Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
We would first like to apologize to the many authors whose studies were excluded due to space constraints. We would like to thank Dr. Andrea Portbury and Chelsea Cyr for critical reading of the manuscript.
The heart begins simply as a bilateral field within the lateral plate mesoderm ( Figure 1.1 , Table 1.1 ). As the early embryo undergoes formation of the gut-tube, these bilateral fields migrate toward the midline, where the cranial-most aspect of the fields will fuse ventrally to form the outer curvature of the heart tube. These fields will continue to migrate together, with more of the heart fields contributing to the forming tube, until the dorsal aspect of the heart fields fuse to form a closed tube. The initial contributors to the heart tube are known as the first heart field. The first heart field gives rise to the left ventricle, with some contributions to the atria and the right ventricle. Additional heart field progenitors from the lateral plate mesoderm continue to add to the arterial and venous poles of the heart tube; these later-adding cells are known as the second heart field. The second heart field gives rise to most of the right ventricle and atria, the most distal myocardium that surrounds the aorta and pulmonary artery, and the most proximal smooth muscle that contributes to the tunica media of the great arteries.

| Milestone | Human | Mouse | Chick | Xenopous | Zebrafish |
|---|---|---|---|---|---|
| Heart is specified | E7.25 | HH 3 | Stage 15 | 8 somites | |
| Heart tube forms | ∼CS 9/10 (2 mm) | E8 | HH 9 | Stage 33 | 24 hpf |
| Heart displays regular contractions | ∼CS 9/10 | E8 | HH 7 ∗ | Stage 35 | 24 hpf |
| Heart begins looping ∗ | CS 13 | E8.5 | HH 9+/10- | Stage 35 | 30 hpf |
| AV cushions begin forming | CS 17 | E9.5 | HH 12/13- | Stage 44 | 48 hpf |
| OFT cushions begin forming | CS 15/16 | E10.5 | HH 12/13- | Stage 39/40 | – |
| Outflow tract is septated | CS 17 | E13.5 | HH 34 | – | – |
| Atria are septated | CS 18 † | E14.5 | HH 46 | Stage 46 | – |
| Ventricles are septated | CS 22 | E13.5 | HH 34 | – | – |
∗ Because the heart tube begins forming as a trough that then closes dorsally, contractions are observed prior to the pinching off of the fully formed heart tube.
† The foramen ovale is still open at this stage. This fenestration is closed at the stages listed for the other species. CS, Carnegie stage ; E, embryonic day; HH, Hamburger-Hamilton stage ; Stage, Nieuwkoop and Faber stage ; hpf, hours post-fertilization. See cited references for more details.
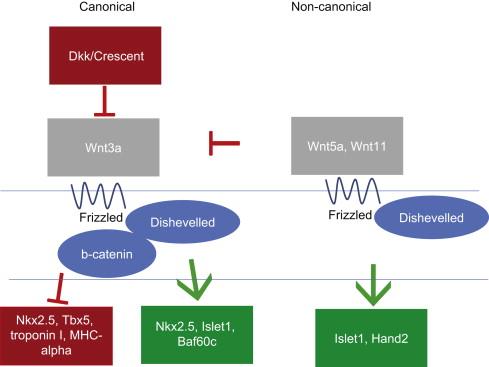
The Wnt family includes the canonical pathway, the non-canonical pathway (also known as the planar cell polarity pathway), and the Wnt/calcium pathway ( Figure 1.2 ). Both the canonical and non-canonical pathways have well-established roles in heart field specification. Temporal waves of canonical and non-canonical Wnt signaling play distinct roles during cardiac specification and morphogenesis. As the heart field forms from the primitive streak, Wnt3a is expressed in the primitive streak and serves as a repulsive cue to the forming heart field. Experiments performed in Xenopus , due to its ease of manipulation and genetic tractability, have shown that the early repression of Wnt signaling in the Xenopus animal cap (i.e., in the ectodermal roof of the blastocele prior to heart field specification) via Dkk-1 and Crescent is necessary for initiation of transcription of cardiac transcription factors Nkx2.5 and Tbx5 and myocardial-specific proteins troponin I and myosin heavy chain α. However, later in cardiac development, canonical Wnt signaling in embryonic mice at E8.75 promotes Nkx2.5, Islet1, and Baf60c within the entire heart. Due to the genetic similarity between Xenopus and mouse, these differences likely reflect different temporal requirements for Wnt signaling as opposed to species-specific differences. In the second heart field, non-canonical Wnts 5a and 11, which act through the non-canonical planar cell polarity pathway, are expressed slightly later in development and co-operatively repress the canonical Wnt pathway while also promoting expression of Islet1 and Hand2, whose expression serves to ‘mark’ the heart field; as such, these genes are commonly referred to as cardiac markers. Both the repression of the canonical Wnts and the induction of the heart field markers require β-catenin in the second heart field. Wnt5a and Wnt11 also promote proliferation within this progenitor region. After the heart tube forms, Wnt-stabilized β-catenin is necessary in the Islet1-expressing second heart field cells to maintain their progenitor status. Loss of either β-catenin or Wnt signaling in the second heart field leads to second heart field