vasculature, can serve not only as a radioprotectant but also as a radiosensitizer. Because of strong preclinical evidence showing synergistic response to radiation therapy when angiogenesis, particularly VEGF signaling, is inhibited, , , , Gutin and associates evaluated the safety and efficacy of bevacizumab with hypofractionated stereotactic irradiation in patients with recurrent malignant glioma. These patients had already received standard radiation therapy (approximately 60 Gy) and were given an additional 30 Gy in five fractions while receiving concurrent bevacizumab. The additional irradiation was well tolerated without any instances of radiation-induced necrosis, possibly a protective anti-VEGF effect of bevacizumab. The 6M-PFS was 65%, the median OS was 12.5 months, and the 1-year survival was 54%. Bevacizumab combined with hypofractionated stereotactic irradiation was well tolerated and showed evidence of activity. Ongoing randomized trials with more moderate radiotherapy dosing may help establish the efficacy of this regimen, although nuances of the approach, including patient selection, radiation target volume, and dose optimization require further assessment. In a multicenter, phase 1 trial, reirradiation using a three-fraction schedule with bevacizumab was found to be feasible and well tolerated with dose-escalation up to 11 Gy × 3, achieving a near doubling in the delivered biologic equivalent dose to normal brain, in comparison to the previous 6 Gy × 5 schedule, with a promising OS in the intention-to-treat population of 13 months. Although no symptomatic radionecrosis was observed, substantial treatment-related effects and necrosis were observed in resected specimens.
Omuro and Gutin extended their work to newly diagnosed glioblastoma, combining bevacizumab, temozolomide, and hypofractionated stereotactic radiotherapy (HSRT) for newly diagnosed glioblastoma. Patients with tumor volumes <60 cm 3 were treated with HSFRT, adjuvant temozolomide, and bevacizumab at standard doses. In 40 patients the PFS was 10 months, without pseudoprogression. An objective response rate was 57%; the OS was comparable to that in historical controls. In two prospective phase 2 trials with small patient numbers, the addition of bevacizumab in patients with newly diagnosed glioblastoma receiving hypofractionated intensity-modulated radiotherapy (IMRT) and temozolomide did not enhance OS, but there was a modest prolongation of PFS.
Anti-VEGF angiotherapy is also being explored in combination with stereotactic radiosurgery. Preliminary studies indicate that for selected patients with heavily treated, recurrent brain metastases, complicated by radiation-induced necrosis, bevacizumab provides a clear radiographic response as well as neurological palliation. In a small trial of 24 patients with recurrent brain metastases, low-dose bevacizumab (5 mg/kg every 3 weeks) was given for four cycles, and the patients underwent a CyberKnife procedure (two to five fractions), with favorable outcomes: 1-year OS was 87.5%, the positive imaging response rate was 96%, the median T2 volume reduction was 14 cm 3 , with significant improvement in Karnofsky performance scale (KPS) scores, suggesting that further studies are warranted. In a large, multicenter, retrospective analysis of glioblastoma patient cohorts, the use of corticosteroids to control edema was linked to a shorter OS, in contrast to bevacizumab, which has biologic and radiologic effects but minimal effect on OS, highlighting the need for a next generation of antiedema agents. In a mouse model, a gene signature was used to characterize the effects of dexamethasone on tumor cell proliferation and cell death, with a murine anti-VEGFA antibody in parallel as an alternative for edema control. The dexamethasone-associated gene expression signature correlated with shorter survival in The Cancer Genome Atlas (TCGA). In the animal model the anti-VEGFA antibody (comparable to bevacizumab) did not affect survival, but did decrease glioma cell proliferation and increased glioma cell death, whereas dexamethasone pretreatment decreased tumor cell proliferation but reduced survival when combined with radiotherapy ; taken together, the study highlights the importance of identifying alternative agents such as VEGF antagonists for managing edema in glioblastoma patients, arguing for prudent and restricted use of corticosteroids in patients with glioblastoma. ,
Despite the initial promising response observed with the addition of HSRT to bevacizumab as salvage treatment for recurrent malignant glioma, approximately one-half of patients ultimately experience failure within the radiation field. The rate of local failure appears lower than that with bevacizumab alone in the recurrent setting, but underscores the radioresistance of malignant glioma and the need for a better understanding of the role of the glioma microvasculature to radiation resistance. For example, the response to a single high dose of radiation (10 Gy) in a laboratory model is an upregulation of VEGF by the normal brain as a response to radiation injury. Temozolomide is clearly effective in the treatment of newly diagnosed glioblastoma, in part because it acts as a radiosensitizer. Temozolomide also suppresses angiogenic pathways; in animal models, bevacizumab increases the antitumor efficacy of temozolomide, supporting the rationale for combining temozolomide with bevacizumab in patients with glioma. In glioblastoma xenograft models, addition of bevacizumab to temozolomide showed a significant antitumor effect only in the IDH1-mutant tumors. However, in a randomized, open-label, phase 2 trial, the TAVAREC trial (Bevacizumab in Recurrent Grade II and III Glioma), in patients with World Health Organization (WHO) grade II or III glioma, and without 1p/19q co-deletion, there was no survival benefit to adding bevacizumab to temozolomide compared with temozolomide monotherapy. In a large phase 3 trial, CheckMate 143 (A Study of the Effectiveness and Safety of Nivolumab Compared to Bevacizumab and of Nivolumab With or Without Ipilimumab in Glioblastoma Patients), for recurrent glioblastoma, bevacizumab (FDA approved) was compared with the immune checkpoint inhibitor (PD-1 blockade), nivolumab. There was no statistical difference in outcome measures between bevacizumab and nivolumab, as single-agent therapy, in terms of OS or treatment-related adverse events. The objective tumor response was higher with bevacizumab (23.1%) versus nivolumab (7.8%); both agents were well tolerated without any unexpected neurological adverse events or deaths due to the drugs.