defects, including right ventricular and outflow tract defects. Even if Wnt signaling is lost under cells expressing one of the first markers of differentiated cardiomyocytes, Mesp1, second heart field proliferation is decreased, and Islet-1 expression is down-regulated. Conversely, overexpressing β-catenin under the Mesp1 promoter expands the Islet-1-positive second heart field and promotes proliferation. Later, Wnt5a specifically acts upstream of the disheveled/planar cell polarity pathway to regulate the addition of the second heart field to the arterial pole. In addition, Wnt signaling also promotes bone morphogenetic protein (BMP) 4 and the non-canonical Wnt 11, which promote myocardial differentiation. Thus, the Wnt pathway is critical for inducing heart field formation, maintaining progenitor status and promoting myocardial differentiation.
One of the earliest required signaling pathways is the retinoic acid pathway. RALDH2, the enzyme that synthesizes retinoic acid, is restricted within the lateral plate mesoderm to a region nearest to the heart field. Expression of RALDH2 progresses in a cranial–caudal direction during heart field induction and heart tube formation and establishes the posterior boundary of the heart field. Within the lateral plate mesoderm, retinoic acid plays an inhibitory role, where it acts both directly and indirectly to restrict cardiac transcription factors Nkx2.5 and FoxF1 to the anterior lateral plate mesoderm, Hand1 to the anterior and middle of the lateral plate mesoderm, and Sal1 to the posterior lateral plate mesoderm. Retinoic acid further represses GATAs 4, 5, and 6. This inhibitory role is required to limit the size of the heart field, and fate-mapping studies in the zebrafish, another experimental model that is valued for its genetic similarity to the mouse and ease of studying embryonic development, have demonstrated that zebrafish embryos with decreased levels of retinoic acid exhibit an increased number of Nkx2.5-positive cells. Conversely, exposing either zebrafish or Xenopus embryos to increasing levels of retinoic acid specifically leads to a reduced number of cardiomyocytes. In chick embryos, which physically develop more similar to humans as compared with mice but lack the ability to manipulate the genome as in mice, antagonizing retinoic acid signaling promotes the ventricular myocardial fate at the expense of the atria. Together, these studies suggest that retinoic acid plays two major roles in the early heart field. First, retinoic acid generally restricts the expression of heart field markers to limit the size of the heart field. Then, it specifically promotes a ‘posterior’ fate within the heart field, which affects the venous precursors that will give rise to the atria.
Several well-known cardiogenic transcription factors are expressed in the early heart fields and play a role in driving lateral plate mesoderm to a cardiac fate. In the first heart field these transcription factors include but are not limited to Nkx2-5; GATAs 4, 5, and 6; and Tbx5. The transcription factor kernel required for driving mesoderm to myocardium has been minimally defined as the transcription factors GATA4 and Tbx5 and the chromatin remodeling subunit Baf60c. Furthermore, Tbx5 genetically interacts with Baf60c in cardiac morphogenesis. These findings have begun to elucidate the interplay between chromatin remodeling factors and cardiogenic transcription factors during the progressive differentiation of mesoderm to cardiac progenitor to cardiomyocyte.
The homeobox transcription factor Nkx2.5 is perhaps the best-known cardiac inducer. Nkx2.5 is expressed in the first and second heart fields. During normal development, Nkx2.5 becomes turned off in the first heart field, allowing these cells to begin differentiating while maintaining the second heart field in a progenitor state. During this process, Nkx2.5 represses the transcription of numerous cardiac progenitor markers, including Islet1, Mef2c, and Tgfβ2. In addition to repressing cardiac progenitor markers, Nkx2.5 also represses the BMP and fibroblast growth factor (FGF) pathways, which promote differentiation into myocardium and inhibit proliferation. This field of cells is the first to differentiate into functional myocardium and express muscle-specific markers such as the myosin light chain protein MLC3F while concomitantly down-regulating cardiac progenitor transcription factors such as Islet1. In the later differentiating second heart field, these pathways must be down-regulated to allow for sufficient proliferation within the progenitor pool, and proliferation in this region is essential for normal development of the arterial pole. In addition, Mef2c is required to allow the transition from specification to myocardial differentiation.
A severe and uniformly lethal heart defect associated with the early heart fields is a failure of the bilateral heart fields to migrate and fuse at the midline, leading to two separate hearts, a condition known as cardia bifida. Though the previously mentioned signaling pathways and transcription factors are crucial for heart formation, loss of only a few specific factors can result in cardia bifida. These factors are described in detail, and Table 1.2 provides the Online Mendelian Inheritance of Man entry numbers if information on available genetic testing for these genes (as well as additional CHD-related genes presented later in the chapter) is of further interest.
| Developmental Process | Transcription Factors and Genes Involved | Related Human Diseases | OMIM Entry # |
|---|---|---|---|
| Heart field specification and heart tube formation | GATA4 Smad6 Nodal |
Cardia bifida | 600576 602931 ∗ 601265 |
| Establishment of laterality | Pitx2 FoxC1 |
Axenfeld-Rieger syndrome | 601542 601090 |
| Conduction system | Pitx2c | Atrial fibrillation | 601542 |
| Valve formation | Fibrillin-1 Elastin Fibulin-4 TGFβ |
Mitral valve prolapse | 134797 130160 604633 190180 |
| Outflow tract formation | Tbx1 Lhx2 Tcf21 β-catenin Neuropilin-1 |
DiGeorge syndrome | 602054 603759 ∗ 603306 ∗ 116806 602069 ∗ |
| Chamber septation | GATA4 Nkx2.5 Tbx20 Tbx5 |
Atrial septal defects | 600576 600584 606061 601620 |
Loss of GATA4 is perhaps the best-known cause of cardia bifida. The GATA4 homozygous null mouse embryo fails to form a heart tube. Furthermore, siRNA knockdown of GATA4 in the chick heart fields results in the formation of bilateral heart tubes, and this effect is caused by a down-regulation of N-cadherin. Similarly, blocking N-cadherin in chick embryos inhibits fusion of the heart fields but does not halt heart development, resulting in bilateral heart tubes. However, although the N-cadherin homozygous null embryo displays heart failure, as evidenced by pericardial effusion, a single heart tube is present. Thus, species-specific differences in the cadherins may underlie the contribution of N-cadherin to GATA4-mediated cardia bifida.
Loss of BMP or Nodal signaling can also result in cardia bifida, and this effect may also occur through the GATA transcription factors. In Xenopus , loss of BMP signaling through injection of mRNA encoding the BMP antagonist Smad6 or a mutated truncated BMP receptor leads to cardia bifida. In these embryos, the heart fields are also specified but fail to fuse at the midline. Consistent with BMP’s previously mentioned role in promoting myocardial differentiation, these embryos also show fewer cardiomyocytes compared with untreated control embryos. Similarly, the zebrafish swirl and one-eyed pinhead mutants, which lack Bmp2b and Cripto, respectively, display reduced Nkx2.5 expression, which can be rescued by the ectopic expression of GATA5. Of these two zebrafish mutants, only the one-eyed pinhead mutant presents with cardia bifida though, and genetic redundancy in the zebrafish may explain why the zebrafish swirl mutant has a less severe phenotype than the Smad6-injected Xenopus embryo. Perhaps unsurprisingly, cardia bifida is almost always incompatible with life. One case report, however, has described an infant who survived with cardia bifida in which each half heart showed characteristics of both ventricles but a single atrium. Each half heart also had its own truncus and sinus venosus, which may have been sufficient in the intrauterine environment.
The human body is asymmetric about the vertical midline, and the heart is among the visceral organs with left/right asymmetry. Left/right asymmetry is established early during embryonic development; left/right gene expression differences are observed in the lateral plate mesoderm from which cardiac progenitors emanate. The molecular mechanisms that dictate the left/right asymmetry of the heart during its morphogenesis are unknown; however, the mechanisms that underlie the initial asymmetry-breaking events in the early embryo have been described.