Currently, there is FDA approval, and NCCN endorsement, for use in the treatment of recurrent glioblastoma. Practical guidelines and caveats are summarized in Box 140.1 . Additional information can be found online or in the package insert. Bevacizumab is generally given at a dosage of 10 mg/kg every 14 days on a 28-day cycle; two infusions constitute one cycle. The maximal tolerable dose is approximately 15 to 20 mg/kg; the optimal biologic dose is unknown. Use of the maximum tolerated dose as opposed to the optimal biologic dose of antiangiogenic therapy is the approach currently used for patients. There are numerous completed and ongoing clinical trials combining antiangiogenic therapy with chemotherapy and molecularly targeted agents. These studies have failed to demonstrate improved outcomes compared with the antiangiogenic agent alone. Standard cytotoxic chemotherapy, including temozolomide, when given in low-dose, continuous (metronomic) fashion is also angiosuppressive. , Metronomic chemotherapy acts directly on the mature endothelial cells within the tumor neovasculature and inhibits the mobilization of circulating endothelial progenitor cells. Accumulating evidence suggests that the efficacy of such treatment may not rely only on antiangiogenic activity. Potential new mechanisms of action, such as restoration of anticancer immune response and induction of tumor dormancy, are relatively new approaches. A new class of repurposed, readily available, and relatively inexpensive drugs (e.g., metformin, celecoxib, valproic acid) are antiangiogenic and show clinical promise.
Bevacizumab provides “pharmacologic debulking” with volumetric reduction in the majority of patients with recurrent malignant glioma as measured by >50% reduction in tumor size (partial response; Macdonald Criteria), which translates to a significant increase in 6-month progression-free survival of >50%, superior to historical controls. However, the increase in the 6-month PFS, also observed for newly diagnosed glioblastoma, does not translate to a significant increase in overall survival. Bevacizumab could increase overall survival in patients with specific genotypes (e.g., proneural).
Bevacizumab can be given long term, for several years. It must be discontinued, as all drugs, if major toxicity (National Cancer Institute [NCI]–Common Terminology Criteria [CTC] grade 3 or 4) occurs. Discontinuation does not appear to cause a “rebound” in angiogenesis and tumor growth, based on preclinical models. There are no clear “stop” rules in place. If a patient does not have a clinical or radiographic response after one cycle, it is unlikely that vascular endothelial growth factor (VEGF) inhibition is being achieved, or it is likely that angiogenesis is driven by another (non-VEGF) angiogenic factor, and therefore long-term benefit of tumor control with bevacizumab is unlikely.
Numerous second-generation angiosuppressive agents are under evaluation. , , Currently, none has a superior radiographic response to bevacizumab—the prototype for angiotherapy, analogous to penicillin at the dawn of the era of antibiotics. As with antibiotics, future generations of antiangiogenics could (1) be used in combinations; (2) be used for a greater range of tumors (e.g., acoustic neuromas); (3) overcome problems of VEGF-escape/resistance; and (4) be more cost-effective.
The majority of patients treated with bevacizumab show a significant decrease in vasogenic edema, often rapidly, with reduced need for corticosteroids. VEGF increases capillary permeability; inhibition of VEGF restores the blood-brain barrier, decreasing edema and contrast enhancement, thereby decreasing mass effect but also obscuring glioma visualization.
The commissioner of the FDA declared antiangiogenesis as a fourth mode of treatment for human cancer (in addition to surgery, chemotherapy, and radiation therapy). The FDA approved several angiogenic inhibitors for treatment of human cancer. Bevacizumab is currently approved for (1) recurrent glioblastoma; (2) advanced non–small cell lung cancer (NSCLC); (3) metastatic renal cell cancer; (4) advanced cervical cancer; and (5) platinum-resistant ovarian cancer ( http://www.avastin.com ).
The NCCN, based on available data, endorses bevacizumab + irinotecan combination, or bevacizumab as a single agent, in the treatment of recurrent glioblastoma; it can also be given for anaplastic gliomas and for recurrent glioblastoma in combination with another agent, such as temozolomide, irinotecan, carmustine or lomustine, or carboplatin (Evidence level 2B).
The cost of bevacizumab, and other molecular targeted agents, is problematic, especially in the current era of the Affordable Care Act. Medicare agreed to consider payments to cover the costs of bevacizumab for patients with recurrent glioblastoma. For the individual patient, the cost can be a significant barrier to acquiring the drug.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here