The cilium, a subcellular organelle that protrudes from the cellular surface, plays an important role in left/right determination. In the gastrulating embryo, rotating cilia in Hensen’s node generate leftward flow that breaks left/right symmetry and establishes differential signaling and gene expression on the left and right sides of the embryo. In the heart field, the cilia initially express FGF receptors. These receptors respond to FGF, which induces them to move Sonic hedgehog and retinoic acid leftward, which results in the release of calcium. Despite this purported leftward movement, though, the asymmetrical expression of Sonic hedgehog and retinoic acid is controversial, with some but not all studies showing asymmetrical expression in the node. These signaling events limit the expression of Nodal, Pitx2c, and Lefty2 to the left side of the embryo. The side-specific expression of these transcriptional regulators activates a gene regulatory network on the left distinct from that on the right, causing sidedness of morphogenesis. The link from the left/right determination transcriptional kernel to the cardiac progenitor transcriptional kernel may be direct. Nkx2.5, a progenitor transcription factor, interacts with the N-terminal end of transcription factor Pitx2c, part of the left-determination transcriptional network. This specific interaction is required for normal heart looping, and antagonizing Pitx2c in the left side of the heart, where it is normally expressed, leads to randomized heart looping.
Cardiac left/right asymmetry is first noticeable during cardiac looping, during which the ventricles undergo characteristic D-handed looping with great fidelity. This rightward looping displaces the ventral midline of the heart towards the right and establishes the outer curvature of the heart tube. The formerly dorsal midline is then rotated to become the inner curvature of the heart tube. From this looped position, the outflow and inflow tracts converge, and the ventricular bend becomes displaced ventrally. Finally, as the outflow tract is septated to form the aorta and pulmonary artery, the aorta is wedged between the pulmonary artery and the atrioventricular valves.
Left/right determination can go awry in distinct ways with predictable results. The entire cascade can be inverted, resulting in normal but mirror image development of the embryo and heart. Consistent with this hypothesis, the inv mutant mice display situs inversus, and in this model, Pitx2c is selectively expressed on the right side instead of the left.
The left-sided program can be disrupted, resulting in hearts with both sides developing as ‘right,’ or right isomerism. Work in mouse embryos suggests that Pitx2c is required for turning off the ‘right-sided’ developmental program on the left side. In Pitx2c homozygous null embryos, both atria display right-sided structures. Within the mature heart, left-sided structures include the left atrium with pulmonary veins, whereas right-sided structures include the right atrium with sinoatrial node. In Pitx2c mutant embryos, the sinoatrial node is duplicated on both sides, and valve and septation defects consistent with right atrial isomerism are observed. The requirement for Pitx2c in structural morphogenesis of the heart may come after heart looping: removing Pitx2c specifically from differentiated myocardium causes right atrial isomerism, including an atrial septal defect and the duplicated sinoatrial node. Thus, Pitx2c is required in the mesoderm for left/right determination and later in the myocardium to repress the ‘right-sided’ developmental program, including sinoatrial node formation within the left atrium.
The right-sided program can also be disrupted, resulting in hearts developing with two ‘left’ sides or left atrial isomerism. Left atrial isomerism is observed in embryos lacking Sonic hedgehog. Sonic hedgehog null mice display isomerism of the left atrial appendages and other malformations that are frequently observed in humans with left isomerism. In these embryos, Pitx2c expression is observed bilaterally, consistent with the instructive role of this transcription factor for left-sided structures. Consistent with the Pitx2c expression pattern in the Sonic hedgehog null mouse, the heart fails to loop in mice lacking hedgehog signaling. Finally, the left/right determination process can be randomized. Mice that lack cilia show randomized expression of left/right determination transcription factors, sidedness, cardiac looping, and sidedness of specific cardiac structures.
Defects in left/right determination are common in humans and are grouped into the heterotaxy syndrome. The observed cardiovascular phenotypes of humans with this syndrome are broad, as predicted from the above discussion. The molecular genetics of heterotaxy syndrome are under current investigation, and rapid progress is being made given the large number of predicted candidate genes identified from animal model studies. In particular and consistent with the importance of cilia in left/right determination, cilia gene mutations have been identified in several heterotaxy syndrome patients, causing a range of cardiac structural abnormalities from situs inversus to left or right isomerisms. Furthermore, proper cardiac looping is required to align the segments of the heart for subsequent septum morphogenesis. Mis-looping would be expected to cause cardiac structural abnormalities in addition to structural isomerisms. Consistent with this hypothesis, cardiac septal defects are commonly observed in patients with heterotaxy syndrome. For example, patients with Axenfeld-Rieger syndrome display a variety of congenital heart defects, including atrial and ventricular septal defects, mitral and tricuspid defects such as prolapse and stenosis, small left ventricular outflow tract, and stenotic or bicuspid aortic and pulmonary valves. An estimated 40–60% of these patients have mutations in Pitx2 or FoxC1.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here